Institute of Oceanology, Chinese Academy of Sciences
Article Information
- SUN Chunyang, HUANG Duanjie, XU Qiang, GAO Fei, LI Xiubao, WANG Aimin
- Diverse habitat preferences of two sea cucumber species and the seasonal change in a coral reef area
- Journal of Oceanology and Limnology, 40(4): 1578-1591
- http://dx.doi.org/10.1007/s00343-021-1254-z
Article History
- Received Aug. 13, 2021
- accepted in principle Oct. 4, 2021
- accepted for publication Oct. 18, 2021
2 College of Marine Science, Hainan University, Haikou 570228, China;
3 State Key Laboratory of Marine Resource Utilization in South China Sea, Hainan University, Haikou 570228, China;
4 Hainan Provincial Ecological and Environmental Monitoring Centre, Haikou 571126, China
Most tropical sea cucumbers have high commercial value, but little is reported about their ecological knowledge (Dissanayake and Stefansson, 2012). Understanding its basic biological and ecological information is conducive to rational utilization and protection of precious tropical sea cucumber resources (Wiedemeyer, 1994; Mercier et al., 1999; Purcell, 2004; Conand, 2008). The distribution variation of benthic marine invertebrate populations over time and space are related to complex ecological processes (Mercier et al., 1999). For example, Aspidochirotida sea cucumbers are ubiquitous and conspicuous members in the coral reef ecosystem and play an important ecological role in nutrient cycling and bioturbation processes (Cowen and Sponaugle, 2009; Purcell et al., 2016). Alvarado et al. (2012) found the coral reefs with high species richness and diversity index of echinoderm have a wide variety of coral species, good growth, and large distribution area. Population dynamics are essential for the determination and management of the bio-resource protecting area, but little is known about the temporal dynamics of sea cucumber populations and how these dynamics are related to seasonal ecological change in their habitats (Eriksson et al., 2013), which makes the spatial planning of sea cucumber protecting area on coral reefs difficult (Parma et al., 2005; Crowder and Norse, 2008).
Holothuria edulis and Stichopus chloronotus are common species in coral reefs in tropical seas, and S. chloronotus have high economic value. The two sea cucumber species inhabit different habitats. H. edulis mainly inhabit in the soft bottom habitat dominated by sand (Fig. 1a). They have irregular feeding behaviors and prefer to eat large-grained sediments (Liao, 1997). S. chloronotus mainly lives in hard bottom habitat such as coral and rocky reefs (Fig. 1b). They feed on the fine sediments on the surface, and their feeding time is relatively regular (Liao, 1997).

|
| Fig.1 Holothuria edulis (a) and Stichopus chloronotus (b) |
Resources are often unevenly distributed in nature, hence animals must constantly adjust in the process of behavioral response (Hutto, 1985; Block and Brennan, 1993), and show the characteristics of seasonal habitat selection (Zhang and Li, 2005; Conand, 2008; Slater and Jeffs, 2010; Yamana et al., 2010). The habitat selection of sea cucumbers is affected by biological and inorganic environmental factors, mainly involving feeding selection, habitat heterogeneity, hidden behaviors, competition, and water flow (Roberts, 1979; Sloan and Von Bodungen, 1980; Entrambasaguas et al., 2008; Morgan, 2011). At present, reports on tropical sea cucumbers mainly focus on population distribution, community structure, and habitat selection. There is still a lack of detailed research on the habitat factors that affect the habitat selection of sea cucumbers and the seasonal changes of their preferred habitats.
The coordination between habitat type and habitat selection of benthic animals are an obvious phenomenon in coral reef area (Entrambasaguas et al., 2008; Shiell and Knott, 2010). It is reported that sea cucumber population demography mostly depends on environmental parameters such as food and hydrodynamics (Byrne et al., 1998; Ebert et al., 1999). Besides, substrate type for inhabitation and available detritus food source are also key factors influencing the population metrics of sea cucumbers (Uthicke and Karez, 1999; Hermosillo-Nuñez et al., 2016). Habitat preferences vary among different species, and are also impacted by habitat profiles and seasonal changes. For example, H. edulis preferred fine sand substrate in winter but it moved to coral rubble habitat in spring. There are also researches reporting conflicting results concerning the habitat preference in different areas. For example, Holothuria atra is reported to prefer seagrass bed (Dissanayake and Stefansson, 2012), while other scholars found they preferred coral sand bottom (Liao, 1997). Therefore, it is particularly necessary to study the habitat preference and seasonal changes of sea cucumbers in the unique coral reef ecosystem in the South China Sea.
