Institute of Oceanology, Chinese Academy of Sciences
Article Information
- GENG Huixia, SUN Hongxiao, LIU Chao, KONG Fanzhou, ZHANG Qingchun, YAN Tian, YU Rencheng
- Screening for lipophilic marine toxins and their potential producers in coastal waters of Weihai in autumn, 2020
- Journal of Oceanology and Limnology, 40(6): 2218-2230
- http://dx.doi.org/10.1007/s00343-022-2301-0
Article History
- Received Aug. 17, 2022
- accepted in principle Sep. 15, 2022
- accepted for publication Sep. 30, 2022
2 Laboratory for Marine Ecology and Environmental Science, Pilot National Laboratory for Marine Science and Technology(Qingdao), Qingdao 266237, China;
3 University of Chinese Academy of Sciences, Beijing 100049, China;
4 Center for Ocean Mega-Science, Chinese Academy of Sciences, Qingdao 266071, China
Some microalgae in the sea can produce potent phycotoxins as secondary metabolites, which give the producers competitive advantages against other microalgae and protect them from predators (Cembella, 2003; Ianora et al., 2011). Such phycotoxins could be transferred via marine food chains, and result in mortality of marine animals, damaging fisheries and aquaculture, and threatening public health (Berdalet et al., 2016; Li et al., 2021). The negative impacts of marine phycotoxins are often associated with harmful algal blooms (HABs) formed by toxic microalgae (Shumway, 1990; Landsberg, 2002). The apparent increase of HABs around the world due to climate change, eutrophication, anthropogenic activities, or increasing awareness of HABs (Gobler, 2020; Hallegraeff et al., 2021), prompted the establishment of monitoring and research on HABs and phycotoxins (Howard et al., 2021; Otero and Silva, 2022).
Marine phycotoxins could be classified into eight major groups based on the chemical structure: okadaic acid (OA) and its derivatives, azaspiracids (AZAs), yessotoxins (YTXs), brevetoxins (BTXs), cyclic imines (CIs), pectenotoxins (PTXs), saxitoxins (STXs), and domoic acid (DA) (Farabegoli et al., 2018). Most of the marine phycotoxins, except for STXs and DA, are lipophilic compounds, which could be collectively named as lipophilic marine toxins and detected simultaneously with liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS). Thousands of human poisoning events related to lipophilic marine toxins occurred around the world every year, particularly in Europe, Asia, and America (De Schrijver et al., 2002; Garcêa et al., 2005; Lin et al., 2015; Nicolas et al., 2017).
In China, poisoning episodes associated lipophilic marine toxins were reported occasionally (Li et al., 2012; Liang et al., 2022). Investigations on lipophilic marine toxins started from the late 1990s (Zhou et al., 1999). The early investigations on lipophilic marine toxins mainly focused on those accumulated in wild or cultured edible shellfish, using the method of mouse bioassay (MBA). This method, however, was gradually given up due to a high proportion of false-positive result (Chapela et al., 2008; Bodero et al., 2018), and other ethical or technical concerns. The methods based on high performance liquid chromatography coupled with mass spectrometry (LC-MS) gradually take the place of MBA for detection of lipophilic marine toxins. Recently, the regulatory limit (GB 2733-2015) and official method based on LC-MS/MS (GB 5009.212-2016) have been established for OA and its derivatives in shellfish in China, but there are still no regulatory limits and methods for other lipophilic marine toxins. Using the state-of-the-art methods of LC-MS/MS, many investigations have been carried out to investigate the composition, distribution, and seasonal dynamics of lipophilic marine toxins during the last decade in the Bohai Sea (Liu et al., 2017, 2019a; He et al., 2020), Yellow Sea (Li et al., 2010; Wang et al., 2015; Chen et al., 2018; Wu et al., 2019, 2020; Liu et al. 2021a), East China Sea (He et al., 2019; Wu et al., 2020), and South China Sea (Jiang et al., 2017; Liu et al., 2019b; 2020, 2021b; Ji et al., 2022). However, lipophilic marine toxins are still not well studied, particularly in some important aquaculture zones near the coast.
Weihai is a coastal city located at the east end of Shandong peninsula, and has important aquaculture industries of seaweed and oysters, scallops, and clams. Shellfish production occupied over 60% of the total aquaculture production in Weihai (Hou et al, 2018). Despite the significance of aquaculture industry, there was little investigation of marine toxins in this region. However, previous investigations in the Bohai Sea and Yellow Sea indicated that this region was likely affected by lipophilic marine toxins, such as PTXs (Chen et al., 2018). Therefore, phytoplankton and shellfish samples were collected in this study during the cruise along the coast of Weihai in 2020 to detect lipophilic marine toxins using LC-MS/MS, and the potential producers of toxins were resolved based on microscopy observation and high throughput sequencing analyses of phytoplankton samples.
2 MATERIAL AND METHOD 2.1 Chemical reagents and standardsHPLC-grade solvents acetonitrile and methanol were purchased from Merck, Germany, and ammonium hydroxide used in this study was from Sigma-Aldrich, Germany. Ultrapure water of 18.2 MΩ·cm prepared with the Milli-Q water purification system (Milli-Q Reference, Millipore Ltd., USA) was used for toxin analysis and molecular experiments. Certified reference materials of lipophilic marine toxins, including OA, dinophysistoxins-1 (DTX1), yessotoxin (YTX), homoyessotoxin (homo-YTX), azaspiracid 1–3 (AZA1–3), 13-desmethyl spirolide C (13-DesMe-C), gymnodimine (GYM), and pectenotoxin-2 (PTX2) were obtained from the Institute for Marine Biosciences, National Research Council of Canada (Halifax, Canada). All the standards were stored at -20 ℃ in the dark.
2.2 Collection of phytoplankton and shellfish samplesPhytoplankton samples for lipophilic marine toxin analysis and high throughput sequencing analysis were collected, following the protocols described by Liu et al. (2017) and Chen et al. (2019). Phytoplankton samples for lipophilic marine toxin analysis were collected from 53 sampling sites in 27 bays along the coast of Weihai (121.6°E–122.7°E, 36.7°N–37.6°N), Shandong province, from October to November in 2020 (Fig. 1). A total of 1 000-L seawater was pumped from the surface and concentrated with a phytoplankton net (pore size 20 m). The net-concentrated phytoplankton sample was filtered by a sieve (pore size 200 m) to remove zooplankton and other organisms, then made up to a volume of 850 mL. A fraction of 50-mL concentrated phytoplankton sample was fixed with Lugol's iodine solution (final concentration ca. 2%) and preserved at room temperature for morphological observation. Another 200 mL of concentrated phytoplankton sample was filtered onto a fiberglass membrane (GF/C, Whatman, nominal pore size 1.2 m), which was preserved in a freezer at -20 ℃ until analysis of lipophilic marine toxins with LC-MS/MS. For high throughput sequencing analysis, 2-L seawater was collected from the surface and passed through a sieve with pore size of 200 μm to remove zooplankton and other organisms, then filtered on a polycarbonate membrane (Millipore, USA, nominal pore size 0.4 m) under 30-kPa vacuum to collect phytoplankton. The membranes were stored at -80 ℃ until analysis.
