Institute of Oceanology, Chinese Academy of Sciences
Article Information
- DONG Chenfan, WU Haiyan, PENG Jixing, GUO Mengmeng, ZHAI Yuxiu, TAN Zhijun
- Thermal processing induced release and degradation of paralytic shellfish toxin from mussels Mytilus edulis
- Journal of Oceanology and Limnology, 40(6): 2267-2276
- http://dx.doi.org/10.1007/s00343-021-1172-0
Article History
- Received Jun. 22, 2021
- accepted in principle Jul. 19, 2021
- accepted for publication Sep. 15, 2022
2 Key Laboratory of Testing and Evaluation for Aquatic Product Safety and Quality, Ministry of Agriculture; Yellow Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences, Qingdao 266071, China;
3 Pilot National Laboratory for Marine Science and Technology(Qingdao), Qingdao 266237, China;
4 Collaborative Innovation Center of Seafood Deep Processing, Dalian Polytechnic University, Dalian 116034, China
During the May Day holidays in 2016, many media outlets reported that consumers in Qinhuangdao in Hebei Province in China were poisoned after consuming mussels (Mytilus edulis) (Yu and Luo, 2016). An investigation indicated that the causative agent was a paralytic shellfish toxins (PSTs). The mussel samples contained 133.40 mg-STX.2HCl eq/kg saxitoxin (Ding et al., 2017), which is more than 166 times the Codex Alimentarius standard (2015) of 0.8 mg STX.2HCl eq/kg. Risk of such exposure is a major problem in China, as shellfish culture constitutes more than 75% of the mariculture production in China, and more than 15 million tons are produced annually (Ministry of Agriculture and Rural Affairs of the People's Republic of China, 2017). The scale and output of the mariculture industry in China rank first worldwide, and most of the fresh live shellfish cultured in China is sold domestically.
Paralytic shellfish poisoning (PSP) is one of the most widely distributed and most harmful type of lethal shellfish toxin poisoning worldwide (Halstead and Schantz, 1984; Ochi, 2021). PSTs are a group of water-soluble sodium channel blockers produced by dinoflagellates and cyanobacteria. There are many analogues, which have complex transformation relationships, as shown in Fig. 1. Toxic dinoflagellates are ingested and concentrated by filter-feeding mollusks, which promote entry of PSTs into the food chain. Mollusk consumption is the traditional exposure pathway for these toxins (Vieites et al., 1999). Symptoms can generally occur within 30 min of consuming contaminated seafood, including tingling sensation of the lips, mouth and tongue, numbness of extremities, nausea, shortness of breath, dizziness, vomiting, headache, dysphagia, and dysarthria (Etheridge, 2010). No effective antidote is currently available.
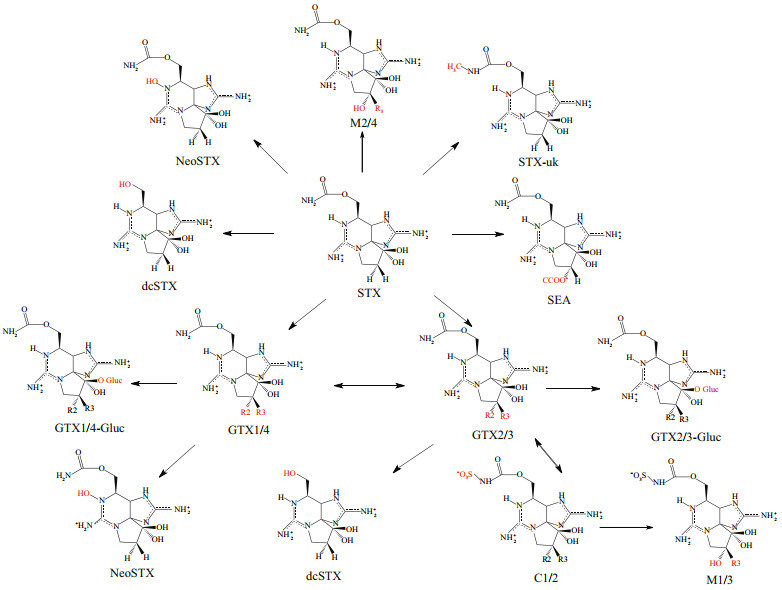
|
| Fig.1 Biotransformation pathways of paralytic shellfish toxins (Wiese et al., 2010) |
Many measures have been implemented to reduce PSTs. Thermal processing is the most common measure which can sharply reduce the total toxicity of PSTs. However, reduction of PSTs cannot be achieved through conventional thermal processing because of the thermal stability of PSTs and it requires harsh alkaline processing conditions. Some studies have reported that bactericidal treatment (115 ℃, 45 min) (Berenguer et al., 1993), pan frying (Indrasena, 1999), and canning (Berenguer et al., 1993) decrease the toxin concentration by more than 70% in mussels. Vieites et al. (1999) stated reduction of toxin concentration in mussel tissue during cooking was a result of toxin transfer to the cooking liquor with more than 50% of toxins removed from the tissue entering the liquor. In addition, some studies have suggested that pH greatly affects the effcacy of thermal processing. Indrasena and Gill (2000) have reported that the higher the pH, the faster the toxin degradation. The purified PST solution was heated at pH 3 for 120 min, and the toxin content remained above 60%, whereas the toxicity decreased to below 10% under neutral conditions (pH 7). Thermal processing in an alkaline environment can significantly decrease the toxin level, as widely supported by research (García et al., 2010).
