Institute of Oceanology, Chinese Academy of Sciences
Article Information
- ZHENG Wenxu, LI Renhui, QIN Wenli, CHEN Binbin, WANG Min, GUAN Wanchun, ZHANG Xiaoling, YANG Qiao, ZHAO Min, MA Zengling
- Tidal water exchanges can shape the phytoplankton community structure and reduce the risk of harmful cyanobacterial blooms in a semi-closed lake
- Journal of Oceanology and Limnology, 40(5): 1868-1880
- http://dx.doi.org/10.1007/s00343-022-2032-2
Article History
- Received Jan. 24, 2022
- accepted in principle Mar. 23, 2022
- accepted for publication Apr. 2, 2022
2 National and Local Joint Engineering Research Center of Ecological Treatment Technology for Urban Water Pollution, Wenzhou University, Wenzhou 325035, China;
3 Department of Marine Biotechnology, School of Laboratory Medicine and Life Science, Wenzhou Medical University, Wenzhou 325035, China;
4 Marine Science and Technology College, Zhejiang Ocean University, Zhoushan 316022, China;
5 School of Petrochemical Engineering and Environment, Zhejiang Ocean University, Zhoushan 316022, China
In estuaries, tides play a vital role in aquatic ecosystems by bringing in seawater with nutrients, salt, and microorganisms, and mixing with freshwater (Choi et al., 2017). Such complex processes can result in high-productivity areas that act as habitats, and feeding areas for many species, such as fish, mammals, and birds (Wu et al., 2021; Yi et al., 2021). Moreover, estuarine islands are vital for the maintenance of biodiversity in the estuarine ecosystem because they provide a transfer point for migratory birds and mammals, and greatly contribute to the protection of freshwater (Li et al., 2021). Lakes play an important role in the control of flooding, water supply, and recreational activities, and they are also the most vulnerable ecological systems that can be disturbed by many natural factors and anthropological activities (Xu et al., 2021a).
Lakes in estuarine islands are a kind of extraordinary natural lake system isolated from the surrounding water. They are affected by the exchange of tidal water and play an important role in the island ecosystem balance. Normally, their elevations are higher than the outside saline water, but the fresh water in the lakes can exchange with the outside brackish water as a result of the tidal movement. The exchange will result in water disturbance, physicochemical alteration, salinity invasion, nutrient input, and biological exchange; and these processes may influence the phytoplankton community structure in regional ecosystems (Ramond et al., 2021). Therefore, lakes in estuarine island are ideal places to study the effects of tide-driven water exchange on the phytoplankton community structure due to their special geographical location, small area, and semi-isolated status. In addition, eutrophication, freshwater salinization, and bio-invasion are becoming heavy environmental stresses for the estuarine island lakes because of anthropogenic and natural causes (Cordeiro et al., 2020; Mo et al., 2021).
Phytoplankton plays a central role in aquatic ecosystems in driving biogeochemical cycles through their nutrient uptake and primary biomass production for supporting the aquatic food webs (Cardinale, 2011; Striebel et al., 2012; Worden et al., 2015). However, if a particular phytoplankton species grows excessively and forms algal blooms, such a massive quantity of algal cells will hinder the sunlight across the water surface and deplete dissolved oxygen, consequently suffocating the aquatic organisms and destroying the balance of the ecosystem (Paerl and Huisman, 2008; Huisman et al., 2018; Pyo et al., 2021). Phytoplankton community responds rapidly to the changes in the environmental conditions of the habitat by altering the factors such as species composition, dominant species, and cell abundance (Griffiths et al., 2016; Zhang et al., 2021a). Growth of phytoplankton is restricted by both biotic and abiotic factors, which include predation, water temperature, salinity, light, nutrient supply, and others (Yu et al., 2008; Zhang et al., 2021b). The phytoplankton community structure of estuaries is mainly affected by extreme rainfall events and nutrient loading (Van Meerssche and Pinckney, 2019; Vizzo et al., 2021). Moreover, hydrography conditions such as tides and coastal freshwater streams play a key role in structuring the estuarine phytoplankton community (Kasai et al., 2010; Paerl et al., 2014; Flores-Melo et al., 2018). Therefore, field investigation on the characteristics of a phytoplankton community can provide a visual result of the environmental influence (Rao et al., 2021). In China, harmful algal blooms formed by cyanobacteria (CyanoHABs) have occurred in the lakes, and even in the reservoirs of drinking water as a result of eutrophication and anthropological activities (Guan et al., 2020; Huang et al., 2020). In addition, in recent years, more CyanoHABs capbable of causing various degrees of marked visible discoloration of water in lakes located in Jiangxin Islet are reported. Such a phenomenon has also aroused public concerns.
Tidal backflow may bring extreme shifts to estuarine island lakes for the following reasons. (1) The invasion of brackish water might favor phytoplankton species that are different from those of freshwater due to environment preferences (Kim et al., 2021; Xu et al., 2021b). (2) The water exchange might exacerbate eutrophication because the frequent water turbulence is a trigger to the release of the nutrients trapped in surface of the sediment (Baek et al., 2020; Wirtz and Smith, 2021). (3) A decrease in water transparency caused by the water turbulence can limit light availability for phytoplankton and impede their growth (Domingues et al., 2005). (4) Intrusion of salinity may become the main threat to the salt intolerant algal species by affecting photosystem Ⅱ (PSII) performance and extracellular osmotic pressure (Corsi et al., 2010). (5) Invasive organisms brought by the seawater might cause unpredictable impacts on the phytoplankton habitat through biotic factors, such as predation, competition, and parasitism (Bailey, 2015). However, to our best knowledge, there has been a paucity of studies focusing on the effects of water exchange (caused by tides) on the phytoplankton community structure and algal bloom outbreak in the estuary island lakes.
