Institute of Oceanology, Chinese Academy of Sciences
Article Information
- WANG Xuemei, SHAN Tifeng, PANG Shaojun
- Potential of Ulva prolifera in phytoremediation of seawater polluted by cesium and cobalt: an experimental study on the biosorption and kinetics
- Journal of Oceanology and Limnology, 40(4): 1592-1599
- http://dx.doi.org/10.1007/s00343-001-1205-7
Article History
- Received Jun. 24, 2021
- accepted in principle Aug. 11, 2021
- accepted for publication Oct. 18, 2021
2 Laboratory for Marine Biology and Biotechnology, Pilot National Laboratory for Marine Science and Technology(Qingdao), Qingdao 266237, China
With rapid worldwide development in the construction of coastal nuclear power plants (NPPs), the safety of NPPs posed a major risk to the public, particularly after the Fukushima 1 accident in 2011. The radionuclides released after the accident could cause serious pollution in coastal waters (Fukuda et al., 2014; Zhuang et al., 2018). After the Fukushima 1 accident, the radiation level near the NPP was 15 000– 30 000 times higher over the normal level (Xiao et al., 2011). One of the major radionuclides released was 137Cs, which has a relatively long half-life of 30 years of radioactivity (Wang et al., 2018a). 60Co was also present in wastewater from NPPs and has a half-life of 5 years (Lingamdinne et al., 2016). Although Co is a source of vitamin B12, an essential trace element for life, it would become toxic at a high concentration, and on the other hand, cause low blood pressure, paralysis, and bone defects, as well as damage in the lungs, heart, and liver in humans (Ahmadpour et al., 2009; Mosoarca et al., 2018).
The removal of radionuclides from polluted seawater should be in high priority because of their negative effects on the marine environment. Several methods—e.g., ion exchange, adsorption, chemical precipitation, and membrane technology—have been proposed for removing radionuclides from seawater (Hu et al., 2019). Biosorbents have been testified to have higher removal efficiency, and they are more abundant, less expensive, and importantly, more environmentally friendly, than both physical and chemical adsorbents (Bailey et al., 1999; Alluri et al., 2007). Therefore, the potential value of living macroalgae as biosorbents has been assessed for the removal of lead, copper, chromium, strontium, and cobalt (Doshi et al., 2008; Southichak et al., 2008; Mosoarca et al., 2018; Wang et al., 2018b). The consumption of algal biomass would be huge once nuclide pollution occurs. The ability to obtain a large biomass in a short period is a major technical bottleneck for using macroalgae in marine nuclide pollution remediation.
Green tides are vast blooms of floating green macroalgae, mainly involving Ulva(or Enteromorpha), which are not distinct genera in taxonomy (Hayden et al., 2003; Nelson et al., 2008). Algal blooms in coastal waters initially are resulted from eutrophication, and they are often harmful to aquaculture and tourism (Sun et al., 2008). The Qingdao coast of the Yellow Sea in China first experienced a small-scale green tide in the summer of 2007. In the following years, massive green tides occurred each summer in the Yellow Sea (Liu et al., 2015; Zhao et al., 2015). Before the 2008 Olympic Regatta in Qingdao, the largest green tide in the world at that time occurred along the Qingdao coast, measuring 3 849 km2 and having an approximate harvested biomass of one million tons (fresh weight) (Pang et al., 2010; Keesing et al., 2011). Through morphological and molecular analyses, the dominant species was determined filamentous Ulva prolifera (Müller) J. Agardh (Sun et al., 2008). The main treatment method for green tides at present is salvage by boat, yet disposing of massive amounts of Ulva biomass remains a major challenge. We may find a novel application for making use of this immense biomass "endowed" by nature if the green algae can be used as a sorbent for nuclide pollution.
