Institute of Oceanology, Chinese Academy of Sciences
Article Information
- SUN Yue, TAO Xin, TANG Boping, LI Xinzheng
- Two new species of Leodamas Kinberg, 1866 (Annelida, Orbiniidae) from China seas
- Journal of Oceanology and Limnology, 40(4): 1654-1661
- http://dx.doi.org/10.1007/s00343-021-1218-3
Article History
- Received Jul. 7, 2021
- accepted in principle Aug. 6, 2021
- accepted for publication Sep. 30, 2021
2 Institute of Oceanology, Chinese Academy of Sciences, Qingdao 266071, China;
3 Center for Ocean Mega-Science, Chinese Academy of Sciences, Qingdao 266071, China;
4 University of Chinese Academy of Sciences, Beijing 100049, China;
5 Laboratory for Marine Biology and Biotechnology, Pilot National Laboratory for Marine Science and Technology(Qingdao), Qingdao 266237, China
Orbiniidae are burrowing deposit feeder annelids, inhabiting from intertidal to the deep sea, but apparently being more common in muddy and sandy shallow intertidal zones (Meca et al., 2021). Leodamas, previously regarded as a subgenus of Scoloplos (Hartman, 1957; Pettibone, 1957; Day, 1973, 1977; Fauchald, 1977; Blake, 1996; Eibye-Jacobsen, 2002) was raised to genus based on the form and arrangement of the thoracic neuropodial uncini by Blake (2000), this proposal has been followed by Blake(2000, 2017, 2020), López et al. (2003), Bleidorn et al. (2009), Dean and Blake (2015), and Zhadan et al. (2015). The species of Leodamas (either as genus or as subgenus of Scoloplos) were divided into two groups without taxonomic range, according to the start of branchiae and the number of vertical rows of uncini (Blake, 2017). With the recent description of two new species (Sun et al., 2018; Blake, 2020), the genus contains 31 species worldwide (Read and Fauchald, 2021).
Leodamas can be diagnosed by having thoracic neuropodial uncini large, conspicuous, arranged in 1–7 (usually 1–4) rows per chaetiger, few relatively inconspicuous capillaries; and posterior thoracic chaetigers with 0–2 postchaetal lobes and 0–2 subpodial lobes, with never more than four lobes of both types combined (Fauchald, 1977; Blake, 2017, 2020).
Sorting the polychaete material deposited in the Marine Biological Museum, Chinese Academy of Sciences in Qingdao, China (MBMCAS), as well as new samples collected from China seas, we identified two morphotypes belonging to Leodamas, which we are here describing as new species. Furthermore, we include a key to Leodamas species from China seas.
2 MATERIAL AND METHODThe studied material were collected from coastal areas of China seas. Specimens were preserved in 75% ethanol solution and deposited in the MBMCAS, except some specimens that were deposited at the Yancheng Teachers University (YCTU). Detailed morphological structures were examined under Zeiss Stemi 2000-C stereomicroscope. Parapodia and chaetae were dissected with iris scissors and mounted on slides. Photographs were taken with AxioCam MRc 5 digital camera attached to stereomicroscope and compound microscope. Line drawings were made in Adobe Photoshop CS6 using a graphic tablet. For scanning electron microscope (SEM), the selected parapodia were detached from the specimens, rinsed in absolute ethanol, critical-point dehydrated, coated in gold, observed, and photographed.
The following abbreviations are used through the text (spec: specimen; Sta.: station; ECS: East China Sea; SCS: South China Sea; BG: Beibu Gulf) and in figures (ac: acicula; br: branchia; neL: neuropodial lobe; noL: notopodial lobe; nuO: nuchal organ; per: peristomium; pigB: pigment band; pr: prostomium; subPFla: subpodial flange; plM: pocket-like membrane).