The Oujiang River originates from the western area of Zhejiang Province and flows into the East China Sea (ECS) and along a large number of islands situated in the river channel. Situated in the middle reaches of the Oujiang River is the Jiangxin Islet. There are two types of lakes in the Jiangxin Islet; a semi-closed lake that can exchange water with the Oujiang River during the tidal period, and a closed lake isolated from the other sources of water, and its water source comes mainly from rainfall. Normally, the elevation of the semi-closed lake, Gongqing Lake, is higher than the water around the islet, and the outside water will rise and flow into the lake during a period of astronomical tides. The closed lake, Bibo Lake, is adjacent to Gongqing Lake and it is completely isolated. Therefore, these two lakes are the ideal research objects for assessing the influences of tidal backflow on the phytoplankton community and the outbreak of algal blooms.
Compared with the characteristics of the phytoplankton community in a completely closed lake, it is reasonable to speculate that the phytoplankton community structure can be shaped by the exchange of water and concomitant physicochemical alterations in a semi-closed lake and the changes may favor the control of harmful cyanobacterial blooms. To test the hypothesis, a nearly one-year bi-weekly field investigation was carried out in the semi-closed Gongqing Lake and nearby closed Bibo Lake from June 29, 2020 to June 14, 2021, during which the changes in phytoplankton community structure and physicochemical parameters were determined.
2 MATERIAL AND METHOD 2.1 Study area and sampling station settingThe Jiangxin Islet (28°03′N, 120°63′E) is in the midstream of the Oujiang River which flows through the northern area of Wenzhou City (Fig. 1b). The annual averaged discharge of the Oujiang River is approximately 470 m3/s, and the maximum discharge can reach 23 000 m3/s, for example, during the flood period in July 1959. The lunar semi-diurnal tide that dominates the estuary during the spring has a tidal range of approximately 6 m (Li et al., 2017; Xu and You, 2017). The semi-closed lake, Gongqing Lake, has a water area of 7.4×104 m2 in water volume of 88 800 m3 and average depth of 1.2 m, and it lies in Jiangxin Islet and connects to the Oujiang River with an exit (Fig. 1c). The brackish water outside the islet flows into the lake through the exit during the astronomical tidal period of the lunar calendar, and after that, the water in the lake returns into the Oujiang River. The closed lake, Bibo Lake beside the Gongqing Lake, has water area of 0.4×104 m2 in water volume of 2 000 m3 and average depth of 0.5 m (Fig. 1c). To investigate the influence of water exchange on the structure of the phytoplankton community at different distances to the exit, five sampling sites were selected at different places (Fig. 1c).

|
| Fig.1 The location of Oujiang River in Zhejiang Province, China (a), the map of Oujiang River estuary (b), and the enlarged map of the Jiangxin Islet, showing Bibo Lake, the semi-closed Gongqing Lake, and the sampling sites (c) |
The water sampling was carried out on the 14th and 29th every month of the Chinese lunar calendar, the days before the astronomical tide. Samples for quantitative analysis of the phytoplankton species were collected from 0.5 m below water surface every station and stored in 1.0-L hydrochloric acid-washed opaque polypropylene bottles. Samples were preserved with Lugol's iodine solution and sit for at least 24 h before analysis. Samples for the analysis of phytoplankton taxonomy were collected by horizontal trawling with 25# plankton net at surface water; and the collected plankton was stored in 100-mL transparent plastic bottles. Water samples for nutrient analysis were stored in a cooler with ice packs, then transported to laboratory and processed within 10 h. Water temperature, dissolved oxygen, and pH were measured in situ using a HACHHD40d portable multi-parameter water quality analyzer (HACH, USA). Salinities were measured in situ using a DDBJ-350F portable conductivity meter (INESA Scientific Instrument, China), and transparency was measured with Secchi disc.
2.3 Determination of nutrients in surface waterTotal nitrogen (TN) and total phosphorus (TP) concentrations were determined using the acidic peroxydisulfate method reported by DElia et al. (1977) and Gales et al. (1966), respectively.
2.4 Determination of chlorophyll-a concentration in surface waterChlorophyll a was collected by filtering 200 mL of water (from surface samples) through a Whatman GF/C filter (47-mm-diameter and 1.2-μm-pore-size) and the filter was then extracted overnight in the dark with absolute methanol at 4 ℃. After centrifugation at 5 000×g for 5 min, the absorption spectra of the supernatants were measured with a spectrophotometer (UV 530; Beckman Coulter, USA). The concentration of chlorophyll a was calculated according to equation of Porra (2002).
2.5 Identification of phytoplankton species and determination of diversity indicesIdentification and quantification of phytoplankton species were performed under a Zeiss Axiolab 5 phase contrast microscopes (Carl Zeiss, Germany). Phytoplankton cells were identified and counted using Groove-type 0.1-mL counting slide with a mold-type grid on its bottom under a working magnification of 400× For each sample, a minimum of 400–500 cells were counted from randomly selected transects at multiple magnifications; and all the samples were counted 2–3 time to improve the accuracy and reproducibility. In addition, to characterize the changes in the phytoplankton community structure, species abundance, species richness, and three other diversity indices were determined. The dominance index (Y) that emphasizes the role of the important species was calculated according to the equation of Mcnaughton (1967). The species richness (R) was calculated according to equation of Margalef (1958). The species diversity (H) was calculated according to equation of Shannon and Weaver (1949). The species evenness (E) was calculated according to equation of Pielou (1967).
Phytoplankton community composition (cell density) at the species level was visualized using non-metric multidimensional scaling (NMDS) based on Bray-Curtis dissimilarities between each pair of samples. Analysis of similarity (ANOSIM) was used to reveal differences in the phytoplankton communities among the sampling sites. The NMDS and ANOSIM analyses were performed using PRIMER v.7.0.21. Graphic displays were performed using Origin 2019 (OriginLab, USA), Adobe Illustration 2020 (Adobe, USA), and Adobe Photoshop 2020 (Adobe, USA).