Different macroalgal species have different adsorption capacities for nuclides. In general, the green algae have a higher adsorption capacity than red algae, but lower than brown algae, due to the different composition of the cell wall (Wang et al., 2018b). Considering the huge biomass requirement of bioremediation and the immense biomass of algal bloom, we explored the removal potential for nuclides of Sargassum horneri from golden tide (Wang et al., 2021). The content of the corresponding heavy metal in U. prolifera could reach 44.67 and 503.63 mg/kg, and the bio-concentration factors (BCFs) could be 625.89 and 9 119.79 after being cultured for 12 d in the seawater containing 80-μg/L Cd2+ and Pb2+, respectively (Liu et al., 2014). However, there was no literature about the scavenging ability of U. prolifera for important nuclides such as 137Cs and 60Co commonly found in polluted water after nuclear accidents. To potentially exploit the waste products of green tides for remediation purposes, we investigated the scavenging ability of U. prolifera for Cs and Co from seawater under simulation conditions and analyzed the biosorption characteristics, kinetics, and models. Based on our results, we discuss the potential of U. prolifera in the remediation of seawater polluted by Cs and Co.
2 MATERIAL AND METHOD 2.1 Biosorption experimentUlva prolifera was collected from the coastal region of Qingdao, China, during a green tide in July 2017. The algae were acclimatized for 2 d in indoor seawater circulation tanks with constantly aerated sand-filtered seawater. U. prolifera was then acclimatized in an intelligent light incubator (Jiangnan Co. Ltd., Ningbo, China) in 3-L beakers (200-g (fresh weight) algea per beaker) at 15 ℃ and a photoperiod of 12 h꞉12 h (light꞉dark) using fluorescent white light at 100 μmol photons/(m2·s). The culture medium was sterilized seawater enriched with 70-mg/L NaNO3 and 10-mg/L NaH2PO4. Chloride salt of Cs or Co was used in the present investigation. U. prolifera was then cultured under the same conditions in the culture medium at four different concentrations of Cs or Co (10, 20, 50, and 100 mg/L). Culture media without Co or Cs were set as controls. The background concentrations of Cs and Co were detected to be 0.023 7 μg/L and 0.004 5 μg/L using an Agilent 7500ce inductively coupled plasma mass spectrometry (ICP-MS) with serial standard solutions under default parameters. All culture media were constantly aerated and renewed daily. The Cs or Co content of algae was measured at 10 different time points. All sampling and cultures were performed in triplicate.
In the tank simulation experiment, 5 kg (fresh weight) of U. prolifera was placed into each 90-L tank. It was cultured in the above-mentioned sandfiltered natural seawater under ambient conditions. Chloride salt of Cs or Co was added to each tank, and the final concentration of Cs or Co was 100 mg/L (calculated value). The Cs content of the algae and Co concentration of the culture medium were measured at different time points.
2.2 Nuclides quantificationCs or Co content in algae and seawater was measured using an inductively coupled plasma optical emission spectrometer (ICP-OES). Algal samples were dried and digested according to methods described in Wang et al. (2018b). Following digestion, the samples were diluted with deionized water, and Cs or Co concentrations were detected with the Agilent 5110 ICP-OES in the presence of serial standard solutions under default parameters. The absorption wavelengths (λ) used to determine the analyzed nuclides were 697.327 nm for Cs and 228.615 nm for Co.
For the tank experiment, the Cs concentration of the culture medium was calculated via the content in algae because certain seawater components may interfere with Cs detection. The formula is as follows:
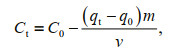 (1)
(1)where V (L) is the volume of solution in the tank, m (g) is the dry weight (DW) of the algal biomass, and q0 (mg/kg DW) and C0 (mg/L) are the background nuclide content in algae and the initial nuclide concentration in the solution, respectively. The parameters qt (mg/kg DW) and Ct (mg/L) represent the nuclide content of the algae and nuclide concentration of the solution at a given time t, respectively. The dry weight/fresh weight ratio for U. prolifera used in the present study was determined to be 20.76%±0.45%.
The bioconcentration factor (BCF) is the ratio of certain compounds or element contents in the biomass (q, mg/kg DW) to the dissolved corresponding chemical concentration in solution (c, mg/L) (meter (ICP-OES). Algal samples were dried and digested according to methods described in Foster, 1976). BCF is calculated by q/c, and it denotes the enrichment level of the given chemical in living organisms.