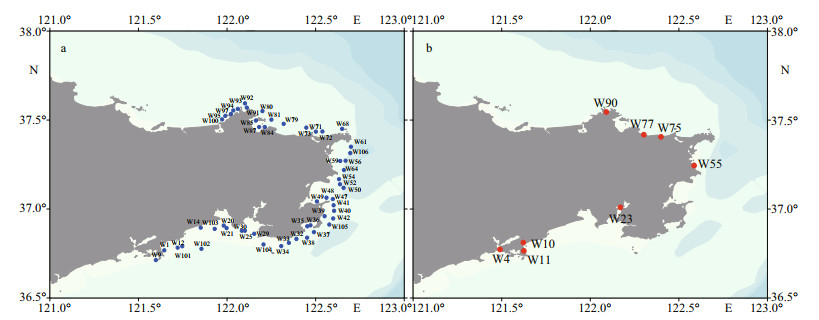
|
| Fig.1 Illustration of the sampling sites for phytoplankton (a) and shellfish (b) in coastal waters of Weihai in autumn, 2020 |
A total of 11 shellfish samples, including 4 species of clams (Ruditapes philippinarum, Anadara kagoshimensis (=Scapharca subcrenata), Meretrix lusoria, and Cetoconcha hyaline), 1 species of scallop (Argopecten irradians), 1 species of mussel (Mytilus galloprovincialis), and 1 species of oyster (Magallana gigas (=Crassostrea gigas)), were collected from 8 sites (Table 1) along the coast of Weihai (Fig. 1). For each species of shellfish, about 2 kg flesh tissues were collected and preserved in a freezer at -20 ℃ until analysis of lipophilic marine toxins with LC-MS/MS.
The extraction of lipophilic marine toxins from net-concentrated phytoplankton samples and shellfish samples referred to the protocols described in Liu et al. (2017, 2019a). Briefly, the membrane containing phytoplankton, or homogenized flesh tissues of shellfish (1 g), were extracted 3 times, each with 3-mL methanol. The supernatants were then combined and made up to 10 mL with methanol in a volumetric flask. A fraction of raw extract from phytoplankton (5 mL) or shellfish (1 mL) in methanol was diluted with ultrapure water to around 30% and loaded into a solid phase extraction (SPE) cartridge (strata-X, 3 cm3, 60 mg, Phenomenex) previously conditioned by alternative treatment with 3-mL methanol and 3-mL deionized water. The cartridge was flushed with 3 mL of methanol/water (20:80, v/v), and dried under mild vacuum. Toxins in the cartridge were then eluted with 1-mL methanol containing 0.3% (v/v) ammonium hydroxide. The eluent was filtered through a 0.22-μm syringe membrane filter, and preserved in a freezer at -80 ℃ prior to the analysis with HPLC-MS/MS.
Lipophilic marine toxins in phytoplankton and shellfish were analyzed using a LC-MS/MS method described in Liu et al. (2017, 2019a). Briefly, a Thermo Fisher UltiMate 3000 HPLC system was used for chromatographic separation of lipophilic marine toxins with a Waters X-Bridge C18 column (150 mm 3 mm, 3.5 mm) and a guard column (10 mm 2.1 mm, 3.5 mm). Mobile phases A (acetonitrile/water, 10:90, v/v) and B (acetonitrile/water, 90:10, v/v), both containing 6.7-mmol/L ammonium hydroxide (pH 11), were used to elute lipophilic marine toxins using a gradient program as described in Liu et al. (2017). A mass spectrometer AB-SCIEX-4500 Q-Trap (Applied Biosystems, Darmstadt, Germany) equipped with a TurboSpray® interface was used to detect lipophilic marine toxins. The mass spectrometer was operated in both negative and positive electrospray ionization (ESI) and in multiple reaction monitoring (MRM) mode for three different retention time windows (Liu et al., 2017).
Instrument response to lipophilic marine toxins were determined with detection limit of 1.42 ng/mL and quantification limit of 4.27 ng/mL (Liu, 2017). The matrix effects, recovery and precision of the method have been examined by Liu et al. (2019a). The results showed that the clean-up procedure using Strata-X cartridges could significantly reduce matrix effects. The method had good performances, with recoveries ranged from 83% to 126% for phytoplankton samples and 83% to 132% for shellfish samples. Relative standard deviation (RSD) was less than 10% for phytoplankton samples and less than 14% for shellfish samples (Liu, 2017). The shellfish samples were treated in duplicate and single phytoplankton samples collected from each site were analyzed in this study.
2.4 High throughput sequencing analysis of phytoplankton samplesIn this study, high-throughput sequencing data for phytoplankton samples were used to screen for potential producers of lipophilic marine toxins in the coastal waters of Weihai. Protocol for sample preparation and analysis refers to Chen et al. (2019). Genomic DNA of phytoplankton samples was extracted following the protocol of Winnepenninckx et al. (1993). A primer pair for eukaryotes (forward primer D514, 5′-TCCAGCTCCAATAGCGTA-3′ and reverse primer B706R, 5′-AATCCRAGAATTTCACCTCT-3′) was used to amplify the V4 region of the 18S rRNA gene (18S rDNA) (Cheung et al., 2010; Zimmermann et al., 2011). The NEB Next® Ultra™ DNA Library Prep Kit for Illumina (Illumina Inc., USA) was used to construct the sequence libraries, which were sequenced on the Illumina HiSeq2500 platform. Reads were truncated and assembled using FLASH (Version 1.2.7), and QIIME (Version 1.7.0) was used to generate high-quality clean tags. After removing singletons from the dataset (Flynn et al., 2015), and sequences were clustered into operational taxonomic units (OTUs, similarity above 97%) using UPARSE software (Version 7.0.1001) (Edgar, 2013; Flynn et al., 2015). Taxonomic assignment of the OTUs related to Alexandrium spp., Dinophysis spp. and Azadinium spp. were completed using both SILVA Database (Release 119, https://www.arb-silva.de/documentation/release-119/) and Nucleotide Database in NCBI (https://blast.ncbi.nlm.nih.gov/).
A total of 35 representative sequences of the 18S rDNA V4 region were selected for the species in genus Alexandrium and downloaded from GenBank. Together with the sequences of 6 OTUs assigned to Alexandrium spp., a maximum-likelihood tree was established using Mega 10 (https://megasoftware.net/) variance estimation method with bootstrap replications of 10 000 and the best-fitting nucleotide substitution model of Tamura 3-parameter+G (Kumar et al., 2018). The sequence of Prorocentrum donghaiense was used as the outside group. The OTUs of Dinophysis spp. and Azadinium spp. identified in the study area were analyzed using the same protocol, with 22 representative sequences of the genus Dinophysis, and 25 representative sequences of the genus Azadinium.