3 RESULT 3.1 TaxonomyFamily Orbiniidae Hartman, 1942
Subfamily Orbiniinae Hartman, 1942
Genus Leodamas Kinberg, 1866
Type-species: Leodamas verax Kinberg, 1866
3.2 Leodamas robustus sp. n.urn: lsid: zoobank.org: act: 129C4300-EA02-4EE4-80D9-318FE843F18D
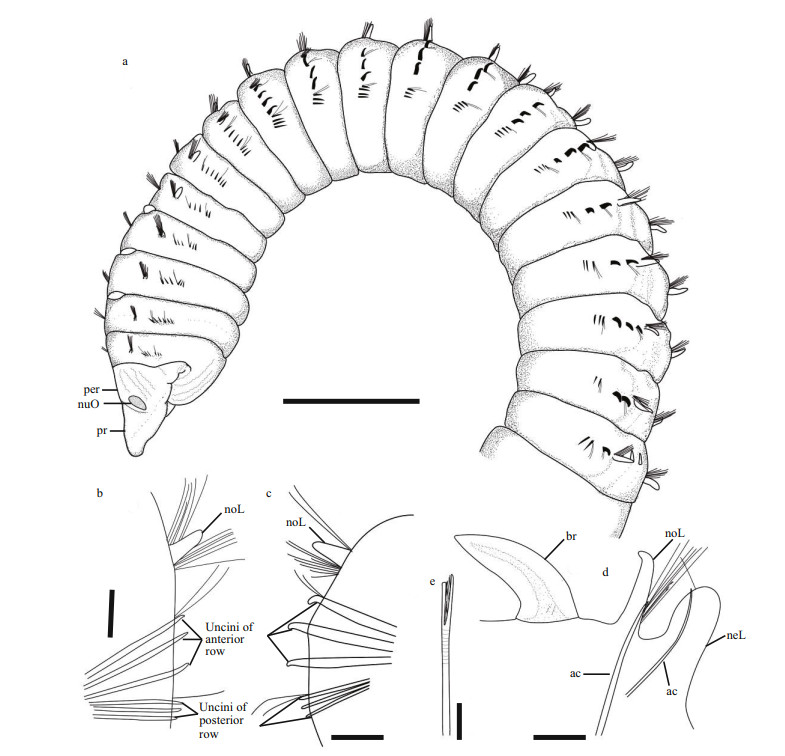
|
| Fig.1 Leodamas robustus sp. n., the holotype a. anterior region, left lateral view. Parapodia: b. 5th, posterior view; c. 15th, posterior view; d. abdominal, posterior view; e. furcate chaeta. Scale bars: a: 1 mm; b: 100 μm; c–d: 200 μm; e: 25 μm. |
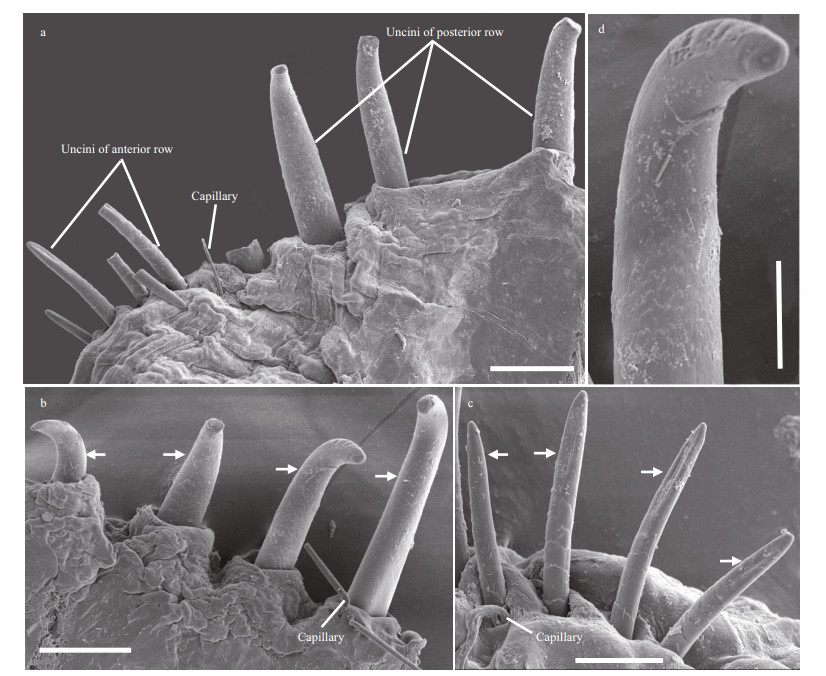
|
| Fig.2 Leodamas robustus sp. n. MBM202033; the SEM micrographs a. posterior thoracic neurochaetae; b, d. uncini of first row, arrows denote uncini; c. second row of uncini. Scale bars: a–b: 50 μm; c: 25 μm; d: 20 μm. |
Diagnosis: Leodamas with prostomium conical, blunt. Branchiae from chaetiger 19. Thorax with 19 chaetigers, thoracic neuropodial postchaetal lobe greatly reduced. Thoracic neuropodia with two rows of uncini; anterior row with 3–5 uncini, increasing in size along thorax, uncini on posterior chaetigers thicker, heavier, more curved than those on precedent chaetigers; posterior row with 2–5 thin, delicate uncini, of similar size along all chaetigers, with 1–3 thin capillaries dorsally to two rows of uncini.