2.3 Quality control and statistical analysisAll chemicals used in the present research were analytical reagents for quality control. The biosorption test was conducted in an incubator with accurately controlled environmental conditions for 10 d. Each concentration group was set up and all measurements were conducted in triplicate. Results are represented as the mean±standard deviation (SD). The tank experiment data were analyzed using a one-way analysis of variance (ANOVA) with SPSS Ver. 22. Homogeneity of variance was checked, and data were transformed if necessary before analysis. The Tukey's multiple comparison method was employed when significant differences (P < 0.05) were observed.
2.4 Kinetics modelPseudo-first-order (Lagergren, 1898) and pseudosecond-order equations (Ho and McKay, 1999) were employed to evaluate the dominating mechanism of the Cs or Co biosorption process by U. prolifera biomass. Linear regression analysis was used to calculate the correlation coefficients (R2). The equations of linear forms are as follows:
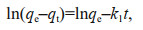 (2)
(2)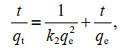 (3)
(3)where the nuclide contents in algae at time t and under equilibrium were designated qt and qe (mg/kg DW), respectively. The parameter k1 (/h) is a pseudo-firstorder rate constant, and k2 (g/(mg·h)) is a pseudosecond-order rate constant.
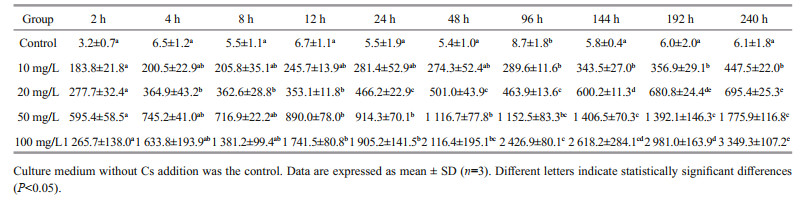
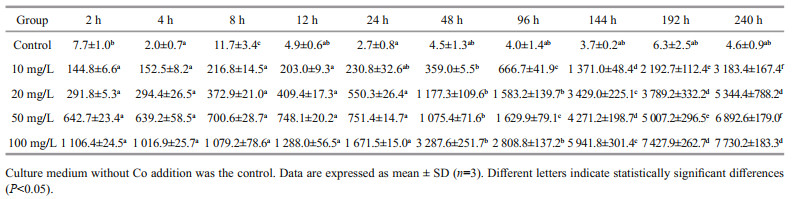
3 RESULT 3.1 Biosorption processThe Cs contents of the U. prolifera biomass in different Cs concentration groups were determined after different culture times ranging from 2 h to 10 d (Table 1). The Cs contents of U. prolifera in the control group were almost the same at different times, except for 96 h, a little higher than other time points. During the experiment, the biosorption capacity increased significantly with increased Cs concentration in the medium. The Cs contents of U. prolifera increased rapidly in the first day, especially in the first 2 h, and then much more slowly over the following 9 d. The Co contents of the U. prolifera biomass in the different Co concentration groups were also determined at different times, from 2 h to 10 d (Table 2). Co contents of U. prolifera in the control group were statistically significant differences (P < 0.05) at some time points. According to our results, the biosorption process of Cs by U. prolifera was similar to that of Co.
|
|
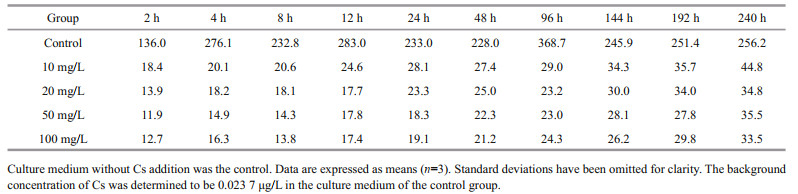
For Cs, the BCF of U. prolifera was 136.0–368.7 in the control group and 11.9–44.8 in the experimental groups (Table 3). On the tenth day, the mean BCFs of U. prolifera at Cs concentrations of 10, 20, 50, and 100 mg/L were 44.8, 34.8, 35.5, and 33.5, respectively. For Co, the BCF was 447.9–2631.4 in the control group and 10.2–318.3 in the experimental groups (Table 4). On the tenth day, the mean BCFs of U. prolifera at different Co concentrations (10, 20, 50, and 100 mg/L) were 318.3, 267.2, 137.9, and 77.3, respectively. We conclude that the increase in the initial Cs or Co concentration reduces the BCFs of U. prolifera for Cs or Co, although the absolute biosorption quantity increases substantially.