Traditional cooking is a thermal processing method commonly used by consumers in their daily life, which is the last line of defense to reduce the risk of PSP before consumption. Different cooking methods affect the toxin distribution and total toxicity in different ways. Therefore, the effects of different cooking methods on the fate of PSTs should be evaluated to better understand the risk of consuming toxin-containing shellfish. PST levels in contaminated mollusks such as clams, mussels (Reboreda et al., 2010), and scallops (Wong et al., 2009) can decrease by more than 50% after steaming or boiling. Moreover, the effects of cooking are significantly different for different shellfish and tissues in shellfish. Lawrence et al. (1994) found that steaming or boiling lowers the concentration of PSP in the lobster hepatopancreases by an average of 54%. In contrast, Turnbull et al. (2018) found no change in PST concentration but a reduction in the hepatopancreas volume of lobsters during cooking, resulting in a decrease in the total amount of toxin present. In a study by Wong (2009), the percentage removal of toxins was higher in mussels than in scallops because most of the PSTs (more than 70%) accumulate in the viscera and adductor muscle of raw scallops and thus become more diffcult to remove.
This study was designed to clarify the effects of thermal processing of daily cooking on the removal and degradation of PSTs in M. edulis. In this study, the effects of the five most common cooking methods (steaming, boiling, frying, water bath heating, and microwaving) on the removal of PSTs from M. edulis samples were evaluated by measuring three indices (toxin concentration, percentage removal, and toxin amounts in the cooking liquor and meat). Such scientific and accurate data related to cooking mussels are needed to reduce the risk of PSP to consumers.
2 MATERIAL AND METHOD 2.1 Sample collection and analysisPST-contaminated M. edulis were obtained from regions where a harmful algal bloom (the causative species was Alexandrium) broke out in 2016 in Qinhuangdao, China. M. edulis of the same size (53.0±1.79 mm) and from the same location (39°56.582′N, 119°44.238′E) were selected, cleaned, and stored at -80 ℃. The toxin concentration in the M. edulis was 29.6-mg STX.2HCl eq/kg for the five main toxins (saxitoxin (STX), 11α-hydroxysulfate-neosaxitoxin (GTX1), 11α-hydroxysulfate-saxitoxin (GTX2), 11β-hydroxysulfate-saxitoxin (GTX3), 11β-hydroxysulfate-neosaxitoxin (GTX4)).
Certified reference standards (N21-sulfocarbamoyl-11α-hydroxysulfate-saxitoxin (C1) & N21-sulfocarbamoyl-11β-hydroxysulfate-saxitoxin (C2), decarbamoyl-11α-hydroxysulfate-saxitoxin (dcGTX2) & decarbamoyl-11β-hydroxysulfate-saxitoxin (dcGTX3), GTX2 & 3, GTX1 & 4, N-sulfocarbamoyl-saxitoxin (GTX5), decarbamoyl-saxitoxin (dcSTX), decarbamoyl-neosaxitoxin (dcNEO), STX, and neosaxitoxin (NEO)) purchased from the National Research Council of Canada (Halifax, Nova Scotia, Canada) were used in this study. Water was deionized and passed through a Milli-Q water purification system (Millipore, Billerica, MA, USA).
2.2 Chemical and reagentThe chemicals and reagents used in this experiment were of analytical grade and included methanol, acetonitrile, formic acid (more than 98%), and ammonium acetate (more than 97%). They were purchased from Merck (Darmstadt, Germany). All aqueous solutions were prepared with water purchased from Millipore (Millipore, Billerica, MA, USA) in optigrade quality. Soybean oil (Luhua, Shanghai, China) was used for frying.