Finding out the seasonal changes in the preferred habitat of sea cucumbers can help us analyze the seasonal changes of its ability to restore a specific coral reef ecosystem and assess the importance of sea cucumbers as an ecological restoration species. Sea cucumbers play a key role in bioturbation and turnover of sediments (Mangion et al., 2004), the deposit fecal can enhance coral calcification and provide a buffer against ocean acidification (Schneider et al., 2011). The seasonal changes of sea cucumber's preferred habitat are closely related to its ecological adaptability, but the precision of previous studies is low on the time scale, so it is difficult to reflect the fine changes of sea cucumber ecological adaptability. It is essential to understand their migration behavior through different coral reef habitats. A fine-scale study is needed to cover the reasons why different species select different habitat types and how it changes throughout the year.
Most previous studies on sea cucumber population or community were conducted by the method of large-scale random sample survey, which is difficult to fix the specific habitat profiles and other external biotic and abiotic factors, such as water depth and benthic substrate, so it is not suitable for the study of sea cucumber habitat preference on a fine time scale. In this study, the fixed transect method was used to select three fixed transects with different habitat combinations, and regular underwater video recordings were conducted within one year to study the population migration of sea cucumbers among different habitat types in details. Therefore, the goals were to find the distribution patterns and preferred habitat changes of two predominant sea cucumber species in the coral reef ecosystem; to explore whether the seasonal change of sea cucumber's preferred habitat is gradually migrated or realized in the short term of seasonal alternation; to explore the effect of depth gradient on the distribution of two sea cucumber species; and to find the habitat factors that affect the distribution of two sea cucumber species.
2 MATERIAL AND METHOD 2.1 Study area and fixed transectWuzhizhou Island (18°31′N, 109°77′E; area 1.48 km2) is a famous tourist region in China with the best coral reef around Hainan Island and excellent water and sediment quality. There are over 90 reef corals with an average coverage of 28.18% (41.85% highest on the south side) (Li et al., 2019). At least 12 sea cucumber species live here with a mean density of 3 individuals per 10 m2 (inds./10 m2) (Huang et al., 2020). The good protection is mainly due to the management of the tourist company and the technical support of our research team. Coral reef restoration has been carried out over six years. Commercial trawling fishing is completely prohibited in the study area, and only recreational fishing on one boat belonging to the tourism company is permitted in Wuzhizhou island sea ranching area. It is a perfect place for the biodiversity preservation of sea cucumber species.
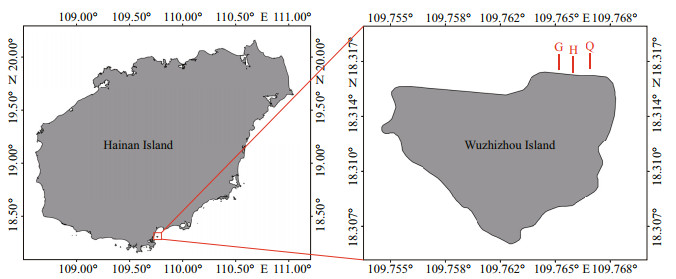
During the preliminary benthic habitat survey, it was found that the coastal area on the north side of the Wuzhizhou Island is a compound distribution area of living coral reefs and sandy bottom, with gentle seabed topography and abundant sea cucumber population resources (predominant species are Holothuria edulis, Holothuria atra, and Stichopus chloronotus). It is also the main area for reef diving tourism in summer. Therefore, the study was carried out in this area (18°19.000′–18°19.067′N, 109°45.890′–109°46.009′E). Three parallel transects which are perpendicular to the coastline were set from the west to the east and abbreviated as G, H, and Q (Fig. 2). The length of each transect is 100 m, using a 100-m long rope (1-cm precision). The interval between transects was 30–50 m. Transects extends from shallow water to deep water. The transects were set within the depth of 15 m, crossing the sand substrate and coral reef substrate, and cover all types of habitat types of the area (the transects snapshots are in the attachment).