Material examined: Holotype: MBM304676: ECS, Sta. DH 9–2, 26°30′N, 121°00′E, 52-m depth, 24 Dec. 2015, fine silt. Paratype: MBM202032: 1 spec, BG, Sta. 6242, 18°30ʹN, 108°00ʹE, 78-m depth, mud, 21 Oct. 1960. MBM202033: 1 spec, BG, Sta. 6254, 18°30ʹN, 107°30ʹE, 72-m depth, silty sandy mud, 22 Aug. 1960. Additional material: MBM023214: 1 spec, SCS, Sta. 6105, 21°00ʹN, 112°00′E, 49-m depth, silty sandy mud, 2 Jul. 1960. MBM202035: 1 spec, BG, Sta. 6235, 20°45ʹN, 108°00ʹE, 42-m depth, sandy mud, 11 Nov. 1960. MBM202036: 1 spec, BG, Sta. 6253, 19°00ʹN, 107°30ʹE, 67-m depth, sandy mud, 22 Apr. 1960. MBM023280: 1 spec, SCS, Sta. 6089, 21°15ʹN, 112°30ʹE, 41-m depth, muddy sand, 7 Apr. 1960. MBM023247: 1 spec, SCS, Sta. 6205, 17°45ʹN, 109°00ʹE, 74-m depth, muddy sand, 11 Apr. 1959. MBM023042: 1 spec, SCS, Sta. 6045, 21°45ʹN, 114°30ʹE, 61-m depth, soft mud, 10 Jan. 1960. YCTU000001: 1 spec, ECS, Sta. DH9–1, 26°30′N, 121°00′E, 52-m depth, fine silt, 24 Dec. 2015. YCTU000002: 1 spec, ECS, Sta. DH11–5, 25°30ʹN, 120°45ʹE, 87-m depth, fine silt, 23 Dec. 2015. YCTU000003: 3 specs, ECS, Sta. DH11–3, 25°45ʹN, 120°30ʹE, 64-m depth, fine silt, 13 Dec. 2015.
Description: All specimens incomplete, posterior end missing; holotype 1.5 mm of maximum width. Thoracic region cylindrical, with 19 chaetigers. Prostomium conical, slightly longer than wide, blunt, lacking eyespots; peristomium with a single, short, achaetous ring; nuchal organs transverse slits on anterior peristomial border (Fig. 1a). Branchiae from chaetiger 19, simple, ligulate, continuing along body through posterior chaetigers (Fig. 1d).
Parapodia biramous. Thoracic notopodia with cirriform postchaetal lobe, short on chaetiger 1, then increasing to reach full size by chaetigers 5–6; abdominal notopodial postchaetal lobe similar, but much elongated, twice longer than those of thoracic chaetigers (Fig. 1d). Interramal cirrus absent. Thoracic neuropodial postchaetal lobe greatly reduced (Fig. 1b–c). Abdominal neuropodia digitate (Fig. 1d).