2.3 Thermal processingSix experimental groups were established, and 500 g of M. edulis was included in each group. The samples in control group were first shelled with a scalpel (keeping shellfish intact as much as possible to reduce the loss of toxin and water), then meat weight measured and homogenized for measuring toxin concentration and moisture content. A similar shelling process was carried out for the fried group. Other groups were cooked in-shell. The M. edulis of five groups were processed by steaming, boiling, frying, water bath heating. The shellfish were processed in an electric stove (Miji Electronics and Appliances Ltd., Shanghai, China), frying pan (26 cm), steamer pot (26 cm), and microwave oven (Midea, Guangdong, China). The temperature was maintained and measured with a digital meat thermometer (Shenzhen Jiayiyang Electronics Co., Ltd., Shenzhen, China) during processing. The meat and cooking liquor were collected separately for analysis (Fig. 2). The specific test conditions were as follows:
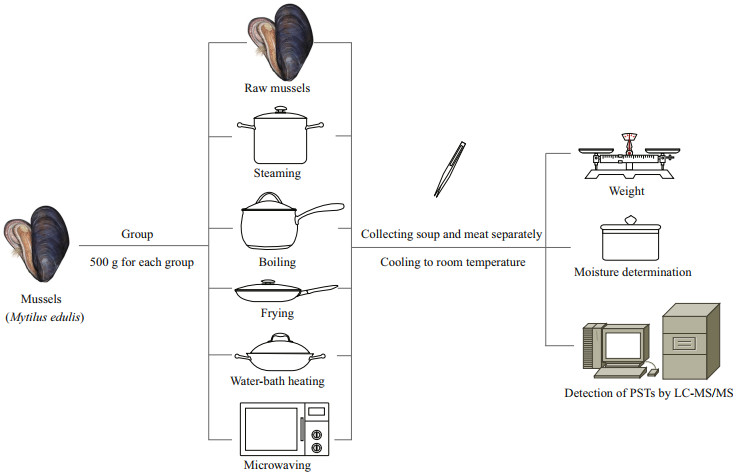
|
| Fig.2 Preparation of shellfish by using different heating methods |
Steaming: 1-L boiling water was added to the steamer, and then a plate of mussels was placed on the upper steamer with a cover for 5, 8, or 10 min. Boiling: 0.5 L of boiling water and mussels were placed in water and heated over medium heat for 5, 8, or 10 min. Frying: 20 mL of oil was heated for 30 s, and then mussels were placed in the pan with a cover for 1, 3, or 5 min. During this period, the mussels were stirred every minute. Water bath heating: the mussels were placed in a beaker covered with preservative film, and heated at 60, 80, or 100 ℃ in preheated water for 5, 8, or 10 min. Water temperature was controlled with an electronic temperature control socket (Pinyi, Shanghai, China). Microwaving: a microwave oven was preheated for 5 min (remove the residual moisture inside and quickly raise the internal temperature to ensure that the shellfish is maintained at a relatively constant temperature), and then mussels were placed in a porcelain dish covered with preservative film and heated for 5, 8, or 10 min at power levels of 140, 280, or 420 W.
After different heat treatments, the shellfish were removed with tweezers after cooling to indoor temperature. The meat and cooking liquor were collected (and the remaining liquid in the shell was considered to be cooking liquor). The weight and the water content of the samples were determined. These samples were homogenized and then used for toxin analysis. Three replicate samples were examined in each group to test the toxin concentration.
2.4 Sample extractionPSTs were analyzed according to published methods (Boundy et al., 2015; Wu et al., 2017) with minor adjustments. This procedure involved extraction of 5±0.05 g of homogenate meat or cooking liquor after centrifugation; the samples were weighed and extracted with 5 mL of 1% acetic acid aqueous solution. After being vortexed for 10 min, the supernatant was boiled at 100 ℃ for 5 min. The samples were removed and cooled in cold water until they reached room temperature, and then centrifuged for 5 min at 8 000×g.
The supernatant (1 mL) was removed, and combined with 5 μL of ammonium hydroxide. The supernatant was then centrifuged for 10 min at 10 000×g for solid-phase extraction purification. An ENVI-Carb solid phase extraction column (3 cc, 200 mg, Sigma-Aldrich, St. Louis, MO, USA) was activated with 3 mL of acetonitrile, 3 mL of 20% acetonitrile aqueous solution (containing 1% acetic acid), and 3 mL of 0.1% ammonium hydroxide. After activation, 500 μL of supernatant was added, and 700 μL of ultrapure water was used for washing. Finally, 1 mL of 75% acetonitrile solution (containing 0.25% formic acid) was used for elution. All extracts were filtered through a 0.22-μm membrane filter prior to liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis. The toxin concentration is expressed as mg STX.2HCl eq/kg (wet weight) and Food and Agriculture Organization of the United Nations (FAO) recommended toxic equivalency factor (TEFs) were used.
2.5 LC-MS/MS conditionsAnalytes were separated on a TSK-Amide-80 3-μm 2.0-mm×150-mm column (TosohBioscience, Tokyo, Japan) at a column temperature of 40 ℃. The LC mobile phases were water (A) and 95% acetonitrile in water (B) (both containing 2-mmol/L ammonium formate and 50-mmol/L formic acid). The injection volume was 5 μL, and the sample temperature was 4 ℃.