|
| Fig.2 Study area and three fixed transects at Wuzhizhou Island |
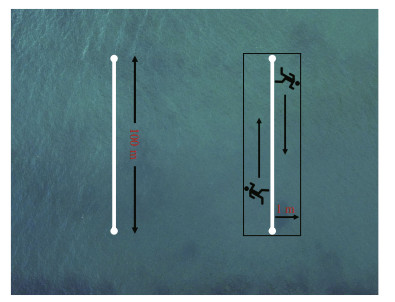
The survey time is January, April–September, 2019, and a 2-person diving team undertook this work. When monitoring, the divers took the videos of each transect with a high-resolution underwater camera (Gopro 5, USA, the resolution is 2.7 kp and the frame rate is 60 frames/s). The average swimming speed of the divers was 0.17 m/s to get high-quality video. During the investigation of each transect, divers started from the left side of the rope to record the habitat and target sea cucumbers within 1 m of the left side, then turned back at the end of 100 m and continued to take the video along the right side of the transect. The target sea cucumbers within 1 m on both sides of the rope were monitored, making a 200-m2 survey area (Fig. 3). The depth gradient of the transects (10-m interval) was measured by the diver with a diving computer watch. When getting back to the lab, the population density of the two species with the habitat profiles were analyzed based on the video.
|
| Fig.3 The layout of fixed transects and shooting mode |
In bioresource evaluations, the mean density is commonly used to represent the distribution of sea cucumbers in a certain area, which will potentially simplify a markedly heterogeneous spatial distribution and mask local variation in population structure, and the mean density has limited reference to spatial scales of populations (Purcell et al., 2009; Eriksson et al., 2010). Thus, a population density profile may be better suited to improving resolution in the analysis of evaluation data (Eriksson et al., 2013). In our study, instead of calculating the population density of each transect totally, we divided each transect into 10 subdistricts of 20 m2 each (10 m×2 m) to count the population density of each sub-district and then combined them into a complete population density profile. It can better reflect the relationship between sea cucumbers' population density variations and habitat profiles.
2.3 Division of dry season and rainy seasonSanya sea area is tropical monsoon climate, the annual variation of temperature in this area is small (22-29 ℃), but the seasonal precipitation has a great impact on the marine environment, showing a distinct division of dry season (January to June) and rainy season (July to November.) (Fig. 4). The monthly precipitation and temperature data of Sanya in 2019 are taken from the reports of the Agricultural Meteorology of Hainan Province, and delivered by the Institute of Meteorology Sciences of Hainan (http://www.hainanqx.cn/UI/Index/Index.aspx). The division method of dry season and rainy season is as follows: Wuzhizhou Island is located in the tropics, affected by the tropical monsoon climate. The precipitation from January to June and December is small, the monthly precipitation is below 65 mm on averaged of 16.8 mm, which is the dry season. The monthly precipitation from July to November is more than 120 mm and averaged as 170 mm, which is the rainy season (Fig. 4).
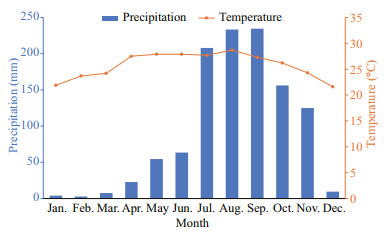
|
| Fig.4 Monthly average precipitation and temperature in Sanya in 2019 |
A multi-parameter water quality detector (YSI-650, USA) was used to measure the water temperature, salinity and dissolved oxygen of the surface water (1-m underwater) in the investigated sea area.
2.4 Determination of habitat types in study areaAccording to the habitat characteristics of the investigated sea area, the habitat types can be divided into the following six types: sand/silt (S), small rock (SR) (diameter below 15 cm), big rock (BR) (diameter above 15 cm), small coral reef (SCR), big coral reef (BCR), and dead coral rubbles (DCR) (Bellchambers et al., 2011; Dissanayake and Stefansson, 2012). The number of H. edulis and S. chloronotus in each subdistrict were counted, and the habitat types of each sea cucumber were distinguished according to the division method of the above six habitat types, then the number of two sea cucumber species in each habitat type in each month was summarized for further statistical analysis.