Thoracic notopodia with fascicles of crenulated capillaries (Fig. 1a). Abdominal notopodia with fascicles of capillaries (Fig. 1d), 1–2 furcate chaetae (Fig. 1e) and 1–2 projecting aciculae (Fig. 1d). Thoracic neurochaetae with two short rows of uncini and 1–3 companion capillaries (Fig. 2a–c); anterior row increasing in size along thorax, with 3–5 uncini, uncini on posterior chaetigers thicker, heavier, and more curved than those on anterior chaetigers (Fig. 1a–c), with blunt tipand 2–4 prominent transverse ribs on convex side (Fig. 2a, b, d); posterior rows all similar size, with 2–5 thin, delicate uncini (Fig. 1a–c), with straight-shaft and weakly developed subapical groove, remaining shaft with weakly developed transverse ribs (Fig. 2a, c). Abdominal neuropodia with 4–8 thin crenulated capillaries and single imbedded acicula, thinner than dorsal ones (Fig. 1d). Flail chaetae not seen.
Variability: 15–19 thoracic chaetigers. Branchiae start from last two thoracic to first abdominal chaetigers.
Etymology: The specific name, robustus, meaning thick and strong in Latin, refers to the cylindrical and robust thoracic region.
Distribution: East China Sea, northern South China Sea, and Beibu Gulf, in sediments at 41–87-m depth.
Remark: Leodamas robustus sp. n. belongs to the species Group B (Blake, 2017) as it has branchiae first present from last 1–2 thoracic or first abdominal chaetigers and thoracic neuropodial uncini arranged in two rows. However, it clearly differs from all other species in this group in having uncini arranged in two short rows, with 1–3 thin companion capillaries; anterior row of uncini increasing in size, with 3–5 uncini and uncini on posterior chaetigers thicker and more curved than those on precedent thoracic chaetigers; posterior row of similar size along thoracic chaetigers, with 2–5 thin, delicate uncini.
Leodamas robustus sp. n. resembles L. agrestis (Nonato and Luna, 1970) in having two different uncini rows, but differs in having these two rows along all thoracic chaetigers (instead of one on anterior thoracic chaetigers, and an additional one anterior row of 2–3 large heavy spines on chaetigers 7–15 in L. agrestis).
3.3 Leodamas weizhouensis sp. n.urn: lsid: zoobank.org: act: 6AAA83BC-D729-4E82-88F2-B0F05A791330

|
| Fig.3 Leodamas weizhouensis sp. n., the holotype a. anterior end, dorsal view, arrows indicate pocket-like membrane; b. anterior chaetigers of abdominal region, arrows point to pocket-like membranes; c. thoracic neuropodial uncini; d. anterior thoracic neuropodium showing arrangement of uncini and capillaries; e. neuropodium of chaetiger 13; f. furcate chaetae, arrows indicate furcate chaetae. Scale bars: a: 1 mm; b: 200 μm; c: 10 μm; d–f, 50 μm. |
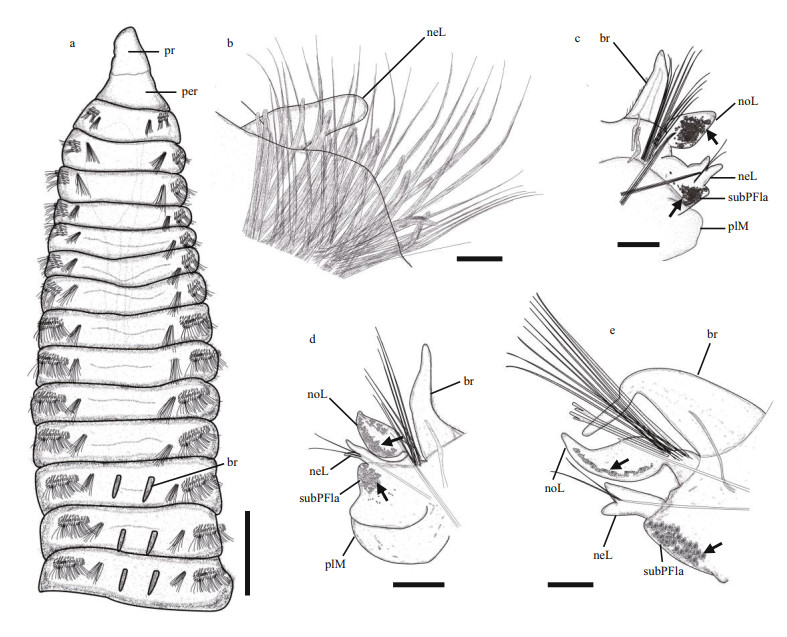
|