The LC-MS/MS system consisted of a high performance liquid chromatography (HPLC) system (LC-20ADXR, Shimadzu, Tokyo, Japan) and a hybrid triple quadrupole linear ion trap mass spectrometer (5500 QTRAP LC-MS/MS system, AB Sciex Instruments, Foster City, CA, USA) with a Turbo V source and electrospray ionization probe. MRM mode was selected. Relevant parameters are shown in Table 1. The turbo ion spray source settings were as follows: ion spray voltage (positive polarity), 5 500 V; ion spray voltage (negative polarity), -4 500 V; curtain gas, 20 psi; ion source gas 1 (GS1) and ion source gas 2 (GS2), 50 psi; ion source temperature, 550 ℃; and collision gas (CAD), medium. Nitrogen served as the nebulizer gas and collision gas in both modes. The LC-MS/MS parameters were as described previously (Boundy et al., 2015; Wu et al., 2017).
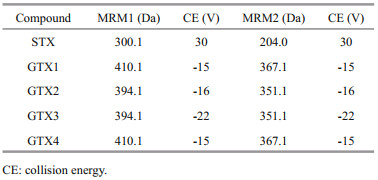
|
We determined the relative retention time drift (≤1%), calibration curve (R2≥0.98 derived from five calibration points), response drift (less than 25% slope variation between the two sets of calibration curve injections), blank QC sample (injected before and after the calibration sets, after every 10 samples, for monitoring the instrumental background), and signal-to-noise ratio of the qualifier transition intensity ≥3. The limit of detection (LOD) for analytes was determined according to a signal-to-noise ratio of 3꞉1, and the limit of quantification (LOQ) was determined according to a signal-to-noise ratio of 10꞉1. The absolute recovery range was 64.5%–93.3%. The quality control are shown in Supplementary Table S1.
2.7 Statistical analysisAll data on toxin content and composition in shellfish samples were analyzed using SPSS 19.0 (IBM Corporation, Armonk, NY, USA). The data are expressed as the mean and standard deviation, three replicates per treatment were examined. The differences in viable resting state was tested by one-way analysis of variance. Statistical significance was defined as P < 0.05. Parameters were as follows:
Toxin concentration is used to characterize the total concentration of PSTs in mg STX.2HCl eq/kg.
Toxin amount means concentration x weight, the unit is mg STX.2HCl eq.
Toxin percentage removal is used to characterize the effect to which PSTs are removed from tissues:
Toxin percentage removal (%)=(a–b)/a×100% (Zou, 2017),
where a: toxin concentration in fresh shellfish (μg/kg); b: toxin concentration in shellfish after processing (μg/kg).
3 RESULT 3.1 PST distribution in tissue vs. cooking liquorIn this study, the toxin concentration of raw tissues were approximately 29.6 mg STX.2HCl eq/kg, which significantly exceeded the standard limit of 0.8 mg STX.2HCl eq/kg. Boiling, steaming, and microwaving significantly decreased the content of PSTs in tissues (Fig. 3a). The effects of microwave treatment were the best: the lowest content was 0.20 mg STX.2HCl eq/kg after microwave heating at a power level of 420 W. During the processing, each method produced different volumes of cooking liquor, which carried some toxins away from tissues. However, the toxin concentration in tissue and cooking liquor still exceeded the regulatory limit. Thus, although cooking may decrease the levels of toxins in tissue, it does not eliminate the danger of toxicity.

|
| Fig.3 Total toxin concentration and sample weight a. toxin concentration in tissue and liquor with five kinds of heating processing (*: P<0.05; **: P<0.01); b. weight of tissue and liquor after five kinds of heating processing. |
The distribution of toxins was closely related to the amount of cooking liquor produced (F=46.9; P < 0.01). The more cooking liquor was produced, the more significant the decrease of toxin concentration in tissues was. The most cooking liquor was produced after steaming and boiling, and the toxin concentration in tissues decreased by more than 85% compared with the control group (Fig. 3b). Thus, these findings indicated that the cooking liquor poses a serious potential risk of PSP poisoning. PSTs concentration and distribution should ideally be controlled by controlling the moisture content during processing.
3.2 Temperature (power) and time effects on the removal and destruction of PSTsIn the process of cooking, the liquor was formed and a considerable amount of toxins was taken away from tissues, which was the main form to reduce the consumptive risk of mussel tissue. We used the removal percentage combined with the change of moisture content to characterize the ability of five methods to reduce toxins in tissues (Fig. 4). The five processing methods tested were effective at removing PSTs from M. edulis tissues (P < 0.01). Among the methods, boiling, steaming, and microwaving had the highest removal percentage, with 89.1%±2.40%, 86.5%±4.02%, and 84.6%±4.87%, respectively. The unconventional methods of water-bath heating at 60 ℃ and 80 ℃ mean to assess the potential hazards of medium cooked shellfish, which led to more retention of the toxin in tissue and inevitably increased the risk of consumption.