2.5 Determination of habitat variablesAfter mapping the detailed habitat types of the three transects, we selected six habitat variables to further explore and quantify the impact of environmental factors on the habitat preference of two sea cucumber species (Eriksson et al., 2012; Li et al., 2012). The chosen principle of habitat variables is to extract and integrate the characteristics of habitat types to make them representative, and their changes will affect the distribution of sea cucumbers.
We divided habitat variables into three categories: soft substrate (including the plain top surface of big coral rock covered with fine deposit), hard substrate, and dead coral rubble. Soft substrate contained two habitat variables: sand coverage (SA) and sand continuity (SC). Hard substrate contained three habitat variables: reef coverage (RC), reef undulation degree (RU), and live coral coverage (LC). Dead coral rubbles (DC) were scattered in soft and hard substrate, which is difficult to be defined as an independent variable. Therefore, DC was taken as a supplemental factor to analyze whether its presence or absence would affect the preferred habitat selection of sea cucumber. Table 1 lists the detailed assignment methods of these habitat variables.
Therefore, each transect was further divided into 20 sub-divisions of 10 m2 each (5 m×2 m) for more precise quantification of habitat variables. Two bottom snapshots at both sides of one sub-division were got, and ImageJ was used to quantify habitat variables based on the snapshots (Bottom snapshots of every 10 m of G, H, and Q transects are attached in the attachment). Then correlation analysis was carried out on the distribution of sea cucumber and habitat variables.
2.6 Data processing 2.6.1 Habitat density of two sea cucumber speciesThe population density of the sea cucumber was calculated as inds./10 m2 in different months and water depths. The formula (Eq.1) is as follows:
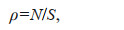 (1)
(1)where ρ is the habitat density of sea cucumber, N is the number of sea cucumbers in the target sub-division, and S is the area of the target sub-division. The unit of habitat density of sea cucumber is inds./10 m2.
2.6.2 Habitat proportion of two sea cucumber speciesThe total number of H. edulis and S. chloronotus in three transects was counted every month. The number of them in the six habitat types was also counted separately, and then the result was transformed to the proportion of the target sea cucumbers in a certain habitat type in all of the population. The formula (Eq.2) is as follows:
 (2)
(2)where P is the proportion of the target sea cucumbers in a certain habitat type in a certain month, n is the number of the sea cucumbers in a certain habitat type in a certain month, and N is the total number of sea cucumbers in the corresponding month.
2.6.3 One-way ANOVAOne-way ANOVA was used to analyze the differences of total number of H. edulis vs. S. chloronotus, the number of H. edulis and S. chloronotus in dry season vs. rainy season, and the habitat density of H. edulis and S. chloronotus in different transects and habitats. Then, one-way ANOVA was also used to analyze the habitat differences between the sandy habitat and coral reef habitat of the three transects under the premise that the data conforms to normal distribution, homogeneity of variance, and independence. R version 4.0.3 was used for the above statistical analyses.
2.6.4 Correlation analysisR version 4.0.3 was used to analyze the correlation between the number of H. edulis and six habitat factors in each transect, and the method used was Spearman's correlation coefficient.
3 RESULT 3.1 Water quality characteristics of the studied sea areaDuring the seven monitoring months in 2019, the water temperature (WT) varied from 23.74 ℃ to 29.31 ℃, with the lowest water temperature existed in January and the highest in September. In July, the water temperature showed a relatively low period due to the dominance of an upwelling cold water mass invasion at the east coast of Hainan Island. The salinity ranged from 33.21 to 34.05, with no remarkable difference in the monthly survey. The range of dissolved oxygen (DO) was 5.16–7.10 mg/L, with the highest in September and the lowest in April (Table 2).
G transect is located at the west. The proportion of sand coverage (SA) and sand continuity (SC) increased with the distance from the shore and water depth, whereas the proportion of live coral coverage (LC) and reef coverage (RC) decreased. The reef undulation degree (RU) is large in G transect. No dead coral rubble (DC) was observed in the first 10 m of the transect.