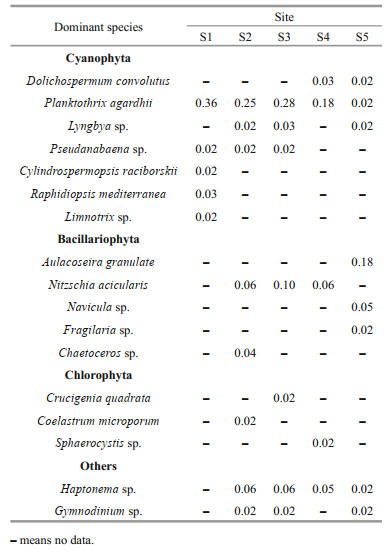
3 RESULT 3.1 Phytoplankton species composition and dominant speciesIn the closed lake (Bibo Lake), 58 species from 7 phyla were identified, and they were Cyanophyta, Bacillariophyta, Chlorophyta, Euglenophyta, Pyrrophyta, Chrysophyta, and Haptophyta (Fig. 2a). Among them, Chlorophyta was the most diverse, consisting of 20 species whereas Cyanobacteria and Bacillariophyta were the second and third most diverse, having 17 and 12 species, respectively (Fig. 2a). In the semi-closed lake (Gongqing Lake), 60 species were identified, of which 21, 15, and 14 species belong to Chlorophyta, Cyanophyta, and Bacillariophyta, respectively (Fig. 2b). In the Oujiang River, 31 species were identified (Fig. 2c), and the most diverse group was diatoms (Bacillariophyta), consisting of 14 species. In addition, in the semi-closed lake, the number of phytoplankton species showed a decreasing trend as 57, 54, and 53 species were found in site S2 (farther away from the lake exit) (Fig. 2d), S3 (Fig. 2e), and S4 (closer to the lake exit) (Fig. 2f), respectively.
|
| Fig.2 Composition of phytoplankton species in the closed (a) and semi-closed (b) lakes on the Jiangxin Islet and in the Oujiang River near the exit (c); d, e, and f show the phytoplankton species composition in semi-closed Gongqing Lake at the sampling sites from the farthest to the closest from the exit |
In the closed lake, the 5 dominant species (Y≥0.02) were all filamentous cyanobacteria, and the most dominant species was Planktothrix agardhii, in dominance of 0.36 (Table 1). In the semi-closed lake and the Oujiang River, the number of dominant species increased, and they belonged to different taxonomical groups, i.e., cyanobacteria, diatoms, green algae.
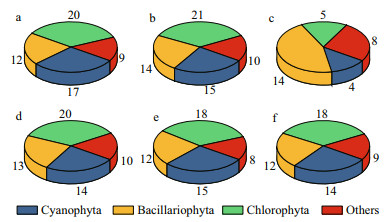
In the closed lake, the monthly abundance of total phytoplankton species was between 1.90×107 and 7.26×108 cells/L, with an annual average abundance of 1.35×108 cells/L during the study period (Fig. 3a). The abundance of total phytoplankton species was the highest in autumn and lowest in spring. In addition, during the entire study period, the abundance of cyanobacteria accounted for more than 96% of the total phytoplankton (Fig. 3a). In the semi-closed lake, the monthly abundance of total phytoplankton species was between 6.22?06 and 4.05?07 cells/L, on annual average abundance of 2.10?07 cells/L (Fig. 3b). Cyanobacteria, diatoms, and green algae appeared throughout the year on average abundance of 1.08×107, 3.55×106, and 2.24×106 cells/L (Fig. 3b), respectively. In the Oujiang River, the monthly abundance of total phytoplankton species was between 1.17×104 and 1.07×106 cells/L, on annual average of 2.04×105 cells/L (Fig. 3c). The average abundance of total phytoplankton species in the closed lake was 6 times higher than in the semi-closed one, and nearly 5 800 times higher than in the Oujiang River (Fig. 3a–c). In other words, the average dominance of cyanobacteria in the closed lake was 0.96, and those in the semi-closed lake and the Oujiang River were 0.51 and 0.22, respectively.
|
| Fig.3 Phytoplankton abundance in the closed (a) and semi-closed (b) lakes on the Jiangxin Islet and in the Oujiang River near the exit (c); d, e, and f show the phytoplankton species composition in the semi-closed Gongqing Lake at the sampling sites from the farthest to the closest from the exit |
Sites S2 (Fig. 3d), S3 (Fig. 3e), and S4 (Fig. 3f) in the semi-closed lake showed similar patterns in phytoplankton abundance, and the annual averages were 2.52×107, 1.93×107, and 1.84×107 cells/L (Fig. 3d–f), respectively. The total phytoplankton population consisted of cyanobacteria, green algae, diatoms, and other species, and their abundance exhibited a decreasing trend from S2 to S4 (Fig. 3d–f).
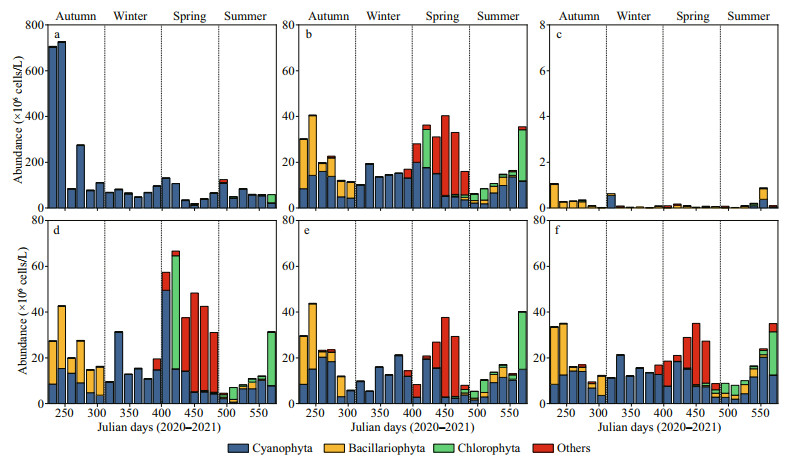
3.3 Relative abundance of different phytoplankton groupsIn the closed lake, cyanobacteria were the most dominant group during the entire study period (Fig. 4a). The relative abundance of diatoms, green algae, and other species accounted for only 0.79%, 5.17%, and 1.14%, respectively. Filamentous cyanobacterium P. agardhii was the most dominant species from September 2020 to February 2021 (Julian day 304?65), and peaked at 1.20×108 cells/L and a relative abundance of 98.8% on January 26, 2021 (Julian day 392).