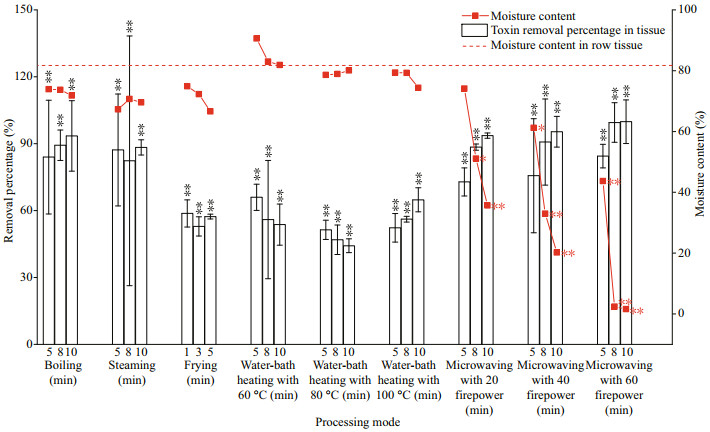
|
| Fig.4 Effects of heating temperature (power) and time on the percentage removal of toxins Samples with a significantly different toxin removal percentage in comparison to the control group are designated by * (P<0.05) and ** (P<0.01). |
Overall, the moisture content in tissues gradually decreased, while the removal percentage increased with the extension of treatment time. This was particularly true for mussels cooked by boiling or microwaving. The removal percentage can be increased by more than 2% and 3% respectively when the heating time was prolonged for 1 min. However, this rule is not absolute. For individual methods, such as water-bath heating, the moisture content of tissues may increase, which affected the removal of toxins with the extension of time. For microwaving, the moisture content decreased most sharply, so its removal percentage also increased significantly. However, during microwaving treatment, the moisture content of tissue decreased sharply from more than 74.1% to 1.60%, which has a very significant impact on the edibility of the M. edulis.
3.3 Toxin transformation in M. edulis during cookingThe total amounts of toxin were analyzed, and the results showed that five methods had effects on the destruction of toxins. For individual analogues (Fig. 5a), their abilities of destruction was different. All methods significantly decreased the amounts of STX and GTX3. Microwaving was particularly effective. However, the destruction of GTX1, GTX2, and GTX4 was not ideal through boiling and steaming. Especially, in steaming and water-bath heating, the amount of GTX4 even exceeded that of the control group. Thus, toxins may be converted to other forms during cooking. In this study, the transformation to GTX4 was mainly manifested.
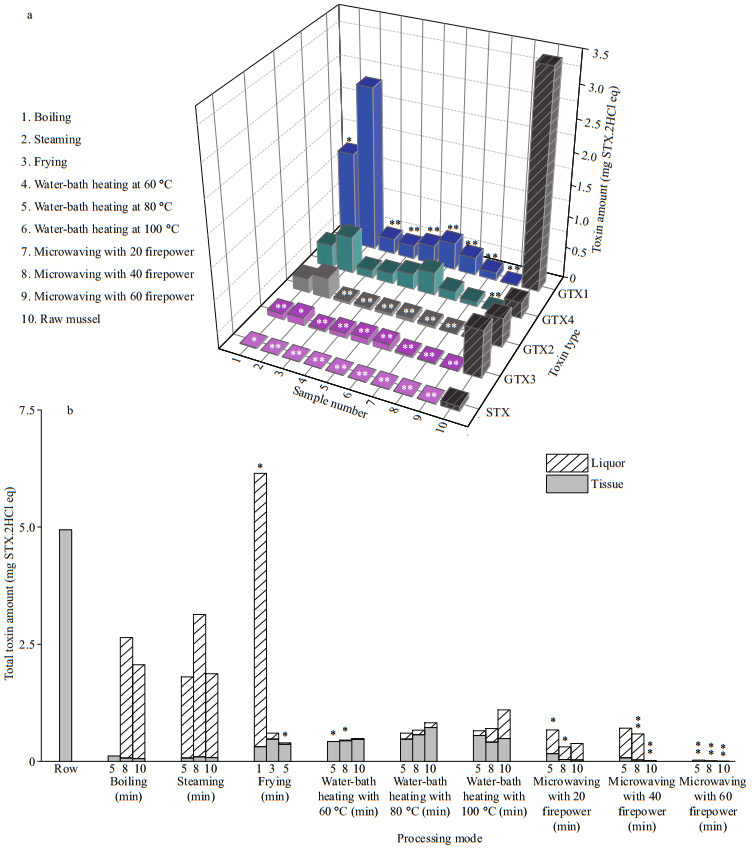
|
| Fig.5 Amount analysis of total and individual toxin a. the total amount of five toxins (GTX1-4, STX) in five cooking methods; b. total amount of toxin and its distribution in liquor and tissue. Compared with control group, *: P < 0.05; **: P < 0.01. |
Comparison of total amounts with control group is shown in Fig. 5b. Overall, the amount of toxins decreased through five methods, and most of the toxins were distributed in the liquor. Thus, choosing a reasonable cooking method and discarding liquor can significantly reduce the risk of poisoning. In terms of safety, microwaving is recommended, which can reduce the amount by 99%, and the toxin in tissues was almost completely removed, but it is inevitable that the food quality deteriorated with the decrease of moisture content. Especially, the amount of total toxins was higher than that of the control group after 1 min of frying, which further confirmed the existence of toxin transformation during cooking.