| Fig.4 Leodamas weizhouensis sp. n., the holotype a. anterior thoracic chaetigers, dorsal view; b. neuropodium of chaetiger 19. Parapodia: c. chaetiger 20, arrows denote pigment spots; d. middle abdominal region; e. posterior abdominal region. Scale bars: a: 0.5 mm; b: 50 μm; c–d: 200 μm; e: 100 μm. |
Diagnosis: Leodamas with prostomium conical, pointed. Branchiae from chaetigers 11–13. Thorax with 20 chaetigers, thoracic neuropodial postchaetal lobe greatly reduced, with delicate pocket-like membrane below, posterior to neuropodia. Thoracic neuropodia with five rows of uncini, last uncini row shorter, only half as long as anterior rows, decreasing to 1–2 rows in posterior thoracic chaetigers; uncini distally curved, with 3–4 transverse rows of ribs along shaft, long crenulated capillaries in single posterior row.
Material examined: Holotype: MBM196722, 16 Apr. 1978, Beigang Village, Weizhou Island, Guangxi Zhuang Autonomous Region, 21°04′N, 109°08′E, intertidal. Paratype: MBM193712: 5 specs, 1 Apr. 1978, ibid. Additional material: MBM193720, 3 specs, 13 Apr. 1978; MBM193721: 7 specs, 28 Apr. 1978; MBM193722: 1 spec, 16 Apr. 1978, ibid.
Description: All specimens incomplete, posterior end missing; holotype 1.3 mm of maximum width (Fig. 3a). Color in alcohol: light brown, with dark brown pigment spots on branchiae, abdominal parapodia and subpodial flange (Fig. 4c–e), with transverse brown pigment band in middle of each chaetigers (Fig. 3a–b).
Thoracic region with 20 chaetigers, first 3–4 swollen, thereafter depressed. Prostomium conical, blunt, as long as wide, lacking eyespots; peristomium single short achaetous ring, nuchal organs not observed. Branchiae from chaetiger 12, with ciliated margins, continuing along body throughout posterior chaetigers, smaller in thoracic chaetigers (Fig. 4a), and then becoming ligulate, with terminal tips nipple-like, longer than notopodial lobes (Fig. 4c–e).
Parapodia biramous. Thoracic notopodia with cirriform postchaetal lobe (Fig. 3a). Abdominal notopodia with postchaetal lobe broadly triangular, tapering to narrow tip (Fig. 4c–e). Interramal cirrus absent. Thoracic neuropodia with a single papillate postchaetal lobe through anterior chaetigers, then two on last 3–5 thoracic chaetigers (Fig. 4b). Abdominal neuropodia elongated, terminally bilobed, inner lobe larger than outer, with fleshy subpodial flange (Fig. 4c–e). Abdominal chaetigers with a delicate pocket-like membrane below, posterior to neuropodia (Fig. 4d).
Thoracic notopodia with fascicles of crenulated capillaries (Fig. 4a). Abdominal notopodia with fascicles of capillaries, 4–5 furcate chaetae, three embedded aciculae, decreasing to one through posterior chaetigers (Fig. 4c–e). Thoracic neuropodia with uncini arranged in five rows, decreasing to 1–2 in posterior chaetigers; posterior row with long crenulated capillaries (Figs. 3d, 4b). Uncini distally curved, with 3–4 transverse rows of ribs along shaft (Fig. 3c, e); abdominal neurochaetae including 4–8 thin crenulated capillaries and 1–3 embedded aciculae (Fig. 4c–e).