H transect is located in the middle, with the sand coverage (SA) accounted for the lowest proportion among three transects, while the reef coverage (RC) accounted for the highest proportion among three transects. The sand continuity (SC) of this transect is the lowest (sand continuity of six sub-divisions is 0), while the reef undulation degree (RU) is the largest. No dead coral rubble (DC) was observed in the habitat within 60–80 m, and the proportion of live coral coverage (LC) decreased with the increase of water depth.
Q transect is located in the northeast corner of Wuzhizhou Island. The proportion of sand coverage (SA) gradually increased with the distance from the shore, while the proportion of reef coverage (RC) decreased. The sand continuity (SC) fluctuates greatly in the second half of the transect, and the reef undulation degree (RU) is lower than other two transects. No dead coral rubble (DC) was observed in the 20–30-m habitat. The proportion of live coral coverage (LC) among different sub-divisions in the transect is relatively stable (S2=0.025).
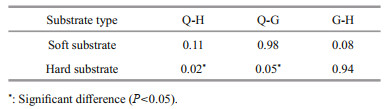
The results of one-way ANOVA showed that there was difference between the soft substrate of H and Q, H and G transects, but not statistically significant (P=0.11 for H-Q and 0.08 for H-G), while a high similarity was found between transect Q and G (P=0.98). For hard substrate, there was significant difference between Q and H, Q and G transects (P=0.02 for Q-H and 0.05 for Q-G), while the similarity of hard substrate of G and H transects was high (P=0.94) (Table 3).
The population density for Holothuria edulis and Stichopus chloronotus varied from 2.0–8.9 and 0.2–0.7 inds./10 m2, respectively. The overall density of S. chloronotus was significantly lower than H. edulis (t-test, P < 0.001). The density of two species showed a different dynamic change throughout the sampling period. The density of H. edulis was low in winter, and gradually increased in spring and summer, reaching a peak value in August. The density variation of S. chloronotus was not obvious from January to September, only showing a slight increase from June to September. Meanwhile, both of the two species showed a significantly higher population density in rainy season than in dry season (t-test, P=0.021 for H. edulis and 0.008 for S. chloronotus). In rainy season, the average population density of H. edulis and S. chloronotus were 7.99 and 0.66 inds./10 m2, respectively. In dry season, the average population density of H. edulis and S. chloronotus were only 4.40 and 0.36 inds./10 m2, respectively (Fig. 5).
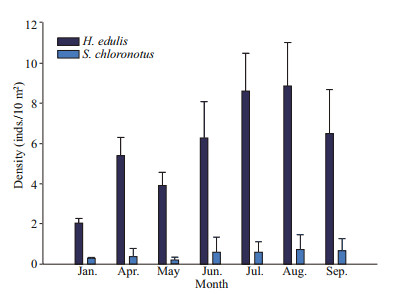
|
| Fig.5 Monthly population density of Holothuria edulis and Stichopus chloronotus |
There was a spatial variation in the number of H. edulis and S. chloronotus among three transects. The results of one-way ANOVA show that the number of H. edulis in G and H transects was higher than that in Q transects, but it was not significant (P>0.05). The number of H. edulis in G and H transects within 50-100 m was much higher than that in Q transect, and the number of H. edulis in G transect was the highest among sampling months (Fig. 6a). For S. chloronotus, the transect of Q that located near the rocky coast at the east had the greatest number of individuals than the other two transects', and the results of one-way ANOVA are significant (P < 0.05). The number of S. chloronotus in Q transect showed a small-clustered distribution in the range of 20–70 m, while it was the least in G transect and absent in April and May (Fig. 6b). In addition, the patching distribution phenomenon of S. chloronotus was more frequent in rainy season.