2.5 Microscopy observation of phytoplankton samplesPhytoplankton samples fixed with Lugol's iodine solution were settled for 24 h to 200 mL following the protocol of the Utermöhl method (Sournia, 1978), and a fraction of 0.2 mL was observed under an inverted light microscope (Primo Vert, Zeiss, Germany). Alexandrium spp., Dinophysis spp. and Azadinium spp. were identified and counted under the microscope.
2.6 Data processingDate acquisition of phycotoxins with LC-MS/MS was performed using software Multiquant 3.0. All the data were processed with Microsoft Excel (Microsoft, USA).
3 RESULT 3.1 Lipophilic marine toxins in phytoplankton and shellfish in coastal waters of WeihaiThree lipophilic marine toxins, namely AZA1/AZA40, 13-DesMe-C, and PTX2, were detected in net-concentrated phytoplankton samples collected from coastal waters of Weihai. Among the three toxins, PTX2 was the ubiquitous toxin detected in 47 phytoplankton samples (Figs. 2 & 3). The maximum content of PTX2 was 354 pg/L in a sample collected from the Weihai Bay north to the city. PTX2 content also had a high value of 192 pg/L in a region close to the Rushan Bay south to Weihai. Besides, on the east coast of Weihai, the PTX2 content also had relatively higher values. Trace amount of AZA1 or AZA40 was detected in 28 phytoplankton samples, which were widely distributed along the coast of Weihai (Figs. 2 & 4). However, the amount of toxin was too low to determine the content quantitatively. Moreover, it is hard to discriminate between AZA1 and AZA40 since the two analogues had the same molecular weight and similar retention time. 13-DesMe-C was detected in 16 phytoplankton samples, which were mainly distributed in the northern and eastern coastal waters of Weihai. The content of 13-DesMe-C was also very low and it is impossible to make quantitative determination.
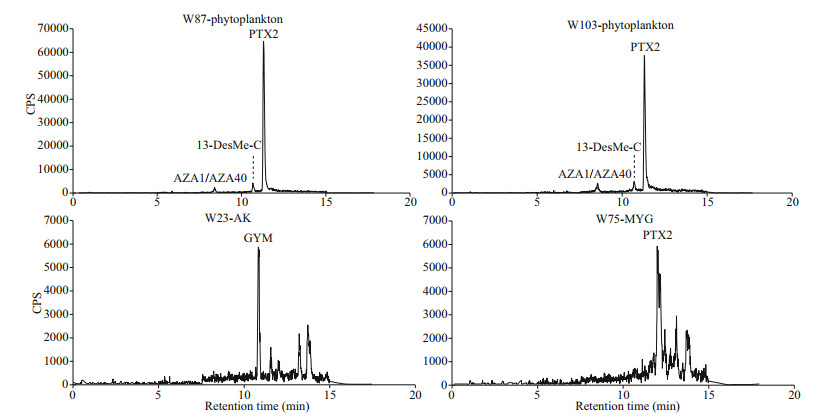
|
| Fig.2 The HPLC-MS/MS chromatograms for lipophilic marine toxins in phytoplankton and shellfish samples collected from coastal waters of Weihai AK: Anadara kagoshimensis; MYG: Mytilus galloprovincialis; AZA1/AZA40: azaspirolide-1/40; 13-DesMe-C: 13-desmethyl spirolide C; PTX2: pectenotoxin-2; GYM: gymnodimine. |
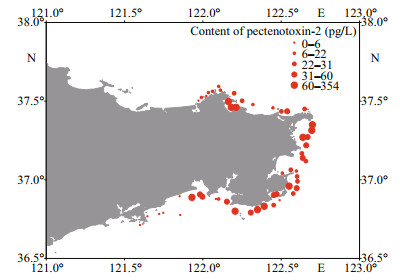
|
| Fig.3 Distribution and content of pectenotoxin-2 (pg/L) in the phytoplankton samples collected from coastal waters of Weihai |
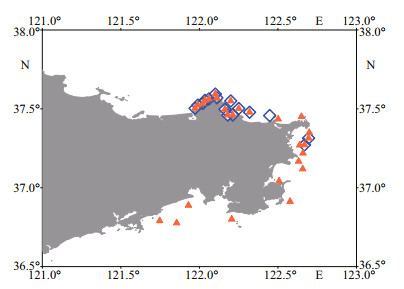
|
| Fig.4 Distribution of azaspirolide-1 (orange triangle) and 13-desmethyl spirolide C (blue diamond) in the phytoplankton samples collected from coastal waters of Weihai |
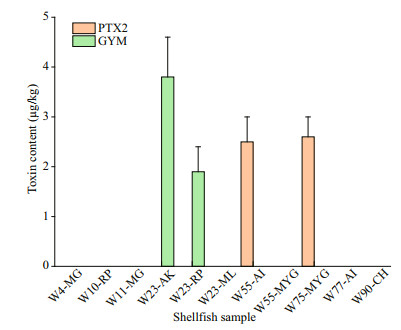
Only two lipophilic marine toxins, namely GYM and PTX2, were detected in shellfish samples collected along the coast of Weihai (Figs. 2 & 5). PTX2 were detected in a scallop sample collected form site W55 and a mussel sample from site W75. Content of PTX2 in the scallops and mussels were 2.45±0.5 and 2.63±0.4 μg/kg, respectively. GYM was detected in the two clam samples of R. philippinarum and A. kagoshimensis from the sampling site W23, and the contents were 1.87±0.5 and 3.77±0.8 μg/kg, respectively.
|
| Fig.5 Component and content of lipophilic marine toxins in the shellfish collected from coastal waters of Weihai MG: Magallana gigas; RP: Ruditapes philippinarum; AK: Anadara kagoshimensis; ML: Meretrix lusoria; AI: Argopecten irradians; MYG: Mytilus galloprovincialis; CH: Cetoconcha hyaline. |
Based on the results of high throughput sequencing data of phytoplankton samples collected from coastal waters of Weihai, a total of 5 300 101 tags were acquired (76 112/sample on average), and 1 703 OTUs were assigned to eukaryotic microalgae. According to the BLASTing results of the NCBI nucleic acid database, 6 OTUs were assigned to the species in genus Alexandrium, and 2 OTUs were assigned to the species in genus Dinophysis, and 2 OTUs were assigned to the species in genus Azadinium.