|
| Fig.4 Relative abundance of dominant phytoplankton groups in the closed (a) and semi-closed (b) lakes situated in the Jiangxin Islet and that in the Oujiang River near the exit (c); d, e, and f show the phytoplankton species composition in the semi-closed Gongqing Lake at the sampling sites from the farthest to the closest from the exit |
In the semi-closed lake, cyanobacteria dominated, and the relative abundance of other algae increased significantly during the study period (Fig. 4b). Planktothrix agardhii was replaced by Haptonema sp. (Haptophyta) as the most abundant species on January 29, 2021 (Julian day 436). In addition, diatoms began to dominate in summer and autumn. Among them, N. acicularis was the most dominant species on June 29, 2020 (Julian day 231) with the highest abundance of 2.15×107 cells/L, accounting for a relative abundance of 72%. In addition, the brackish water diatom Chaetoceros sp. began to be dominated in autumn and reached a peak abundance of 7.44×107 cells/L, equivalent to a relative abundance of 63.4%. Green algae dominated in winter and summer, and Crucigenia quadrata, Sphaerocystis sp., and Coelastrum microporum assumed the dominant position alternately (Fig. 4b).
In the Oujiang River, the diatom Aulacoseira granulate was the most dominant species during most of the study period, with the highest abundance of 4.42×105 cells/L and a relative abundance of 60.5%. However, in some of the months, cyanobacteria (Dolichospermum convolutus), diatom (Navicula sp.), euglena (Gymnodinium sp.), and haptonema (Haptonema sp.) also became the dominant species in the Oujiang River (Fig. 4c).
Furthermore, for the sampling stations S2 (Fig. 4d), S3 (Fig. 4e), and S4 (Fig. 4f), cyanobacteria were still the dominant phytoplankton during the study period. Nevertheless, diatoms, green algae, and other algae (especially Haptonema sp. and Gymnodinium sp.) were the dominant groups for nearly half of the study time.
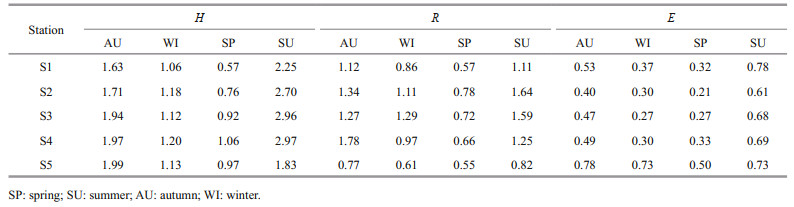
3.4 Diversity indices of phytoplankton speciesThe diversity index (H) of the phytoplankton species in the closed lake, the semi-closed lake, and the Oujiang River was 0.57–2.25, 0.76–2.97, and 0.97–1.99, respectively (Table 2). In addition, the diversity index showed a season-dependent pattern, with the highest H value in summer, followed by autumn, winter, and spring. For the sampling sites, the order of increasing H index was S2 < S3 < S4. The richness (R) in the closed lake, the semi-closed lake, and the Oujiang River was 0.57–1.12, 0.66–1.78, and 0.55–0.82, respectively, displaying a similar trend in temporal and spatial changes to that of H. The evenness (E) of phytoplankton species in the closed, the semi-closed lakes and the Oujiang River was 0.32–0.78, 0.21–0.69, and 0.50–0.78, respectively, during the study period and it showed the same season-dependent patterns to that of H (Table 2).
|
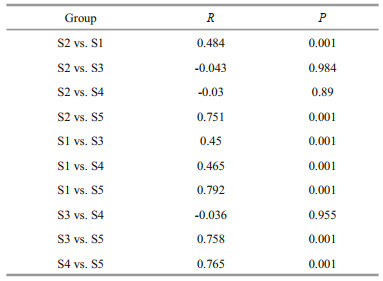
Phytoplankton communities were closely clustered, and the community composition at S1 and S5 sites were significantly separated from the other sites (Fig. 5; Table 3). Although the two lakes are very close with a straight-line distance of about 50 m, the phytoplankton communities in the closed lake were significantly different from those found in the semi-closed lake. Furthermore, the phytoplankton community at S2 was more similar to that at S1 than those at other sampling sites in the semi-closed lake. However, the phytoplankton communities among sampling sites in the semi-closed lake did not show a significant difference.

|
| Fig.5 Non-metric multidimensional scaling (NMDS) of phytoplankton community composition in the five sampling sites in the closed lake (S1), semi-closed lake (S2, S3, S4), and the Oujiang River near the exit (S5) |
|
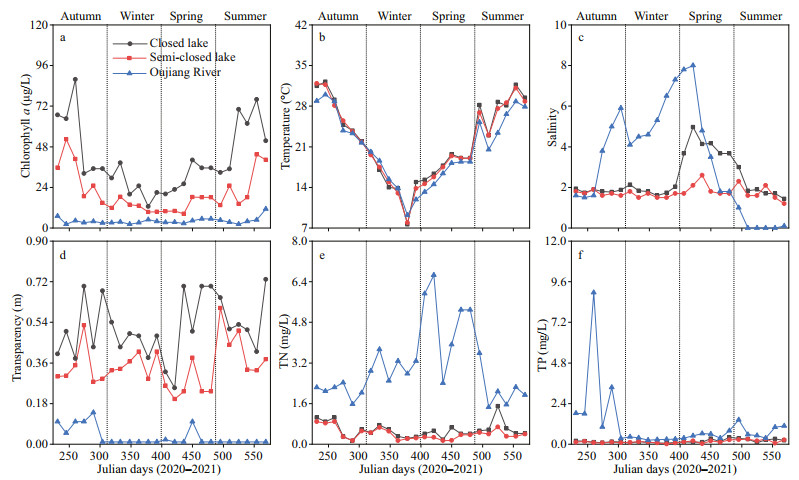
The concentrations of chlorophyll a in the water at the surface of both lakes and the Oujiang River exhibited a similar trend with a peak in summer. In addition, during a same sampling time, the chlorophyll-a concentration in the closed lake was higher than that in the semi-closed one, and both were above that of the Oujiang River (Fig. 6a). The concentration of chlorophyll a in the closed lake, the semi-closed lake, and the Oujiang River were 12.7–87.8, 8.4–52.4, and 2.2–11.3 μg/L, respectively.