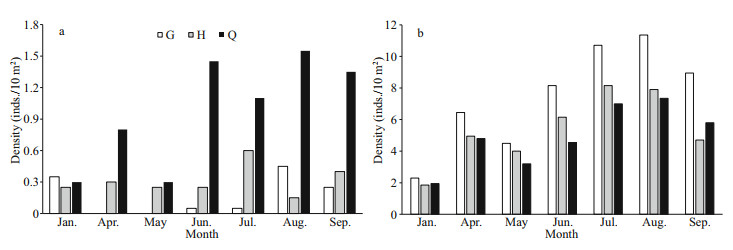
|
| Fig.6 Monthly number of Holothuria edulis (a) and Stichopus chloronotus (b) in three transects |
The mean depth of the transects ranged 3.3–10.7 m. We divided the transects into 10 sub-districts at the interval of 10 m from shore side as the start point, and each one had a population density that corresponds to depth. Results show that higher density of H. edulis was found in deeper water, mainly distributed in the depth of 4.5–10.7 m (Fig. 7a), but most of S. chloronotus lived in the depth of 4.2–8.5 m, and there has no obvious relationship between density and depth (Fig. 7b). S. chloronotus tended to be distributed in patch, and their number could reach up to 9 in one sub-district. The population density of H. edulis increased with the water depth; for example, the population density in the first sub-district (near the shore side) was 0.15 inds./10 m2, and increased to 1.16 inds./10 m2 in the last sub-district (Fig. 7a). When rainy season arrived in July, the density of H. edulis in the depth of 6.4–10.7 m increased dramatically, indicating that more H. edulis aggregated in deeper water (Fig. 7a).

|
| Fig.7 Monthly relationship between the population density of Holothuria edulis (a) and Stichopus chloronotus (b) with depth gradient |
The density of the two sea cucumber species at each habitat type varied significantly among different months throughout the sampling period. There was a distinct different temporal dynamic change between two species.
3.6.1 Holothuria edulisIn dry season, sand (S), small rock (SR), and dead coral rubble (DCR) were the preferred habitats and the number of H. edulis inhabited accounted for about 88.5%. In January, over half of the H. edulis individuals lived in the S (52%) followed by the DCR (21%). From April to July, SR was another most preferred habitat of H. edulis, accounting for 25.3%–37.2%. As the rainy season came at July, small coral reef (SCR) became one of the preferred habitats of H. edulis, and its population proportion gradually increased from 9.5% to 37%. The proportion of S and SR habitats decreased in varying degrees. In August and September, 32.3%–35.2% of H. edulis lived in DCR habitat. The habitat preference difference of H. edulis between dry season and rainy season was realized through monthly migration (Fig. 8a).

|
| Fig.8 The proportion of Holothuria edulis (a) and Stichopus chloronotus (b) in six habitat types in each month S: sand; SR: small rock; BR: big rock; SCR: small coral reef; BCR: big coral reef; DCR: dead coral rubble. |
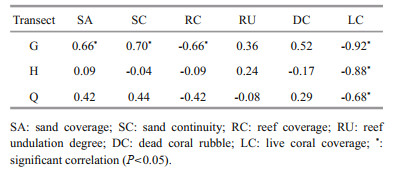
In G transect, the results of Spearman's correlation coefficient showed that there was a strong negative correlation between the number of H. edulis and the live coral coverage (LC) in each month (P < 0.05). The number of H. edulis per month was positively correlated with sand coverage (SA) and sand continuity (SC), and negatively correlated with reef coverage (RC) (P < 0.05). In Q and H transects, the monthly number of H. edulis was negatively correlated with the live coral coverage (LC) (P < 0.05), but no significant correlation with other habitat variables. In all three transects, H. edulis has no significant correlation with reef undulation degree (RU) and dead coral rubble (DC) (P>0.05) (Table 4).