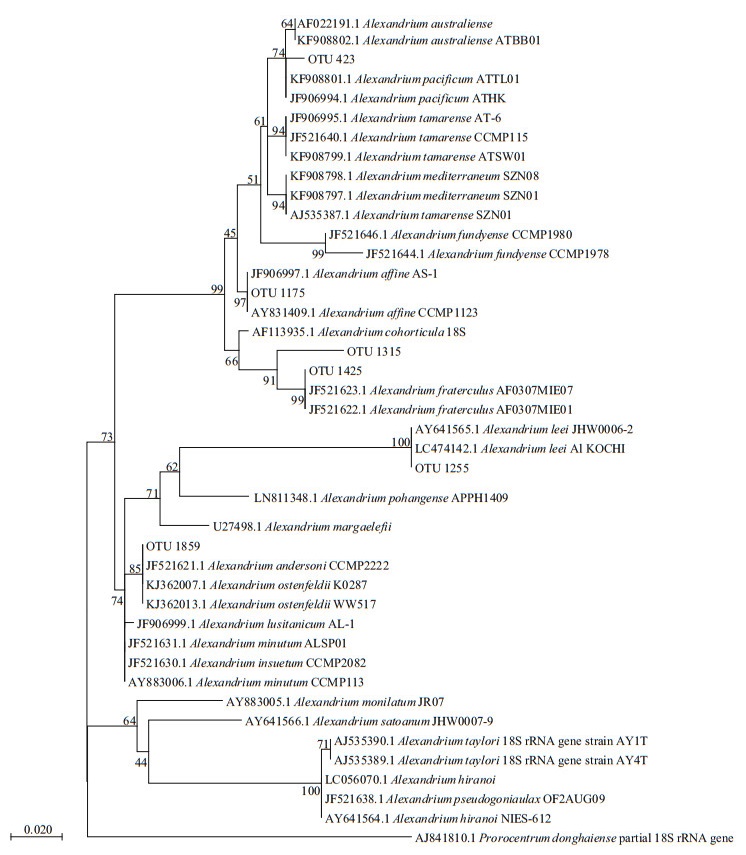
Although vegetative cells of Alexandrium spp. were observed in net-concentrated phytoplankton samples, it is difficult to identify them to the species level. Based on the data of high throughput sequencing and phylogenetic results, 6 OTUs related to Alexandrium were assigned to A. pacificum, A. affine, A. fraterculus, A. leei and A. ostenfeldii/A. andersoni (Fig. 6). It is impossible to discriminate A. ostenfeldii from A. andersoni since the two species have the same sequence in the V4 region. All the OTUs had a high identity to their corresponding species (> 99%), except for the OTU 1315 assigned to A. fraterculus (96.3%).
|
| Fig.6 Phylogenetic tree established for species in genus Alexandrium and the OTUs derived from high throughput sequencing results of phytoplankton samples in coastal waters of Weihai based on the V4 region of the LSU |
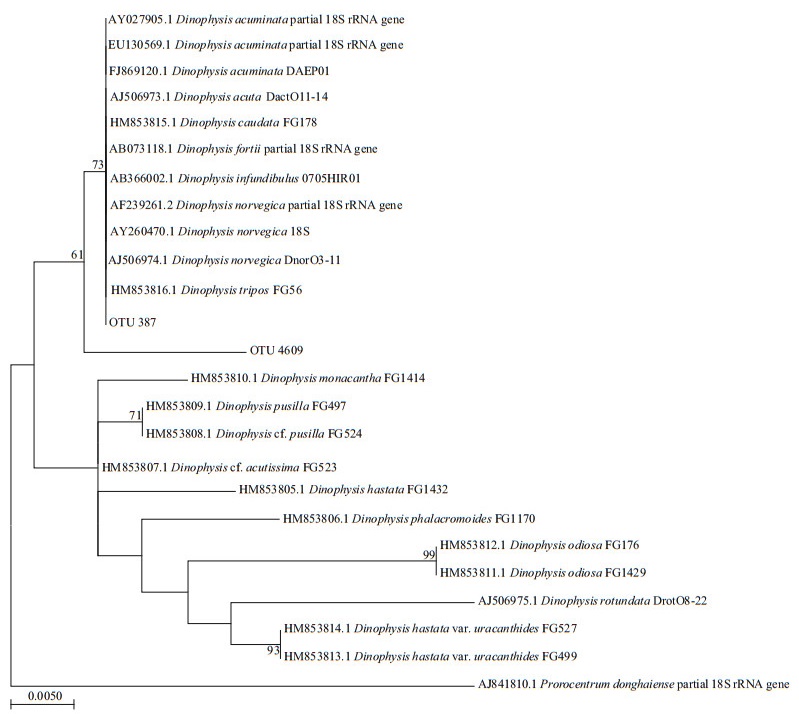
Vegetative cells of Dinophysis were observed in net-concentrated phytoplankton samples, and the species could be identified primarily as D. caudata. Based on the phylogenetic analysis of V4 sequences for Dinophysis spp., they could be roughly divided into two groups (Fig. 7). Toxin-producing species, such as D. acuminata, D. acuta, D. caudata, D. fortii, D. infundibulus, D. norvegica, and D. tripos were grouped in the same cluster. Two OTUs derived from high throughput sequencing in this study were grouped together within this cluster, with high identity (> 98%). However, it is impossible to further identify the species based on the high throughput sequencing results using V4 region of the LSU.
|
| Fig.7 Phylogenetic tree established for species in genus Dinophysis and the OTUs derived from high throughput sequencing results of phytoplankton samples in coastal waters of Weihai based on the V4 region of the LSU |
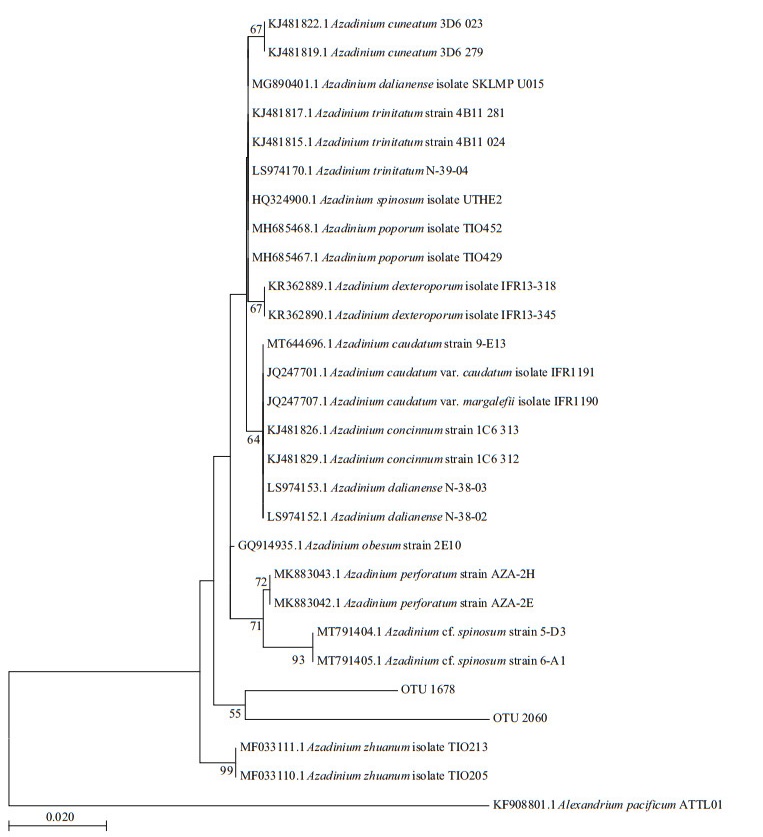
No vegetative cells of Azadinium were observed in net-concentrated phytoplankton samples. However, two OTUs derived from high throughput sequencing were found closely related to the genus Azadinium based on the BLASTing results. However, the phylogenetic analysis did not support the identification of these OTUs to any known species in the genus Azadinium (Fig. 8).