|

|
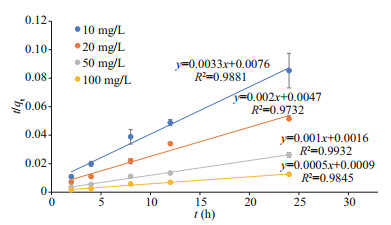
The pseudo-first-order and pseudo-second-order equations were applied to the Cs and Co biosorption processes during the first day before the medium was renewed. The data did not fit the pseudo-first-order model but fit the pseudo-second-order equation well. The physical meaning of pseudo-second-order kinetics is the reaction rate that is proportional to the quadratic power of reactant concentration. It is suitable for reactions with saturation sites, for example, the adsorption of heavy metals. The plots of t/qt versus t for Cs biosorption are shown in Fig. 1. The correlation coefficients (R2) are higher than 0.95 for all the Cs concentrations. The plots of t/qt versus t for Co biosorption are shown in Fig. 2. The correlation coefficients (R2) are higher than 0.95 for all the Co concentrations. Our results revealed that the pseudosecond-order equation provides a high correlation for the biosorption process of Cs and Co by U. prolifera. This indicates that in the total biosorption process, adsorption is the rate-limiting step.
|
| Fig.1 Plots of the pseudo-second-order kinetic model for Cs biosorption using U. prolifera Vertical bars indicate standard deviations. |

|
| Fig.2 Plots of the pseudo-second-order kinetic model for Co biosorption using U. prolifera Vertical bars indicate standard deviations. |
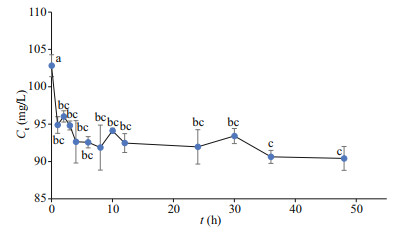
The Cs concentrations in the culture medium at different times were calculated using the Cs contents in the algae in the tank simulation experiment (Fig. 3). The Ct of Cs decreased sharply in the first 2 h and gradually thereafter. The Cs concentration in the culture medium decreased by 25.2% throughout the experimental period. Additionally, the biosorption process was fast in 12 h, although a slight decline was observed after that. The Co concentrations in the culture medium were determined at different times in 2 days (Fig. 4). As with Cs, the Ct of Co decreased sharply in the first 2 h and gradually over the remaining time. The Co concentration in the culture medium decreased by 12.1%, less than the Cs reduction percentage, during the experimental period. Similar to Cs, the biosorption process for Co was also rapid in 12 h, followed by a slight decline.

|
| Fig.3 Cs concentrations of culture medium in tank simulation experiment after disposing with U. prolifera for different time in two days The initial Cs concentration in culture medium was 101.3 mg/L. Vertical bars indicate standard deviations. Different letters indicate statistically significant differences (P<0.05). |
|
| Fig.4 Co concentrations of culture medium in tank simulation experiment after disposing with U. prolifera for different time in two days The initial Co concentration in culture medium was 102.8 mg/L. Vertical bars indicate standard deviations. Different letters indicate statistically significant differences (P<0.05). |
The macroalga U. prolifera, the dominant species in the recurrent green tide in the Yellow Sea of China, was found to be effective for the remediation of seawater containing the nuclides Cs and Co. Previous research has shown that the biosorption of metal ions by living biomass involves a fast passive adsorption process, followed by a slower active bioaccumulation process, which is dependent on metabolism (Mehta and Gaur, 2005). From the biosorption and tank simulation experiments, we found that the biosorption process of Cs and Co by U. prolifera was rapid on the first day, especially in the first few hours. It may be that adsorption is the dominant process in this remediation process. The fitting results of the pseudosecond-order equation, which identified a strong correlation for the biosorption process of Cs and Co during the first day, substantiated this conclusion. This characteristic of the remediation process can significantly reduce the processing time in the following practical application, achieving the goal of rapid interception once the nuclear leakage accident occurred.