4 DISCUSSION 4.1 Water loss was the most important reason for decreased toxin concentration in cookingIf the initial toxicity level is low, cooking may effectively decrease the toxicity below standard limits (Food and Agricultural Organization, 2004) for the tissue. This study showed that cooking styles must be distinguished in this case. When toxin level exceeded the regulatory limit by more than 30 times, boiling, steaming, and microwaving were able to decrease the toxin concentration in mussel tissues significantly. However, cooked mussels remained above the regulatory level. Frying and water bath treatments resulted in a smaller decrease in toxin concentration. This difference may be closely related to the generation and transfer of cooking liquor. The related literature has reported that more than 50% (32%–64%) of the total PST is secreted via exudation of water from tissues during the processing of seafood (Wong et al., 2009; Zhang, 2015). Even repeated freezing and thawing can cause PSTs to penetrate into the exudate (Reboreda et al., 2010). In our study, the M. edulis lost approximately 10%–90% of the meat weight during processing and produced cooking liquor. The transfer rate of toxins ranged from 20% to nearly 90%, and significant differences were observed among treatment groups. In the current study, the total sample weight of tissue was highest after water-bath heating, which indicates relatively little water loss and therefore higher levels of toxins preserved. In contrast, microwaving resulted in the highest water loss and the lowest toxin concentration in the tissue. The amount of toxins in processed shellfish was essentially the same after conventional cooking via boiling and steaming (about 2.17 mg STX.2HCl eq/kg in the cooking liquor). Therefore, water loss is the main reason underlying the decreased toxin concentration in shellfish.
4.2 Appropriate cooking methods and conditions to the destruction of PSTsPrevious studies suggested that elimination of toxins by heating is mainly due to the loss of water but that heating does not cause damage to the toxin components themselves through chemical decomposition (Etheridge, 2010). Some data from our study support this view. Steaming and boiling resulted in a high percentage removal (more than 87%), but in fact, more than 50% of the toxins removed were found in the cooking liquor and were not destroyed. However, results of the current study demonstrate that toxins were damaged by microwaving. The removal percentages were more than 80% under different power levels, and the highest can reach 99.51%. At power 420 W, all the removed toxins were destroyed in tissue. Therefore, microwaving has advantages over conventional heating techniques because the temperature distribution may be more uniform than that of conventional heating (Ştefănoiu et al., 2016). We agree to it, and aim to know about the profound influence of uniform heating. Wang et al. (2019) have reported that microwave irradiation sharply decreases the bound endotoxin concentration and accelerates toxin release from bacterial cells. In addition, compared with other heating technologies, microwaving results in more water loss in our study. Therefore, we further speculated that microwaving might break the cell membrane structure and release cytoplasm containing toxins with the help of uniform heating, thereby leading to more water loss and toxin removal.
4.3 Toxin profile of PSTs in M. edulis—toxin transformation by cookingAt present, more than 50 PSTs have been studied qualitatively (Wiese et al., 2010). During their accumulation and metabolism along the food chain from algae to shellfish, the complex toxin transformation process produces molecular forms with greater or lesser toxicity. For example, 11β-epimer toxin 2 and GTX-4 are predominant toxins produced by the dinoflagellate Alexandrium tamarense, whereas 11α-epimer toxin 1 is predominant in blue mussels (Kim and Shin, 2015). In this study, toxin transformation exists, and the main evidence was that the total toxin amount exceeded the raw group in frying and the amount of GTX4 increased abnormally after cooking by several methods. In addition, elevated GTX4 was the most important transformation feature. GTX4, as the main toxin of A. tamarense, had a high amount, and further increases through boiling, frying, and water-bath heating. GTX4 and GTX1 are isomers, and there is conversion. Qiu (2014) proposed it could promote the conversion from GTX1 to GTX4 with enough glutathione reductase (GR) in mussels. In this study, it is reasonable to assume that the stress of mussels increased the enzyme activity of GR to promote transformation. In addition, Zou (2000) proposed that GTX2 and GTX3 can also be transformed into GTX4 under the action of N-oxidase, but it was first found in A. tamarense, not mussels. After mussels eat toxic algae, they may follow into the body to catalyze the transformation of toxicity. They may follow the toxin algae into the body to catalyze the transformation of toxicity with the feeding process.