|
| Fig.8 Phylogenetic tree established for species in genus Azadinium and the OTUs derived from high throughput sequencing results of phytoplankton samples in coastal waters of Weihai based on the V4 region of the LSU |
The coastal waters of Weihai, which host nearly 30 bays, is an important mariculture zone in the Yellow Sea. However, there is little information on potential risks of phycotoxins in this region. In this study, although results of single phytoplankton samples at each station were provided, it should not have significant impact on the risk assessment of lipophilic marine toxins in coastal waters of Weihai. Based on the investigation, 4 typical lipophilic marine phycotoxins, namely AZA1/AZA40, 13-DesMe-C, PTX2, and GYM, were detected in phytoplankton and shellfish samples. Among the toxins, PTX2 was detected in both phytoplankton and shellfish samples, and widely distributed in coastal waters of Weihai in autumn. In China, PTX2 was detected for the first time in oyster Magallana gigas from the northern Yellow Sea, and in clam Mactra chinensis and mussel Mytilus galloprovincialis from the East China Sea in 2007 (Liu et al., 2011). The following investigations also demonstrated the importance of PTX2 in the Bohai Sea (Wang et al., 2015; Liu et al., 2017, 2019a) and the South China Sea (Jiang et al., 2017; Liu et al., 2021b). In the Yellow Sea, PTX2 was widely detected in shellfish, phytoplankton, seawater, and sediment (Luo, 2011; Wang et al., 2015; Chen et al., 2017). In most of the investigations, PTX2 was found as the dominant toxin component (Luo, 2011; Liu et al., 2017, 2020, 2021b; Chen et al., 2018). The investigations indicated that PTX2 content in shellfish or phytoplankton samples had notable seasonal variations. An investigation in the coastal waters of Qinhuangdao found that content of PTX2 in phytoplankton reached the maximum from April to May (Sun et al., in press). In Jiaozhou Bay of the Yellow Sea, PTX2 content in phytoplankton had the maximum value in June and July (Luo, 2011), which was basically in accordance with the variation pattern of the PTX2 in seawater (Li et al., 2014; Wu et al., 2019) and in shellfish (Wang et al., 2016). In Daya Bay of the South China Sea, PTX2 in net-haul phytoplankton samples reached the highest level during the autumn and winter (Jiang et al., 2017). Such different seasonal patterns of PTX2 may reflect diverse producers of PTX2, or various seasonal dynamics of the PTX2 producers.
Trace amounts of AZA1/AZA40 was detected in a part of phytoplankton samples in the coastal waters of Weihai. Analogues of AZA toxins, mainly AZA2, have been detected in shellfish (Wu et al., 2018; Liu et al., 2019a), phytoplankton (Li et al., 2017), and suspended particle material (Chen et al., 2018) in the coastal waters of China. AZA1 was also detected in shellfish samples collected from the northern Yellow Sea and the South China Sea (Yao et al., 2010), and in phytoplankton and suspended particle materials in the Bohai Sea and the Yellow Sea (Liu et al., 2017; Chen et al., 2018). However, Krock et al. (2014) has indicated that AZA1 detected in coastal waters of China might be a misidentification of AZA40, an isomer of AZA1 identified in strains of Azadinium poporum isolated from China. AZA1 is the major toxin component produced by Azadinium spinosum in European waters, but it has not been detected in the toxic isolates of Azadinium from the China Seas. So far, there is little information available on the seasonal variation patterns of AZAs in the coastal waters of China. An investigation in the northern Yellow Sea indicated that the highest content of AZA-2 (21.5 μg/kg) was detected in June in Zhangzidao island (Wu et al., 2018).
Both 13-DesMe-C and GYM belong to CIs, a group of toxins encompassing a macrocyclic ring with a conserved cyclic imine group. In this study, trace amounts of 13-DesMe-C and GYM were detected in phytoplankton and shellfish samples respectively. Ji et al. (2018) also detected 13-DesMe-C and GYM in shellfish samples cultured in Rongcheng, Weihai. In previous studies, 13-DesMe-C and GYM have been detected in phytoplankton, seawater, and shellfish samples in the Bohai Sea, Yellow Sea, and South China Sea (Yao et al., 2010; Liu et al., 2017; Ji et al., 2018, 2022; Wu et al., 2019; Liang et al., 2022), and the contents of the two toxins in the Yellow Sea and the Bohai Sea were much lower than the South China Sea and the East China Sea.
In the shellfish samples collected from coastal waters of Weihai in this study, only PTX2 and GYM were detected, and toxin content was very low. Content of PTX2 in the scallop and mussel samples was only 2.63±0.4 μg/kg, which is far below the regulatory limit for seafood safety of the European Union (160 μg/kg). Content of GYM (3.77±0.8 μg/kg) is also much lower than those previously reported in this region (for example, 20.23 μg/kg in an oyster sample from Rongcheng) (Ji et al., 2018). Lipophilic marine toxins with low content in coastal waters of Weihai would not lead to potent risks of seafood consumers. Moreover, PTXs have been removed from the standards for live bivalves in Commission Delegated Regulation (EU) 2021/1374, since the European Food Safety Authority (EFSA) has stated that there is no evidence of adverse effects linked with PTXs (Otero and Silva, 2022). Although CIs have potent neurotoxicity, they are not regulated in most of the countries now. Therefore, it can be addressed that lipophilic marine phycotoxins in coastal waters of Weihai in autumn have little risk to the health of human beings. It should be noted, however, the investigation was only carried out in autumn, while the abundance of toxic algae and content of phycotoxins could be much higher in spring and summer. Therefore, the finding of this study is not enough to reflect the potential risks of lipophilic marine toxins in a comprehensive way, and more time-series investigations are still needed.