|
| Fig.6 Water environmental parameters in the closed lake, the semi-closed lake, and the Oujiang River |
Surface water temperature presented an obvious seasonal pattern and showed no significant differences among different sites (Fig. 6b). Salinity in the Oujiang River was higher than that in the semi-enclosed lake, and both were higher than that in the enclosed lake except in summer due to high flooding tides (Fig. 6c). The degree of water transparency in the closed lake was higher than that in the semi-closed lake, and both were much higher than that in the Oujiang River (Fig. 6d). A similar concentration of TN was found in the water of both lakes, and this TN concentration was much lower than that detected in Oujiang River during the entire study period (Fig. 6e). However, there was no significant difference in TP concentration in the water between the two lakes, and the water of both lakes showed a similar TP concentration to that found in the Oujiang River except for autumn (Fig. 6f).
4 DISCUSSIONPhytoplankton communities in freshwater ecosystems usually exhibit a stronger response to environmental changes and are considered good indicators of environmental change and ecosystem status because of their fast responses to environmental disturbances (Liu et al., 2015; Xue et al., 2018). The mixing of water bodies is also considered an effective method for controlling cyanobacterial blooms (Visser et al., 1996, 2016). In the present study, we found that the abundance, diversity, and dominant phytoplankton species in the semi-closed lake were profoundly affected by the water exchange caused by tides compared with those in the closed lake. In addition, the overwhelmingly dominant position of cyanobacteria throughout the year was weakened and tended to be replaced by diatoms and green algae in some of the months, thereby reducing the risk of cyanobacterial blooms.
The tide-dependent water exchange had little effect on the species composition of phytoplankton community between the closed and semi-closed lakes, but it exerted significant effects on the abundance, dominance, and diversity of the dominant species. It is well known that gas vesicles contained in the cyanobacterial cells can provide the cells with buoyancy in an aqueous environment, and give them an advantage when competing for light and CO2 (Walsby, 1975; Pfeifer, 2012). Therefore, dense cyanobacterial blooms usually occur in lakes and reservoirs with stagnant waters, little wind mixing, and small fluctuations in the water level (Huisman, 2018). In addition, if the vertical mixing rates exceed the flotation velocities of the cyanobacteria, then it can no longer benefit from the gas vesicles that provide buoyancy, and they tend to be replaced by diatoms and green algae (Huisman et al., 2004; Visser et al., 2016).
In the present study, the numbers of species and taxonomical groups of phytoplankton found in the closed and semi-closed lakes are similar. However, the abundance of the total phytoplankton and dominance of cyanobacteria profoundly decreased and those of diatoms and green algae appreciably increased in the semi-closed lake compared with the closed lake, and the trends were more obvious in Site S4 near the exit where brackish water of the Oujiang River flowed into the semi-closed lake during the period of astronomical tides. The diversity index H and richness index R of the phytoplankton species in the semi-closed lake were higher than those in the closed one, while the evenness index E showed the opposite trend. This suggested that the exchange of water in the semi-closed lake significantly increased the diversity of the phytoplankton community and reduced the cyanobacterial dominance, and this could decrease the risk of harmful cyanobacterial blooms (Gomaa et al., 2018). In addition, there was a distinct difference between the compositions of the phytoplankton communities in the two lakes, with the highest degree of similarity between S1 and S2 when compared with the other sampling sites in the semi-closed lake. These differences showed an obvious effect of the tidal water exchange on the phytoplankton community of the semi-closed lake. These changes could be attributed to the turbulence caused by the water exchange. In addition, it is reported that cyanobacteria are more easily replaced by diatoms and green algae under turbulence conditions (Yang et al., 2020). Furthermore, the development of cyanobacterial blooms takes time, shortening the residence time by increasing the water flow could weaken their competitive advantage, thereby, preventing the outbreak of cyanobacterial blooms (Verspagen et al., 2006; Mitrovic et al., 2011).
Chlorophyll a is generally regarded as an effective indicator to algal blooms (Guo et al., 2018; Ding et al., 2020), and a concentration of 10 μg/L is usually regarded as the threshold for triggering algal bloom although some studies have selected higher thresholds, such as 20 μg/L and 30 μg/L (Wu and Xu, 2011; Liao et al., 2021). In the present study, the chlorophyll-a concentration in the water near the surface in the closed lake was much higher than 10 μg/L for most of the study period, and reached a peak of 87.8 μg/L in summer. This indicated that cyanobacterial blooms might occur all year round because the abundance of cyanobacteria detected during a one-year period was more than 96% that of the total abundance. Furthermore, the chlorophyll-a concentration was higher than the threshold almost throughout the year in the semi-closed lake. In contrast, the chlorophyll-a concentration in the Oujiang River was below the threshold during the study period. Therefore, the decrease in chlorophyll-a concentration and phytoplankton abundance in the semi-closed lake could be attributed to the exchange of water between the semi-closed lake and the Oujiang River.
Water temperature is always the main environmental factor affecting the growth of cyanobacterial species; and the global warming had favored the outbreak of cyanobacterial blooms (Huisman et al., 2018; Yan et al., 2020). However, in this study, water temperature varied in the same pattern and no significant difference was detected among the closed lake, the semi-closed lake, and the Oujiang River. Therefore, the significant decreases in phytoplankton abundance, chlorophyll-a concentration, and diversity of dominant phytoplankton species in the semi-closed lake was unlikely to be caused by variation of water temperature.
On the other hand, the outbreak of algal bloom is mainly triggered by eutrophication. In general, TN and TP concentrations in stagnant water that exceed the eutrophication threshold of 0.2 mg/L and 0.02 mg/L, respectively, would indicate a high risk of algal bloom (Carey and Migliaccio, 2009; Guo et al., 2017; Rice and Westerhoff, 2017; Lv et al., 2020). All the TN and TP concentrations in the two lakes and Oujiang River were above the eutrophication thresholds, and were much higher in the Oujiang River than in the two lakes, indicating a much higher level of nutrients in the Oujiang River. Therefore, it was unlikely that the very low phytoplankton abundance and chlorophyll-a concentration in semi-closed lakes were caused by nutrient changes.