To compare the nuclide adsorption capacity of U. prolifera with other algal species, the efficiency of different algal species to remove nuclides from contaminated water is listed in Table 5. The removal rate (%) for Cs and Co of U. prolifera was lower than Myriophyllum spicatum, a kind of common freshwater alga. For another algal bloom species, Sargassum horneri, the removal rate for Co was about 40% when the concentration of Co was 1 mg/L. However, we cannot draw the conclusion which alga has higher adsorption capacity for Co, as the concentration of Co was different.
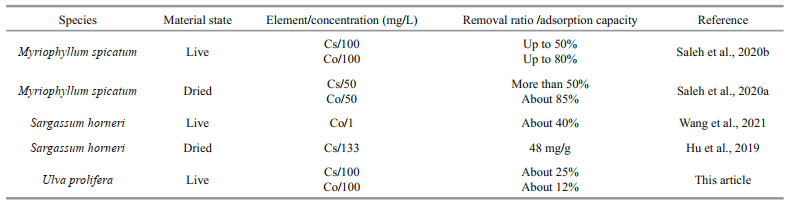
|
The biosorption capabilities of different algal species likely vary due to their specific cell wall structure and composition. The adsorption force is mainly the crosslinking between the positively charged nuclides and negatively charged chemicals in the cell wall, such as the carboxyl in the alginate (Mata et al., 2009). U. prolifera has tubular structure of monolayer cells, favorable to adsorbing pollutants in theory. However, in our previous study, brown algae exhibited higher removal capacity for Sr compared with green algae such as Ulva (Wang et al., 2018b). This is likely caused by fewer negatively charged binding groups on the cell wall of Ulva than brown algae, which contain a large amount of alginates in their cell walls.
The survival of U. prolifera would decrease without renewal of the culture medium owing to nutrient depletion or exudate build-up. This would complicate evaluations of the remediation potential of U. prolifera in an open sea area. However, renewing the culture medium would have interrupted the kinetics process during the experiment. Therefore, we only used the data from the first day in the biosorption process to investigate the kinetics models. In fact, we attempted to apply the pseudo-second-order equation to the whole Cs and Co biosorption process, but the experimental data did not fit the pseudo-first-order model (results not shown). By the end of the biosorption experiment, the Cs and Co contents in algae were relatively high and still increasing. Equilibrium states have been reached in the remediation of Sr and Cs by the algae Sargassum horneri (Wang et al., 2018b; Hu et al., 2019). Here, however, equilibrium was not reached until the tenth day in the constant external concentration conditions of Cs and Co. This is likely caused by the different components of cell wall between brown and green macroalgae. Overall, our results suggest that live macroalgae U. prolifera likely has remediation potential for the nuclides Cs and Co, and future studies should focus on the removal mechanism.
5 CONCLUSIONThe live green alga U. prolifera can potentially remove Cs and Co from contaminated seawater. Given that U. prolifera is the dominant species in green tides and that a large quantity of biomass is usually available, it has a potential to be employed in the bioremediation of seawater contaminated by Cs or Co. Its use may resolve, to some extent, the high demand for biomass in the bioremediation of seawater polluted by nuclides. After all, the large harvests of Ulva during green tide events have made disposal a challenge in recent years. This study provides some direction for the potential practical exploitation of Ulva biomass. Practical application would be doubly beneficial by providing an outlet for excess Ulva biomass during large-scale green tides and remediating seawater nuclide pollution.
6 DATA AVAILABILITY STATEMENTThe data that support the findings of this study are available from the corresponding author upon reasonable request.
Ahmadpour A, Tahmasbi M, Bastami T R, Besharati J A. 2009. Rapid removal of cobalt ion from aqueous solutions by almond green hull. Journal of Hazardous Materials, 166(2-3): 925-930.
DOI:10.1016/j.jhazmat.2008.11.103 |
Alluri H, Ronda S R, Settalluri V, Bondili J, Suryanarayana V, Venkateshwar P. 2007. Biosorption: an eco-friendly alternative for heavy metal removal. African Journal of Biotechnology, 6(25): 2924-2931.