4.2 Potential producers of lipophilic marine toxins in coastal waters of WeihaiPTXs are produced by toxic species in genus Dinophysis, which can produce both okadaates (okadaic acid and its analogues dinophysistoxins) and PTXs (Reguera et al., 2012). At present, 12 species in the genera Dinophysis and Phalacroma are capable of producing okadaates or PTXs. In the Yellow Sea and the East China Sea, toxic dinoflagellates Dinophysis acuminata, D. fortii, D. caudata, and Phalacroma rotundatum (=D. rotundata) are widely distributed, and some of them are capable of producing PTX2, OA, and DTX1 (Luo, 2011; Li et al., 2015, 2017; Gao et al., 2017). Therefore, OA and DTXs were often detected simultaneously with PTX2. In this study, however, no OA or DTXs were detected, probably due to the unique species of Dinophysis present in autumn along the coast of Weihai. During the investigation, only D. caudata with low abundance were observed in net-concentrated phytoplankton. Dinophysis caudata is generally considered as a tropical or temperate species, which appear later in autumn following the bloom of D. acuminata complex in spring and summer in temperate waters (Reguera et al., 2012). In the southern part of China, such as Daya Bay, D. caudata was often present as a dominant Dinophysis species (Jiang et al., 2017; Liu et al. 2021b), and PTX2 was detected as a major toxin component in the concentrated phytoplankton samples. Therefore, D. caudata is most likely to be the producer of PTX2 in the coastal waters of Weihai in autumn.
Dinophysis seldom dominates the phytoplankton (Reguera et al., 2012), and it is difficult to make accurate identification of the Dinophysis species simply based on their morphological features. Therefore, some co-occurring species with similar morphological features have been termed as species complex, such as "D. acuminata complex". In this study, high throughput sequencing was used to assist the identification of toxin producer. A total of two OTUs were assigned to Dinophysis species. The target V4 region, however, have very low ability to resolve the different species in genus Dinophysis. The results of phylogenetic analysis grouped Dinophysis species with known V4 sequences into two major groups (Sun et al., in press). Most of the toxic Dinophysis species were grouped together, but it is difficult to further identify the species based on the V4 region. In previous studies, it was also found that ribosomal RNA genes are not applicable to infer taxonomic and/or phylogenetic relationships for Dinophysis spp. (Reguera et al., 2012). These results suggest that the rDNA molecular markers are not appropriate to discriminate Dinophysis spp., and high-resolution plastid or mitochondrial DNA markers should be tested in the future.
Dinoflagellates Karenia selliformis and Alexandrium ostenfeldii/A. peruvianum have been implicated in the biosynthesis of CIs like GYMs and spirolides (SPXs) (Molgó et al., 2017). Dinoflagellates A.ostenfeldii and A.peruvianum produce a large family of SPXs, and Karenia selliformis produces different groups of GYMs including A, B, and C. Recently, it was found that A. ostenfeldii and A. peruvianum also produced GYM analogs (Molgó et al., 2017). The simultaneous occurrence of GYM and 13-DesMe-C suggest they could derive from the same toxic algae. In this study, an OTU was assigned to A. ostenfeldii/ A. andersonii, suggesting that A. ostenfeldii could be the origin of the two CIs. However, no strains of A. ostenfeldii isolated from the coastal waters of China produce GYM or SPX (Gu, 2011). The origin of CIs still needs to be further confirmed.
The Amphidomataceae is an increasingly growing family of Dinophyceae (Salas et al., 2021). AZAs were mainly produced by toxic dinoflagellates from the genera Azadinium (Aza. poporum, Aza. spinosum and Aza. dexteroporum) and Amphidoma (Amph. languida). In this study, two OTUs related to Azadinium were grouped together to form a cluster different from all the known toxic species in the genus Azadinium. The results suggest that unidentified Azadinium species exist in this region, which may account for the AZA1/AZA40 detected in the coastal waters of Weihai.
5 CONCLUSIONIn this study, LC-MS/MS and high throughput sequencing methods were applied to screen for lipophilic marine toxins and their potential producers in coastal waters of Weihai. Four typical lipophilic marine toxins, PTX2, AZA1/AZA4, 13-DesMe-C, and GYM, were detected in phytoplankton and shellfish samples. The content of lipophilic marine toxins in shellfish was low and has little risk to seafood consumers. Based on the microscopy and high throughput sequencing results, it can be deduced that D. caudata and A. ostenfeldii are likely to be the producers of PTX2 and CIs (GYM and 13-DesMe-C). The producer of AZAs could not be determined but may linked to unidentified species in the genus Azadinium.
6 DATA AVAILABILITY STATEMENTThe datasets analyzed during the current study are available from the corresponding author on reasonable request.
7 ACKNOWLEDGMENTThe authors thank Dr. Xiaotian HAN for the information of morphological observation of phytoplankton samples.
Berdalet E, Fleming L E, Gowen R, et al. 2016. Marine harmful algal blooms, human health and wellbeing: challenges and opportunities in the 21st century. Journal of the Marine Biological Association of the United Kingdom, 96(1): 61-91.
DOI:10.1017/S0025315415001733 |
Bodero M, Gerssen A, Portier L, et al. 2018. A strategy to replace the mouse bioassay for detecting and identifying lipophilic marine biotoxins by combining the neuro-2a bioassay and LC-MS/MS Analysis. Marine Drugs, 16(12): 501.
DOI:10.3390/md16120501 |
Cembella A D. 2003. Chemical ecology of eukaryotic microalgae in marine ecosystems. Phycologia, 42(4): 420-447.
DOI:10.2216/i0031-8884-42-4-420.1 |
Chapela M J, Reboreda A, Vieites J M, et al. 2008. Lipophilic toxins analyzed by liquid chromatography-mass spectrometry and comparison with mouse bioassay in fresh, frozen, and processed molluscs. Journal of Agricultural and Food Chemistry, 56(19): 8979-8986.
DOI:10.1021/jf801572j |
Chen J H, Han T Z, Li X T, et al. 2018. Occurrence and distribution of marine natural organic pollutants: lipophilic marine algal toxins in the Yellow Sea and the Bohai Sea, China. Science of the Total Environment, 612: 931-939.
DOI:10.1016/j.scitotenv.2017.08.304 |
Chen J H, Li X, Wang S, et al. 2017. Screening of lipophilic marine toxins in marine aquaculture environment using liquid chromatography-mass spectrometry. Chemosphere, 168: 32-40.
DOI:10.1016/j.chemosphere.2016.10.052 |
Chen Z F, Zhang Q C, Kong F Z, et al. 2019. Resolving phytoplankton taxa based on high-throughput sequencing during brown tides in the Bohai Sea, China. Harmful Algae, 84: 127-138.
DOI:10.1016/j.hal.2019.03.011 |
Cheung M K, Au C H, Chu K H, et al. 2010. Composition and genetic diversity of picoeukaryotes in subtropical coastal waters as revealed by 454 pyrosequencing. The ISME Journal, 4(8): 1053-1059.
DOI:10.1038/ismej.2010.26 |
De Schrijver K, Maes I, De Man L, et al. 2002. An outbreak of diarrhoeic shellfish poisoning in Antwerp, Belgium. Euro Surveillance, 7(10): 138-141.