Overall, each of the toxin analogies decreased significantly, so the destruction was the main reason, but there may be other factors that lead to this result together. Highly toxic analogues (e.g., GTXs and STX) that do not decompose easily have been reported to have a tendency to transform into the unstable toxin M-toxin in the detoxification process (Che et al., 2020). Therefore, we suspected that the transformation process for new C-11 hydroxyl metabilites (so-called M-toxins) also occurred in the cooking processes in this study, thus further indicating transformation and decomposition. However, studies on the identification and biotransformation of these molecules are limited, and these processes require further investigation.
In addition, to decrease the toxin content, creating a high temperature and high pH cooking environment is essential. GTX 1/4 toxins decrease at high temperatures and at high pH. GTX 2/3 toxins increase initially at low pH and then decline with subsequent heating, whereas STX increases consistently at pH 3 to 4 (Indrasena and Gill, 2000). Adding edible soda to scallops for curing before cooking has been shown to decrease PSP toxins to varying degrees in the alkaline environment (Zhang, 2015). The results of this study may be improved by combining edible soda with cooking.
5 CONCLUSIONThe effects of five cooking methods (steaming, boiling, frying, water bath heating, and microwaving) on the removal and destruction of PSTs in M. edulis samples were determined. The results showed that conventional heating methods (steaming, boiling, and water-bath heating) are not effective at destroying toxins. With these methods, toxins drain from mussel tissues, mainly into the cooking liquor, and can still pose a safety risk. However, higher temperature treatments such as microwaving can destroy toxins in shellfish. The results of this study suggest that the risk of PSP can be minimized by steaming, boiling, or microwaving mussels before eating and discarding the cooking liquor. Otherwise, heating methods at higher temperatures, such as microwaving, should be used to ensure safety.
6 DATA AVAILABILITY STATEMENTThe datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Electronic supplementary material
Supplementary material (Supplementary Table S1) is available in the online version of this article at https://doi.org/10.1007/s00343-021-1172-0.
Berenguer J A, Gonzalez L, Jimenez I, Legarda T M, Olmedo J B, Burdaspal P A. 1993. The effect of commercial processing on the paralytic shellfish poison (PSP) content of naturally-contaminated Acanthocardia tuberculatum L. Food Additives & Contaminants, 10(2): 217-230.
|
Boundy M J, Selwood A I, Harwood D T, McNabb P S, Turner A D. 2015. Development of a sensitive and selective liquid chromatography-mass spectrometry method for high throughput analysis of paralytic shellfish toxins using graphitised carbon solid phase extraction. Journal of Chromatography A, 1387: 1-12.
DOI:10.1016/j.chroma.2015.01.086 |
Che Y J, Ding L, Qiu J B, Ji Y, Li A F. 2020. Conversion and stability of new metabolites of paralytic shellfish toxins under different temperature and pH conditions. Journal of Agricultural and Food Chemistry, 68(5): 1427-1435.
DOI:10.1021/acs.jafc.9b07063 |
Ding L, Qiu J B, Li A F. 2017. Proposed biotransformation pathways for new metabolites of paralytic shellfish toxins based on field and experimental mussel samples. Journal of Agricultural and Food Chemistry, 65(27): 5494-5502.
DOI:10.1021/acs.jafc.7b02101 |
Etheridge S M. 2010. Paralytic shellfish poisoning: seafood safety and human health perspectives. Toxicon, 56(2): 108-122.
DOI:10.1016/j.toxicon.2009.12.013 |
Food and Agricultural Organization. 2004. Marine Biotoxins FAO Food and Nutrition Paper 80. FAO, Rome.
|
García C, Barriga A, Díaz J C, Lagos M, Lagos N. 2010. Route of metabolization and detoxication of paralytic shellfish toxins in humans. Toxicon, 55(1): 135-144.
DOI:10.1016/j.toxicon.2009.07.018 |
Halstead B W, Schantz E J. 1984. Paralytic shellfish poisoning. Geneva: World Health Organization; Albany, NY: WHO Publications Centre USA.
|
Indrasena W M, Gill T A. 2000. Thermal degradation of partially purified paralytic shellfish poison toxins at different times, temperatures, and pH. Journal of Food Science, 65(6): 948-953.