DOI:10.5897/AJB2007.000-2461 |
Bailey S E, Olin T J, Bricka R M, Adrian D D. 1999. A review of potentially low-cost sorbents for heavy metals. Water Research, 33(11): 2469-2479.
DOI:10.1016/S0043-1354(98)00475-8 |
Doshi H, Seth C, Ray A, Kothari I L. 2008. Bioaccumulation of heavy metals by green algae. Current Microbiology, 56(3): 246-255.
DOI:10.1007/s00284-007-9070-z |
Foster P. 1976. Concentrations and concentration factors of heavy metals in brown algae. Environmental Pollution, 10(1): 45-53.
DOI:10.1016/0013-9327(76)90094-x |
Fukuda S Y, Iwamoto K, Atsumi M, Yokoyama A, Nakayama T, Ishida K I, Inouye I, Shiraiwa Y. 2014. Global searches for microalgae and aquatic plants that can eliminate radioactive cesium, iodine and strontium from the radiopolluted aquatic environment: a bioremediation strategy. Journal of Plant Research, 127(1): 79-89.
DOI:10.1007/s10265-013-0596-9 |
Hayden H S, Blomster J, Maggs C A, Silva P C, Stanhope M J, Waaland J R. 2003. Linnaeus was right all along: Ulva and Enteromorpha are not distinct genera. European Journal of Phycology, 38(3): 277-294.
DOI:10.1080/1364253031000136321 |
Ho Y S, McKay G. 1999. Pseudo-second order model for sorption processes. Process Biochemistry, 34(5): 451-465.
DOI:10.1016/S0032-9592(98)00112-5 |
Hu Y M, Guo X, Chen C, Wang J L. 2019. Algal sorbent derived from Sargassum horneri for adsorption of cesium and strontium ions: equilibrium, kinetics, and mass transfer. Applied Microbiology and Biotechnology, 103(6): 2833-2843.
DOI:10.1007/s00253-019-09619-z |
Keesing J K, Liu D Y, Fearns P, Garcia R. 2011. Inter- and intra-annual patterns of Ulva prolifera green tides in the Yellow Sea during 2007-2009, their origin and relationship to the expansion of coastal seaweed aquaculture in China. Marine Pollution Bulletin, 62(6): 1169-1182.
DOI:10.1016/j.marpolbul.2011.03.040 |
Lagergren S. 1898. Zur theorie der sogenannten adsorption gelöster stoffe[About the theory of so-called adsorption of soluble substances]. Kungliga Svenska Vetenskapsakademiens Handlingar Band, 4: 1-39.
|
Lingamdinne L P, Koduru J R, Roh H, Choi Y L, Chang Y Y, Yang J K. 2016. Adsorption removal of Co(II) from waste-water using graphene oxide. Hydrometallurgy, 165: 90-96.
DOI:10.1016/j.hydromet.2015.10.021 |
Liu Q, Yu R C, Yan T, Zhang Q C, Zhou M J. 2015. Laboratory study on the life history of bloom-forming Ulva prolifera in the Yellow Sea. Estuarine, Coastal and Shelf Science, 163: 82-88.
DOI:10.1016/j.ecss.2014.08.011 |
Liu Z Y, Wu J N, Li L, Yang M Feng, He C B, Zheng S H, Chen S J. 2014. Kinetics of cadmium, lead and aluminum bioaccumulation by Enteromorpha prolifera. Modern Food Science & Technology, 30(10): 154-158, 211.
(in Chinese with English abstract) |
Mata Y N, Blázquez M L, Ballester A, González F, Muñoz J A. 2009. Biosorption of cadmium, lead and copper with calcium alginate xerogels and immobilized Fucus vesiculosus. Journal of Hazardous Materials, 163(2-3): 555-562.
DOI:10.1016/j.jhazmat.2008.07.015 |
Mehta S K, Gaur J P. 2005. Use of algae for removing heavy metal ions from wastewater: progress and prospects. Critical Reviews in Biotechnology, 25(3): 113-152.