However, in comparison with that in the closed lake, the salinity in the semi-closed lake was significantly higher but the transparency significantly lower. In addition, it is reported that cyanobacteria are more sensitive to salinity increase than diatoms and green algae, and many cyanobacterial species are able to grow in freshwater only (Babu et al., 2020; Wiśniewska et al., 2021), thus the decrease in cyanobacterial abundance observed in the semi-closed lake was the result of higher salinity due to the mixing with seawater. The lower transparency in the semi-closed lake caused by water exchange with the Oujiang River suppressed the growth of phytoplankton and thus decreased the phytoplankton abundance and chlorophyll-a concentration. Therefore, it is reasonable to speculate that the changes in salinity and transparency in the semi-closed lake shaped the phytoplankton community structure and reduced the risk of cyanobacterial blooms to a certain extent.
5 CONCLUSIONThe water exchange caused by tidal movement significantly decreased the dominance of cyanobacterial species, and thus rose the dominance of diatoms and green algae in the semi-closed lake. In addition, the diversity index and richness index of the phytoplankton species in the semi-closed lake were higher than those in the closed lake, while the evenness index showed the opposite trend, indicating that water exchange was beneficial to the growth of diatoms and green algae, thereby increasing the diversity of phytoplankton species. The NMDS and ANOSIM analysis also illustrated a significant difference in phytoplankton community between the two lakes. The high salinity and low transparency of water in the semi-closed lake were duo to the variation in phytoplankton community structure, which reduced the risk of cyanobacterial bloom in the lake. These findings provided a good example and insightful clue that the outbreak of cyanobacterial blooms could be prevented and controlled by applying hydrodynamic methods.
6 DATA AVAILABILITY STATEMENTThe datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
7 ACKNOWLEDGMENTThe authors highly appreciate Dr. Alan K CHANG (Wenzhou University) for his effort in the linguistic revision of the manuscript, and the anonymous reviewers for their very useful and insightful comments.
Babu R, Park N, Nam B. 2020. Regional and well-scale indicators for assessing the sustainability of small island fresh groundwater lenses under future climate conditions. Environmental Earth Sciences, 79(1): 47.
DOI:10.1007/s12665-019-8773-3 |
Baek S H, Lee M, Park B S, et al. 2020. Variation in phytoplankton community due to an autumn typhoon and winter water turbulence in southern Korean coastal waters. Sustainability, 12(7): 2781.
DOI:10.3390/su12072781 |
Bailey S A. 2015. An overview of thirty years of research on ballast water as a vector for aquatic invasive species to freshwater and marine environments. Aquatic Ecosystem Health & Management, 18(3): 261-268.
DOI:10.1080/14634988.2015.1027129 |
Cardinale B J. 2011. Biodiversity improves water quality through niche partitioning. Nature, 472(7341): 86-89.
DOI:10.1038/nature09904 |
Carey R O, Migliaccio K W. 2009. Contribution of wastewater treatment plant effluents to nutrient dynamics in aquatic systems: a review. Environmental Management, 44(2): 205-217.
DOI:10.1007/s00267-009-9309-5 |
Choi B J, Lee A J, Choi J S, et al. 2017. Influence of the tidal front on the three-dimensional distribution of spring phytoplankton community in the eastern Yellow Sea. Chemosphere, 173: 299-306.
DOI:10.1016/j.chemosphere.2017.01.048 |
Cordeiro R, Luz R, Vilaverde J, et al. 2020. Distribution of toxic cyanobacteria in Volcanic Lakes of the Azores Islands. Water, 12(12): 3385.
DOI:10.3390/w12123385 |
Corsi S R, Graczyk D J, Geis S W, et al. 2010. A fresh look at road salt: aquatic toxicity and water-quality impacts on local, regional, and national scales. Environmental Science & Technology, 44(19): 7376-7382.
DOI:10.1021/es101333u |
D'Elia C F, Steudler P A, Corwin N. 1977. Determination of total nitrogen in aqueous samples using persulfate digestion. Limnologyand Oceanography, 22(4): 760-764.
DOI:10.4319/lo.1977.22.4.0760 |
Ding X K, Guo X Y, Zhang C, et al. 2020. Water conservancy project on the Yellow River modifies the seasonal variation of chlorophyll-a in the Bohai Sea. Chemosphere, 254: 126846.
DOI:10.1016/j.chemosphere.2020.126846 |
Domingues R B, Barbosa A, Galvão H. 2005. Nutrients, light and phytoplankton succession in a temperate estuary (the Guadiana, south-western Iberia). Estuarine, Coastal and Shelf Science, 64(2-3): 249-260.
DOI:10.1016/j.ecss.2005.02.017 |
Flores-Melo X, Schloss I R, Chavanne C, et al. 2018. Phytoplankton ecology during a spring-neap tidal cycle in the southern tidal front of San Jorge Gulf, Patagonia. Oceanography, 31(4): 70-80.
DOI:10.5670/oceanog.2018.412 |
Gales M E Jr, Julian E C, Kroner R C. 1966. Method for quantitative determination of total phosphorus in water. Journal AWWA, 58(10): 1363-1368.
DOI:10.1002/j.1551-8833.1966.tb01703.x |
Gomaa M N, Hannachi I, Carmichael W W, et al. 2018. Low diversity triggers harmful algae bloom (HAB) occurrence adjacent to desalination plants along the Red Sea. Desalination and Water Treatment, 114: 1-12.
DOI:10.5004/dwt.2018.22323 |
Griffiths J R, Hajdu S, Downing A S, et al. 2016. Phytoplankton community interactions and environmental sensitivity in coastal and offshore habitats. Oikos, 125(8): 1134-1143.