DOI:10.2807/esm.07.10.00363-en |
Edgar R C. 2013. UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nature Methods, 10(10): 996-998.
DOI:10.1038/nmeth.2604 |
Farabegoli F, Blanco L, Rodríguez L P, et al. 2018. Phycotoxins in marine shellfish: origin, occurrence and effects on humans. Marine Drugs, 16(6): 188.
DOI:10.3390/md16060188 |
Flynn J M, Brown E A, Chain F J J, et al. 2015. Toward accurate molecular identification of species in complex environmental samples: testing the performance of sequence filtering and clustering methods. Ecology and Evolution, 5(11): 2252-2266.
DOI:10.1002/ece3.1497 |
Gao H, An X L, Liu L, et al. 2017. Characterization of Dinophysis acuminata from the Yellow Sea, China, and its response to different temperatures and Mesodinium prey. Oceanological and Hydrobiological Studies, 46(4): 439-450.
DOI:10.1515/ohs-2017-0043 |
Garcêa C, Truan D, Lagos M, et al. 2005. Metabolic transformation of dinophysistoxin-3 into dinophysistoxin-1 causes human intoxication by consumption of O-acyl-derivatives dinophysistoxins contaminated shellfish. The Journal of Toxicological Sciences, 30(4): 287-296.
DOI:10.2131/jts.30.287 |
Gobler C J. 2020. Climate change and harmful algal blooms: insights and perspective. Harmful Algae, 91: 101731.
DOI:10.1016/j.hal.2019.101731 |
Gu H F. 2011. Morphology, phylogenetic position, and ecophysiology of Alexandrium ostenfeldii (Dinophyceae) from the Bohai Sea, China. Journal of Systematics and Evolution, 49(6): 606-616.
DOI:10.1111/j.1759-6831.2011.00160.x |
Hallegraeff G M, Anderson D M, Belin C, et al. 2021. Perceived global increase in algal blooms is attributable to intensified monitoring and emerging bloom impacts. Communications Earth & Environment, 2: 117.
|
He X P, Chen J H, Wu D N, et al. 2019. Distribution characteristics and environmental control factors of lipophilic marine algal toxins in Changjiang estuary and the adjacent East China Sea. Toxins, 11(10): 596.
DOI:10.3390/toxins11100596 |
He X P, Chen J H, Wu D N, et al. 2020. Occurrence, distribution, source, and influencing factors of lipophilic marine algal toxins in Laizhou Bay, Bohai Sea, China. Marine Pollution Bulletin, 150: 110789.
DOI:10.1016/j.marpolbul.2019.110789 |
Hou S Y, Xiang C H, Dong H W, et al. 2018. Assessment of carbon sink and shellfish culture from 2007 to 2016 in Weihai. Fisheries Science & Technology Information, 45(1): 48-51.
(in Chinese) |
Howard M D A, Smith J, Caron D A, et al. 2022. Integrative monitoring strategy for marine and freshwater harmful algal blooms and toxins across the freshwater-to-marine continuum. Integrated Environmental Assessment and Management.
DOI:10.1002/ieam.4651 |
Ianora A, Bentley M G, Caldwell G S, et al. 2011. The relevance of marine chemical ecology to plankton and ecosystem function: an emerging field. Marine Drugs, 9(9): 1625-1648.
DOI:10.3390/md9091625 |
Ji Y, Hu Y, Song J L, Chen H D, et al. 2018. Characteristics of components and regional distribution of lipophilic shellfish toxins in bivalves cultured along the Chinese Coast in spring. Chinese Fishery Quality and Standards, 8(4): 15-24.
(in Chinese with English abstract) |
Ji Y, Yan G W, Wang G X, et al. 2022. Prevalence and distribution of domoic acid and cyclic imines in bivalve mollusks from Beibu Gulf, China. Journal of Hazardous Materials, 423: 127078.
DOI:10.1016/j.jhazmat.2021.127078 |
Jiang T, Liu L, Li Y, et al. 2017. Occurrence of marine algal toxins in oyster and phytoplankton samples in Daya Bay, South China Sea. Chemosphere, 183: 80-88.
DOI:10.1016/j.chemosphere.2017.05.067 |
Krock B, Tillmann U, Witt M, et al. 2014. Azaspiracid variability of Azadinium poporum (Dinophyceae) from the China Sea. Harmful Algae, 36: 22-28.
DOI:10.1016/j.hal.2014.04.012 |
Kumar S, Stecher G, Li M, et al. 2018. MEGA X: molecular evolutionary genetics analysis across computing platforms. Molecular Biology and Evolution, 35(6): 1547-1549.
DOI:10.1093/molbev/msy096 |
Landsberg J H. 2002. The effects of harmful algal blooms on aquatic organisms. Reviews in Fisheries Science, 10(2): 113-390.
DOI:10.1080/20026491051695 |
Li A F, Ma J G, Cao J J, et al. 2012. Toxins in mussels (Mytilus galloprovincialis) associated with diarrhetic shellfish poisoning episodes in China. Toxicon, 60(3): 420-425.
DOI:10.1016/j.toxicon.2012.04.339 |
Li A F, Sun G, Qiu J B, et al. 2015. Lipophilic shellfish toxins in Dinophysis caudata picked cells and in shellfish from the East China Sea. Environmental Science and Pollution Research, 22(4): 3116-3126.
DOI:10.1007/s11356-014-3595-z |
Li J, Ruan Y F, Mak Y L, et al. 2021. Occurrence and trophodynamics of marine lipophilic phycotoxins in a subtropical marine food web. Environmental Science & Technology, 55(13): 8829-8838.
|
Li M H, Sun G, Qiu J B, et al. 2017. Occurrence and variation of lipophilic shellfish toxins in phytoplankton, shellfish and seawater samples from the aquaculture zone in the Yellow Sea, China. Toxicon, 127: 1-10.
DOI:10.1016/j.toxicon.2016.12.009 |
Li X, Li Z Y, Chen J H, et al. 2014. Detection, occurrence and monthly variations of typical lipophilic marine toxins associated with diarrhetic shellfish poisoning in the coastal seawater of Qingdao City, China. Chemosphere, 111: 560-567.
DOI:10.1016/j.chemosphere.2014.05.006 |
Li Z X, Guo M M, Yang S G, et al. 2010. Investigation of pectenotoxin profiles in the Yellow Sea (China) using a passive sampling technique. Marine Drugs, 8(4): 1263-1272.
DOI:10.3390/md8041263 |
Liang Y B, Li A F, Chen J H, et al. 2022. Progress on the investigation and monitoring of marine phycotoxins in China. Harmful Algae, 111: 102152.
DOI:10.1016/j.hal.2021.102152 |
Lin C, Liu Z S, Tan C Y, et al. 2015. Contamination of commercially available seafood by key diarrhetic shellfish poisons along the coast of China. Environmental Science and Pollution Research, 22(2): 1545-1553.