DOI:10.1080/07388550500248571 |
Mosoarca G, Vancea C, Popa S, Boran S. 2018. Adsorption, bioaccumulation and kinetics parameters of the phytoremediation of cobalt from wastewater using Elodea canadensis. Bulletin of Environmental Contamination and Toxicology, 100(5): 733-739.
DOI:10.1007/s00128-018-2327-3 |
Nelson T A, Karalon H, Nelson A V, Ribarich H, Hotchkiss R, van Alstyne K L, Buckingham L, Simunds D J, Fredrickson K. 2008. Ecological and physiological controls of species composition in green macroalgal blooms. Ecology, 89(5): 1287-1298.
DOI:10.1890/07-0494.1 |
Pang S J, Liu F, Shan T F, Xu N, Zhang Z H, Gao S Q, Chopin T, Sun S. 2010. Tracking the algal origin of the Ulva bloom in the Yellow Sea by a combination of molecular, morphological and physiological analyses. Marine Environmental Research, 69(4): 207-215.
DOI:10.1016/j.marenvres.2009.10.007 |
Saleh H M, Moussa H R, El-Saied F A, Dawoud M, Nouh E S A, Abdel Wahed R S. 2020a. Adsorption of cesium and cobalt onto dried Myriophyllum spicatum L. from radiocontaminated water: experimental and theoretical study. Progress in Nuclear Energy, 125: 103393.
DOI:10.1016/j.pnucene.2020.103393 |
Saleh H M, Moussa H R, Mahmoud H H, El-Saied F A, Dawoud M, Abdel Wahed R S. 2020b. Potential of the submerged plant Myriophyllum spicatum for treatment of aquatic environments contaminated with stable or radioactive cobalt and cesium. Progress in Nuclear Energy, 118: 103147.
DOI:10.1016/j.pnucene.2019.103147 |
Southichak B, Nakano K, Nomura M, Chiba N, Nishimura O. 2008. Marine macroalga Sargassum horneri as biosorbent for heavy metal removal: roles of calcium in ion exchange mechanism. Water Science & Technology, 58(3): 697-704.
DOI:10.2166/wst.2008.696 |
Sun S, Wang F, Li C L, Qin S, Zhou M J, Ding L P, Pang S J, Duan D L, Wang G C, Yin B S, Yu R C, Jiang P, Liu Z L, Zhang G T, Fei X G, Zhou M. 2008. Emerging challenges: massive green algae blooms in the Yellow Sea. Nature Precedings.
DOI:10.1038/npre.2008.2266.1 |
Wang J L, Zhuang S T, Liu Y. 2018a. Metal hexacyanoferratesbased adsorbents for cesium removal. Coordination Chemistry Reviews, 374: 430-438.
DOI:10.1016/j.ccr.2018.07.014 |
Wang X M, Shan T F, Pang S J. 2018b. Phytoremediation potential of Saccharina japonica and Sargassum horneri(phaeophyceae): biosorption study of strontium. Bulletin of Environmental Contamination and Toxicology, 101(4): 501-505.
DOI:10.1007/s00128-018-2435-0 |
Wang X M, Shan T F, Pang S J. 2021. Removal of Sr, Co, and Mn from seawater by Sargassum horneri in mono- and multi-nuclide contamination scenarios. Journal of Applied Phycology, 33(4): 2587-2596.
DOI:10.1007/s10811-021-02477-1 |
Xiao H Q, Zhu S, Wang H W. 2011. Risk assessment on the Japan Fukushima nuclear accident. Inspection and Quarantine Science, 21(2): 68-71.
(in Chinese with English abstract) |
Zhao J, Jiang P, Qin S, Liu X J, Liu Z Y, Lin H Z, Li F C, Chen H X, Wu C H. 2015. Genetic analyses of floating Ulva prolifera in the Yellow Sea suggest a unique ecotype. Estuarine, Coastal and Shelf Science, 163: 96-102.
DOI:10.1016/j.ecss.2015.05.027 |
Zhuang S T, Yin Y N, Wang J L. 2018. Simultaneous detection and removal of cobalt ions from aqueous solution by modified chitosan beads. International Journal of Environmental Science and Technology, 15(2): 385-394.
DOI:10.1007/s13762-017-1388-x |
 2022, Vol. 40
2022, Vol. 40