DOI:10.1111/oik.02405 |
Guan Q, Feng L, Hou X J, et al. 2020. Eutrophication changes in fifty large lakes on the Yangtze Plain of China derived from MERIS and OLCI observations. Remote Sensing of Environment, 246: 111890.
DOI:10.1016/j.rse.2020.111890 |
Guo J B, Zhang C J, Zheng G C, et al. 2018. The establishment of season-specific eutrophication assessment standards for a water-supply reservoir located in Northeast China based on chlorophyll-a levels. Ecological Indicators, 85: 11-20.
DOI:10.1016/j.ecolind.2017.09.056 |
Guo X Y, Feng C H, Zhang J H, et al. 2017. Role of dams in the phase transfer of antibiotics in an urban river receiving wastewater treatment plant effluent. Science of the Total Environment, 607-608: 1173-1179.
DOI:10.1016/j.scitotenv.2017.07.096 |
Huang J C, Zhang Y J, Arhonditsis G B, et al. 2020. The magnitude and drivers of harmful algal blooms in China's lakes and reservoirs: a national-scale characterization. Water Research, 181: 115902.
DOI:10.1016/j.watres.2020.115902 |
Huisman J, Codd G A, Paerl H W, et al. 2018. Cyanobacterial blooms. Nature Reviews Microbiology, 16(8): 471-483.
DOI:10.1038/s41579-018-0040-1 |
Huisman J, Sharples J, Stroom J M, et al. 2004. Changes in turbulent mixing shift competition for light between phytoplankton species. Ecology, 85(11): 2960-2970.
DOI:10.1890/03-0763 |
Kasai A, Kurikawa Y, Ueno M, et al. 2010. Salt-wedge intrusion of seawater and its implication for phytoplankton dynamics in the Yura Estuary, Japan. Estuarine, Coastal and Shelf Science, 86(3): 408-414.
DOI:10.1016/j.ecss.2009.06.001 |
Kim D, Lim J H, Chun Y, et al. 2021. Phytoplankton nutrient use and CO2 dynamics responding to long-term changes in riverine N and P availability. Water Research, 203: 117510.
DOI:10.1016/j.watres.2021.117510 |
Li W, Hu Z X, Zhang W G, et al. 2017. Influence of provenance and hydrodynamic sorting on the magnetic properties and geochemistry of sediments of the Oujiang River, China. Marine Geology, 387: 1-11.
DOI:10.1016/j.margeo.2017.03.002 |
Li Y C, Chen Z W, Peng C, et al. 2021. Assessment of habitat change on bird diversity and bird-habitat network of a Coral Island, South China Sea. BMC Ecology and Evolution, 21(1): 137.
DOI:10.1186/s12862-021-01865-y |
Liao A R, Han D M, Song X F, et al. 2021. Impacts of storm events on chlorophyll-a variations and controlling factors for algal bloom in a river receiving reclaimed water. Journal of Environmental Management, 297: 113376.
DOI:10.1016/j.jenvman.2021.113376 |
Liu L M, Yang J, Lv H, et al. 2015. Phytoplankton communities exhibit a stronger response to environmental changes than bacterioplankton in three subtropical reservoirs. Environmental Science & Technology, 49(18): 10850-10858.
DOI:10.1021/acs.est.5b02637 |
Lv X M, Zhang J, Liang P, et al. 2020. Phytoplankton in an urban river replenished by reclaimed water: features, influential factors and simulation. Ecological Indicators, 112: 106090.
DOI:10.1016/j.ecolind.2020.106090 |
Margalef R. 1958. Information theory in ecology. General Systems, 3: 36-71.
|
Mcnaughton S J. 1967. Relationships among functional properties of Californian grassland. Nature, 216(5111): 168-169.
DOI:10.1038/216168b0 |
Mitrovic S M, Hardwick L, Dorani F. 2011. Use of flow management to mitigate cyanobacterial blooms in the lower Darling River, Australia. Journal of Plankton Research, 33(2): 229-241.
DOI:10.1093/plankt/fbq094 |
Mo Y Y, Peng F, Gao X F, et al. 2021. Low shifts in salinity determined assembly processes and network stability of microeukaryotic plankton communities in a subtropical urban reservoir. Microbiome, 9: 128.
DOI:10.1186/s40168-021-01079-w |
Paerl H W, Hall N S, Peierls B L, et al. 2014. Hydrologic variability and its control of phytoplankton community structure and function in two shallow, coastal, lagoonal ecosystems: the Neuse and New River Estuaries, North Carolina, USA. Estuaries and Coasts, 37(1): 31-45.
DOI:10.1007/s12237-013-9686-0 |
Paerl H W, Huisman J. 2008. Blooms like it hot. Science, 320(5872): 57-58.
DOI:10.1126/science.1155398 |
Pfeifer F. 2012. Distribution, formation and regulation of gas vesicles. Nature Reviews Microbiology, 10(10): 705-715.
DOI:10.1038/nrmicro2834 |
Pielou E C. 1967. The measurement of diversity in different types of biological collections. Journal of Theoretical Biology, 15(1): 177.
DOI:10.1016/0022-5193(67)90048-3 |
Porra R J. 2002. The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. Photosynthesis Research, 73(1): 149-156.
DOI:10.1023/A:1020470224740 |
Pyo J, Cho K H, Kim K, et al. 2021. Cyanobacteria cell prediction using interpretable deep learning model with observed, numerical, and sensing data assemblage. Water Research, 203: 117483.
DOI:10.1016/j.watres.2021.117483 |
Ramond P, Siano R, Schmitt S, et al. 2021. Phytoplankton taxonomic and functional diversity patterns across a coastal tidal front. Scientific Reports, 11(1): 2682.
DOI:10.1038/s41598-021-82071-0 |
Rao K, Zhang X, Wang M, et al. 2021. The relative importance of environmental factors in predicting phytoplankton shifting and cyanobacteria abundance in regulated shallow lakes. Environmental Pollution, 286: 117555.
DOI:10.1016/j.envpol.2021.117555 |
Rice J, Westerhoff P. 2017. High levels of endocrine pollutants in US streams during low flow due to insufficient wastewater dilution. Nature Geoscience, 10(8): 587-591.