Variability: Nineteen to twenty thoracic chaetigers, with branchiae first present on chaetiger 11–13 and pocket-like membrane first present from last thoracic or first abdominal chaetigers.
Etymology: The specific name is derived from the type locality, Weizhou Island, with the Latin suffix, ensis, indicating living in it.
Distribution: Known only from Weizhou Island (South China Sea), sandy beach.
Remark: Leodamas weizhouensis sp. n. is included in Leodamas due to the presence of large, conspicuous uncini and capillaries in thoracic neuropodia. It belongs to Group A (Blake, 2017) as it has 19–20 thoracic chaetigers, branchiae from chaetigers 11–13, and thoracic neuropodial uncini arranged in five rows. However, it can be distinguished by having a delicate pocket-like membrane below and posterior to neuropodia. The new species closely resemble Scoloplos marsupialis (Southern, 1921) in having a pocket-like membrane, even they belong to different genus, but can be distinguished having four rows of conspicuous uncini and few capillaries behind them, instead of 2–3 rows and numerous capillaries in S. marsupialis.
3.4 Key to species of Leodamas from China seas1.a.- Branchiae from anterior thoracic chaetiger (4–7) and thoracic neuropodial uncini typically in three or more rows …………………………………2
1.b.- Branchiae from posterior thoracic or anterior abdominal setiger, thoracic neuropodial uncini in two rows ……………………………… L. robustus sp. n.
2.a.- Thoracic neuropodia postchaetal lobe with broad tori and a digitate to triangular papilla at midlength …………… L. sinensis Sun, Sui & Li, 2018
2.b.- Thoracic neuropodia postchaetal lobe reduced or undeveloped, median papilla small or reduced ……3
3.a.- Abdominal chaetigers with delicate pocket-like membrane below and posterior to neuropodia ……………………………………L. weizhouensis sp. n.
3.b.- Delicate pocket-shaped membrane absent ……………………………………L. bathyalis Blake, 2020
4 CONCLUSIONFour species of Leodamas are currently known to occur in China seas, including the two species described in this study and Leodamas bathyalis (Blake, 2020) and Leodamas sinensis (Sun et al., 2018). Leodamas robustus sp. n. belongs to group B and can be easily identified by the morphology and arrangement of uncini. The other three species belong to group A, including L. weizhouensis sp. n. that is characterized by having a delicate pocket-like membrane below and posterior to neuropodia.
5 DATA AVAILABILITY STATEMENTThe authors declare that the data supporting the findings of this study are available within the article. The data will be available on request from the corresponding author.
6 ACKNOWLEDGMENTWe thank the MBMCAS managers for their help in sorting the studied material.
Blake J A. 1996. Family orbiniidae Hartman, 1942. In: Blake J A, Hilbig B, Scott P H eds. Taxonomic Atlas of the Benthic Fauna of the Santa Maria Basin and Western Santa Barbara Channel. Santa Barbara Museum of Natural History, Santa Barbara. p. 1-26.
|
Blake J A. 2000. A new genus of polychaete worm (Family Orbiniidae) from methane seeps in the Gulf of Mexico, with a review of the systematics and phylogenetic interrelationships of the genera of Orbiniidae. Cahiers de Biologie Marine, 41(4): 435-449.
|
Blake J A. 2017. Polychaeta Orbiniidae from Antarctica, the Southern Ocean, the abyssal Pacific Ocean, and off South America. Zootaxa, 4218(1): 1-145.
DOI:10.11646/zootaxa.4218.1.1 |
Blake J A. 2020. New species and records of deep-water Orbiniidae (Annelida, Polychaeta) from the Eastern Pacific continental slope, abyssal Pacific Ocean, and the South China Sea. Zootaxa, 4730(1): 1-61.