DOI:10.1007/s11356-014-3494-3 |
Liu C, Ji Y, Zhang L, et al. 2021b. Spatial distribution and source of biotoxins in phytoplankton from the South China Sea, China. Journal of Hazardous Materials, 418: 126285.
DOI:10.1016/j.jhazmat.2021.126285 |
Liu R Y, Liang Y B, Wu X L, et al. 2011. First report on the detection of pectenotoxin groups in Chinese shellfish by LC-MS/MS. Toxicon, 57(7-8): 1000-1007.
DOI:10.1016/j.toxicon.2011.04.002 |
Liu Y, Chen Z F, Wang J X, et al. 2021a. Distribution characteristics of lipophilic marine phycotoxins in the sediment: A case study in Jiaozhou Bay, China. Marine Pollution Bulletin, 162: 111908.
DOI:10.1016/j.marpolbul.2020.111908 |
Liu Y, Yu R C, Kong F Z, et al. 2017. Lipophilic marine toxins discovered in the Bohai Sea using high performance liquid chromatography coupled with tandem mass spectrometry. Chemosphere, 183: 380-388.
DOI:10.1016/j.chemosphere.2017.05.073 |
Liu Y, Yu R C, Kong F Z, et al. 2019a. Contamination status of lipophilic marine toxins in shellfish samples from the Bohai Sea, China. Environmental Pollution, 249: 171-180.
DOI:10.1016/j.envpol.2019.02.050 |
Liu Y, Zhang P, Du S, et al. 2019b. Sediment as a potential pool for lipophilic marine phycotoxins with the case study of Daya Bay of China. Marine Drugs, 17(11): 623.
DOI:10.3390/md17110623 |
Liu Y, Zhang P, Du S, et al. 2020. Occurrence and distribution of lipophilic phycotoxins in a subtropical bay of the South China Sea. Chemosphere, 243: 125352.
DOI:10.1016/j.chemosphere.2019.125352 |
Liu Y. 2017. Studies of Phycotoxin Contamination in the Coastal Waters of China and Preparation of Toxin Reference Materials. Institute of Oceanology, Chinese Academy of Sciences, Qingdao. (in Chinese with English abstract)
|
Luo X. 2011. Population Dynamics and Toxin Production of Dinophysis Species in the Coastal Area of Qingdao. Institute of Oceanology, Chinese Academy of Sciences, Qingdao. (in Chinese with English abstract)
|
Molgó J, Marchot P, Aráoz R, et al. 2017. Cyclic imine toxins from dinoflagellates: a growing family of potent antagonists of the nicotinic acetylcholine receptors. Journal of Neurochemistry, 142: 41-51.
|
Nicolas J, Hoogenboom R L A P, Hendriksen P J M, et al. 2017. Marine biotoxins and associated outbreaks following seafood consumption: prevention and surveillance in the 21st century. Global Food Security, 15: 11-21.
DOI:10.1016/j.gfs.2017.03.002 |
Otero P, Silva M. 2022. Emerging marine biotoxins in European waters: potential risks and analytical challenges. Marine Drugs, 20(3): 199.
DOI:10.3390/md20030199 |
Reguera B, Velo-Suárez L, Raine R, et al. 2012. Harmful Dinophysis species: a review. Harmful Algae, 14: 87-106.
DOI:10.1016/j.hal.2011.10.016 |
Salas R, Tillmann U, Gu H F, et al. 2021. Morphological and molecular characterization of multiple new Azadinium strains revealed a high diversity of non-toxigenic species of Amphidomataceae (Dinophyceae) including two new Azadinium species in Irish waters, North East Atlantic. Phycological Research, 69(2): 88-115.
DOI:10.1111/pre.12448 |
Shumway S E. 1990. A review of the effects of algal blooms on shellfish and aquaculture. Journal of the World Aquaculture Society, 21(2): 65-104.
DOI:10.1111/j.1749-7345.1990.tb00529.x |
Sournia A. 1978. Phytoplankton Manual. UNESCO, Paris.
|
Sun H X, Tang W J, Liu C et al. Contamination status and origins of lipophilic marine toxins in Qinhuangdao coastal waters. Marine Sciences, in press. (in Chinese with English abstract)
|
Wang X Z, Cheng Y, Li N, et al. 2016. Occurrence and seasonal variations of lipophilic marine toxins in commercial clam species along the coast of Jiangsu, China. Toxins, 8(1): 8.
DOI:10.3923/rjt.2016.8.14 |
Wang Y L, Chen J H, Li Z Y, et al. 2015. Determination of typical lipophilic marine toxins in marine sediments from three coastal bays of China using liquid chromatography-tandem mass spectrometry after accelerated solvent extraction. Marine Pollution Bulletin, 101(2): 954-960.
DOI:10.1016/j.marpolbul.2015.10.038 |
Winnepenninckx B, Backeljau T, De Wachter R. 1993. Extraction of high molecular weight DNA from molluscs. Trends in Genetics, 9(12): 407.
DOI:10.1016/0168-9525(93)90102-N |
Wu D N, Chen J H, He X P, et al. 2019. Distribution, partitioning, and seasonal variation of lipophilic marine algal toxins in aquatic environments of a typical semi-closed mariculture bay. Environmental Pollution, 255: 113299.
DOI:10.1016/j.envpol.2019.113299 |
Wu D N, Chen J H, Wang J M, et al. 2020. Monitoring and warning of lipophilic marine algal toxins in mariculture zone based on toxin profiles of phytoplankton. Ecotoxicology and Environmental Safety, 197: 110647.
DOI:10.1016/j.ecoenv.2020.110647 |
Wu H Y, Luan Q S, Guo M M, et al. 2018. Phycotoxins in scallops (Patinopecten yessoensis) in relation to source, composition and temporal variation of phytoplankton and cysts in North Yellow Sea, China. Marine Pollution Bulletin, 135: 1198-1204.
DOI:10.1016/j.marpolbul.2018.08.045 |
Yao J H, Tan Z J, Zhou D Q, et al. 2010. Determination of azaspiracid-1 in shellfishes by liquid chromatography with tandem mass spectrometry. Chinese Journal of Chromatography, 28(4): 363-367.
(in Chinese with English abstract) DOI:10.3724/SP.J.1123.2010.00363 |
Zhou M J, Li J, Luckas B, et al. 1999. A recent shellfish toxin investigation in China. Marine Pollution Bulletin, 39(1-12): 331-334.
DOI:10.1016/S0025-326X(99)00026-0 |
Zimmermann J, Jahn R, Gemeinholzer B. 2011. Barcoding diatoms: evaluation of the V4 subregion on the 18S rRNA gene, including new primers and protocols. Organisms Diversity & Evolution, 11(3): 173-192.
|
 2022, Vol. 40
2022, Vol. 40