DOI:10.1038/ngeo2984 |
Shannon C E. 1948. The Mathematical Theory of Communication. Bell System Technical Journal, 27(4): 623-656.
DOI:10.1002/j.1538-7305.1948.tb00917.x |
Striebel M, Singer G, Stibor H, et al. 2012. "Trophic overyielding": phytoplankton diversity promotes zooplankton productivity. Ecology, 93(12): 2719-2727.
DOI:10.1890/12-0003.1 |
Van Meerssche E, Pinckney J L. 2019. Nutrient loading impacts on estuarine phytoplankton size and community composition: community-based indicators of eutrophication. Estuaries and Coasts, 42(2): 504-512.
DOI:10.1007/s12237-018-0470-z |
Verspagen J M H, Passarge J, Jöhnk K D, et al. 2006. Water management strategies against toxic Microcystis blooms in the Dutch delta. Ecological Applications, 16(1): 313-327.
DOI:10.1890/04-1953 |
Visser P M, Ibelings B W, Bormans M, et al. 2016. Artificial mixing to control cyanobacterial blooms: a review. Aquatic Ecology, 50(3): 423-441.
DOI:10.1007/s10452-015-9537-0 |
Visser P, Ibelings B, Van der Veer B, et al. 1996. Artificial mixing prevents nuisance blooms of the cyanobacterium Microcystis in Lake Nieuwe Meer, the Netherlands. Freshwater Biology, 36(2): 435-450.
DOI:10.1046/j.1365-2427.1996.00093.x |
Vizzo J I, Cabrerizo M J, Helbling E W, et al. 2021. Extreme and gradual rainfall effects on winter and summer estuarine phytoplankton communities from Patagonia (Argentina). Marine Environmental Research, 163: 105235.
DOI:10.1016/j.marenvres.2020.105235 |
Walsby A E. 1975. Gas vesicles. Annual Review of Plant Physiology, 26: 427-439.
DOI:10.1146/annurev.pp.26.060175.002235 |
Wirtz K, Smith S L. 2021. Author Correction: vertical migration by bulk phytoplankton sustains biodiversity and nutrient input to the surface ocean. Scientific Reports, 11(1): 9486.
DOI:10.1038/s41598-021-88994-y |
Wiśniewska K, Śliwińska-Wilczewska S, Lewandowska A, et al. 2021. The effect of abiotic factors on abundance and photosynthetic performance of airborne cyanobacteria and microalgae isolated from the Southern Baltic Sea region. Cells, 10(1): 103.
DOI:10.3390/cells10010103 |
Worden A Z, Follows M J, Giovannoni S J, et al. 2015. Rethinking the marine carbon cycle: factoring in the multifarious lifestyles of microbes. Science, 347(6223): 1257594.
DOI:10.1126/science.1257594 |
Wu G Z, Xu Z X. 2011. Prediction of algal blooming using EFDC model: case study in the Daoxiang Lake. Ecological Modelling, 222(6): 1245-1252.
DOI:10.1016/j.ecolmodel.2010.12.021 |
Wu X W, Liu P, Wang H Y, et al. 2021. Photo aging of polypropylene microplastics in estuary water and coastal seawater: important role of chlorine ion. Water Research, 202: 117396.
DOI:10.1016/j.watres.2021.117396 |
Xu D, Bai H W, Li Z X, et al. 2021a. Acclimation of phytoplankton to diverse environmental variables at the initial operation stage of a free water surface constructed wetland. Journal of Cleaner Production, 317: 128442.
DOI:10.1016/j.jclepro.2021.128442 |
Xu D, Wang H, Han D Y, et al. 2021b. Phytoplankton community structural reshaping as response to the thermal effect of cooling water discharged from power plant. Environmental Pollution, 285: 117517.
DOI:10.1016/j.envpol.2021.117517 |
Xu T, You X Y. 2017. Effects of large-scale embankments on the hydrodynamics and salinity in the Oujiang River Estuary, China. Journal of Marine Science and Technology, 22(1): 71-84.
DOI:10.1007/s00773-016-0394-x |
Xue Y Y, Chen H H, Yang J R, et al. 2018. Distinct patterns and processes of abundant and rare eukaryotic plankton communities following a reservoir cyanobacterial bloom. The ISME Journal, 12(9): 2263-2277.
DOI:10.1038/s41396-018-0159-0 |
Yan D N, Xu H, Lan J H, et al. 2020. Warming favors subtropical lake cyanobacterial biomass increasing. Science of the Total Environment, 726: 138606.
DOI:10.1016/j.scitotenv.2020.138606 |
Yang C, Wang L R, Yu Y Q, et al. 2020. Highly efficient removal of amoxicillin from water by Mg−Al layered double hydroxide/cellulose nanocomposite beads synthesized through in-situ coprecipitation method. International Journal of Biological Macromolecules, 149: 93-100.
DOI:10.1016/j.ijbiomac.2020.01.096 |
Yi Y J, Liu Q, Zhang J, et al. 2021. How do the variations of water and sediment fluxes into the estuary influence the ecosystem?. Journal of Hydrology, 600: 126523.
DOI:10.1016/j.jhydrol.2021.126523 |
Yu Y H, Yan Q Y, Feng W S. 2008. Spatiotemporal heterogeneity of plankton communities in Lake Donghu, China, as revealed by PCR-denaturing gradient gel electrophoresis and its relation to biotic and abiotic factors. FEMS Microbiology Ecology, 63(3): 328-337.
DOI:10.1111/j.1574-6941.2007.00430.x |
Zhang M, Dong J, Gao YN, et al. 2021a. Patterns of phytoplankton community structure and diversity in aquaculture ponds, Henan, China. Aquaculture, 544: 737078.
DOI:10.1016/j.aquaculture.2021.737078 |
Zhang M, Shi X L, Chen F Z, et al. 2021b. The underlying causes and effects of phytoplankton seasonal turnover on resource use efficiency in freshwater lakes. Ecology and Evolution, 11(13): 8897-8909.
DOI:10.1002/ece3.7724 |
 2022, Vol. 40
2022, Vol. 40