DOI:10.1111/j.1365-2621.2000.tb09398.x |
Indrasena W M. 1999. Detection and thermal degradation of paralytic shellfish poisoning toxins. DalTech-Dalhousie University, Canada.
|
Kim H Y, Shin I S. 2015. Comparison of paralytic shellfish toxin profiles of Alexandrium tamarense and blue mussel(Mytilus edulis) in Korea. Food Science and Biotechnology, 24(2): 751-756.
DOI:10.1007/s10068-015-0097-9 |
Lawrence J F, Maher M, Watson-Wright W. 1994. Effect of cooking on the concentration of toxins associated with paralytic shellfish poison in lobster hepatopancreas. Toxicon, 32(1): 57-64.
DOI:10.1016/0041-0101(94)90021-3 |
Ministryof Agriculture and Rural Affairs of the People's Republic of China. 2017. China Fishery Statistical Yearbook. China Agriculture Press, Beijing.
(in Chinese)
|
Ochi N. 2021. Simultaneous determination of ten paralytic shellfish toxins and tetrodotoxin in scallop and short-necked clam by ion-pair solid-phase extraction and hydrophilic interaction chromatography with tandem mass spectrometry. Journal of Chromatography A, 1651: 462328.
DOI:10.1016/j.chroma.2021.462328 |
Qiu J B. 2014. Metabolic Transformation of Paralytic Shellfish Toxins by Bivalve Molluscs and Their Physiological and Biochemical Responses. Ocean University of China, Qingdao. (in Chinese)
|
Reboreda A, Lago J, Chapela M J, Vieites J M, Botana L M, Alfonso A, Cabado A G. 2010. Decrease of marine toxin content in bivalves by industrial processes. Toxicon, 55(2-3): 235-243.
DOI:10.1016/j.toxicon.2009.07.029 |
Ştefănoiu G A, Tănase E E, Miteluţ A C, Popa M E. 2016. Unconventional treatments of food: microwave vs. radiofrequency. Agriculture and Agricultural Science Procedia, 10: 503-510.
DOI:10.1016/j.aaspro.2016.09.024 |
Turnbull A, Malhi N, Tan J, Harwood D T, Madigan T. 2018. Fate of paralytic shellfish toxins in southern rock lobster (Jasus edwardsii) during cooking: concentration, composition, and distribution. Journal of Food Protection, 81(2): 240-245.
DOI:10.4315/0362-028X.JFP-17-280 |
Vieites J M, Botana L M, Vieytes M R, Leira F J. 1999. Canning process that diminishes paralytic shellfish poison in naturally contaminated mussels (Mytilus galloprovincialis). Journal of Food Protection, 62(5): 515-519.
DOI:10.4315/0362-028X-62.5.515 |
Wang C, Zhang Z W, Liu H. 2019. Microwave-induced release and degradation of airborne endotoxins from Escherichia coli bioaerosol. Journal of Hazardous Materials, 366: 27-33.
DOI:10.1016/j.jhazmat.2018.11.088 |
Wiese M, D'Agostino P M, Mihali T K, Moffitt M C, Neilan B A. 2010. Neurotoxic alkaloids: saxitoxin and its analogs. Marine Drugs, 8(7): 2185-2211.
DOI:10.3390/md8072185 |
Wong C K, Hung P, Lee K L H, Mok T, Kam K M. 2009. Effect of steam cooking on distribution of paralytic shellfish toxins in different tissue compartments of scallops Patinopecten yessoensis. Food Chemistry, 114(1): 72-80.
DOI:10.1016/j.foodchem.2008.09.018 |
Wu H Y, Guo M M, Bing X F, Zheng G C, Peng J X, Tan Z J, Zhai Y X. 2017. Simultaneous identification and detection of paralytic shellfish toxin in bivalve mollusks by liquid chromatography coupled with quadrupole/linear ion trap tandem mass spectrometry. Oceanologia et Limnologia Sinica, 48(3): 508-515.
(in Chinese with English abstract) |
Yu R C, Luo X. 2016. Status and research perspectives on toxic algae and phycotoxins in the coastal waters of China. Studia Marina Sinica, (51): 155-166.
|
Zhang S S. 2015. Research on Control and Degradation of Paralytic Shellfish Poisoning Toxins. Chinese Academy of Agricultural Sciences, Beijing. (in Chinese)
|
Zou Y L. 2000. The production of paralytic shellfish poisoning (PSP) toxins during the growth of toxic algae Alexandrium, and the uptake, accumulation, and detoxinfication of PSP in Chinese scallop Chlamys farreri. The First Institute of Oceanography, Qingdao.
|
Zou Z Y, Sun J L, Feng Z, Shi R T, Hou S, Li H J. 2017. Removal of deoxynivalenol and T-2 toxin by thermal treatment. Food Research and Development, 38(13): 6-10.
(in Chinese with English abstract) DOI:10.3969/j.issn.1005-6521.2017.13.002 |
 2022, Vol. 40
2022, Vol. 40