DOI:10.11646/zootaxa.4730.1.1 |
Bleidorn C, Hill N, Erséus C, Tiedemann R. 2009. On the role of character loss in orbiniid phylogeny (Annelida): molecules vs. morphology. Molecular Phylogenetics and Evolution, 52(1): 57-69.
DOI:10.1016/j.ympev.2009.03.022 |
Day J H. 1973. New Polychaeta from Beaufort: with a Key to all Species Recorded from North Carolina. NOAA Technical Report NMFS CIRC, Seattle.
|
Day J H. 1977. A review of the Australian and New Zealand Orbiniidae (Annelida: Polychaeta). In: Reish D J, Fauchald K eds. Essays on Polychaetous Annelids in Memory of Dr. Olga Hartman. Allan Hancock Foundation, University of Southern California, Los Angeles. p. 217-246.
|
Dean H K, Blake J A. 2015. The Orbiniidae (Annelida: Polychaeta) of pacific Costa Rica. Zootaxa, 3956(2): 183-198.
DOI:10.11646/zootaxa.3956.2.2 |
Eibye-Jacobsen D. 2002. The Orbiniidae (Annelida: Polychaeta) of the BIOSHELF Project, Andaman Sea, Thailand. Phuket Marine Biological Center, Special Publication, 24: 77-99.
|
Fauchald K. 1977. The polychaete worms. Definitions and keys to the orders, families and genera. Natural History Museum of Los Angeles County. Science Series, 28: 1-190.
|
Hartman O. 1942. A review of the types of polychaetous annelids at the Peabody Museum of Natural History, Yale University. Bulletin of the Bingham Oceanographic Collection, 8: 11-98.
|
Hartman O. 1957. Orbiniidae, apistobranchidae, paraonidae and longosomidae. Allan Hancock Pacific Expeditions, 15(3): 211-393.
|
Kinberg J G H. 1866. Annulata nova. Öfversigt af Kongl. Vetenskaps-Akademiens Förhandlingar, 23: 97-103.
|
López E, Cladera P, San Martín G. 2003. Two new species of the genus Leodamas (Orbiniidae: Scolecida: Polychaeta) from the Pacific coast of Panama. Journal of the Marine Biological Association of the United Kingdom, 83(2): 367-374.
DOI:10.1017/S0025315403007215h |
Meca M A, Zhadan A, Struck T H. 2021. The early branching group of Orbiniida sensu Struck et al., 2015: parergodrilidae and orbiniidae. Diversity, 13(1): 29.
DOI:10.3390/d13010029 |
Nonato E F, Luna J A C. 1970. Anelídeos poliquetas do Nordeste do Brasil: Ⅰ-Poliquetas bentônicos da costa de Alagoas e Sergipe. Boletim do Instituto Oceanografico, 19: 57-130.
DOI:10.1590/S0373-55241970000100004 |
Pettibone M H. 1957. North American genera of the family Orbiniidae. Journal of the Washington Academy of Science, 47: 159-167.
|
Read G, Fauchald K. 2021. World Polychaeta database. Leodamas Kinberg, 1866. http://www.marinespecies.org/aphia.php?p=taxdetails&id=325863. Accessed on 2021-08-26.
|
Southern R. 1921. Polychaeta of the Chilka Lake and also of fresh and brackish waters in other parts of India. Memoirs of the Indian Museum, 5(8): 563-659.
|
Sun Y, Sui J X, Li X Z. 2018. A new species of Leodamas Kinberg, 1866 (Polychaeta: Orbiniidae) from the Yellow Sea and the East China Sea. Acta Oceanologica Sinica, 37(10): 130-135.
DOI:10.1007/s13131-018-1313-2 |
Zhadan A, Stupnikova A, Neretina T. 2015. Orbiniidae (Annelida: Errantia) from Lizard Island, Great Barrier Reef, Australia with notes on orbiniid phylogeny. Zootaxa, 4019(1): 773-801.
DOI:10.11646/zootaxa.4019.1.27 |
 2022, Vol. 40
2022, Vol. 40