Comparatively, the total observed number of S. chloronotus was much less than H. edulis (201 vs. 2 489), and it showed a different habitat preference and temporal dynamic. In dry season, the preferred habitats of S. chloronotus were small rock (SR), sand (S), and small coral reef (SCR), and the number of them account for about 87%. The most preferred habitat of S. chloronotus in dry season is SR, the number of it in January, April, May, and June accounted for 38.9%, 50%, 50%, and 50%, respectively. Meanwhile, S. chloronotus living in sand (S) habitat reduced remarkably (44.4%–5.9%) from January to June, while it gradually increased in dead coral rubble (DCR) habitat (0–17.6%). In rainy season, SCR habitat was the most preferred habitat of S. chloronotus, accounting for 51.4%–65.1% of the total population. The proportion of S. chloronotus in SR habitat decreased remarkably (42.9%–5.1%), while the proportion of it in DCR habitat increased gradually (2.9%–23.1%). The proportion of S. chloronotus in S habitat was less than 10%. Meanwhile, this species rarely selects big rock (BR) and big coral reef (BCR) habitats (total≤2.3%). The habitat preference difference of S. chloronotus between dry season and rainy season was also realized through monthly migration (Fig. 8b).
By observing the proportion of S. chloronotus in each habitat type, we can conclude that S. chloronotus has obvious preference for hard bottom habitat, and mainly concentrated in SR and SCR habitats.
4 DISCUSSION 4.1 Seasonal population density variation of the two speciesThe study found that the population density of Holothuria edulis was significantly higher than that of Stichopus chloronotus, and it may because of the strong adaptability or a higher reproductive efficiency of H. edulis. Another possible reason is that S. chloronotus has a high economic and edible value, and "sea cucumber rush" raised in recent years has caused the overfishing of it. Bellchambers et al. (2011) found that the population densities of H. edulis and S. chloronotus in the coral reef habitat of the tropical Indian Ocean were only 0.05 and 0.04 inds./10 m2, which is much lower than that in our study sea area. It is reported that there existed overfishing of S. chloronotus in the Indian Ocean (Dissanayake and Stefansson, 2010), whereas our study area is well protected by the Tourism Management Department of Wuzhizhou Island. The compound habitat of living coral reefs and sandy bottom in the northern part of the island showed a highly habitat heterogeneity, which is suitable for the growth of sea cucumbers.
The results show that the population density of two species varied among seasons. Both of the two species showed the lowest population density in winter (January), and the population gradually increased with the rising of water temperature. In winter, the study area is affected by the northeast monsoon, leading to a hostile water environment with turbulent flow. Sea cucumbers tend to migrate to deep water to find mild habitats, so the number of sea cucumbers in the fixed transects was low. In addition, the population density of the two species was quite different in dry season and rainy season, which may be related to the food richness. Li et al. (2015) found that there are many kinds of macroalgae in the adjacent coastal area of the Haitang Bay in Sanya. Abundant rainfall in rainy season brought a large amount of fresh water with rich nutrients into the sea, consequently supported the bloom of the macroalgae and pelagic and benthic microalgae, supplying sufficient food for sea cucumbers.
4.2 Reasons for spatial distribution difference of the two speciesThe results of one-way ANOVA show that the number of H. edulis in G and H transects was more than that in Q transect (P>0.05), and the number of S. chloronotus in Q transect was significantly higher than that in G and H transects (P < 0.05). It probably relates to the habitat heterogeneity of the three transects. A similar phenomenon was also reported on the species of Stichopus herrmanni in Shark Alley (Australia) with significant population variation among sites, which was related to habitat heterogeneity (Eriksson et al., 2013). We found that more H. edulis distributed in deeper water and the number of H. edulis in G and H transects within 50–100 m was remarkably higher than that in Q transect. Water flow and sediment type can influence the habitat selection of H. edulis (Dar and Ahmad, 2006; Dissanayake and Stefansson, 2012), and the deeper water area in the north of the island is mainly sandy bottom. The water flow in G and H transects is relatively stable, which are very suitable for the survival of H. edulis with sedimentary food habit and hiding behavior. However, Q transect is located in the northeast corner of the island with turbulent flow.
Meanwhile, as a tourist area, the species density difference in our study may also be related to human activities. There are frequent diving and sea sport activities in the shallow water area of G and H transects, which may drive sea cucumbers to migrate to deep water or other places not affected by human activities. Another possible reason may relate to the weak tube foot adsorption ability of H. edulis compared with S. chloronotus, so it cannot stay firm on sand bottom with turbulent water flow. Interestingly, S. chloronotus with strong adsorption foot mainly distributed in Q transect with more hard substrates and strong waves. In January, April, and May, the population number of S. chloronotus was very small among all of the study areas, while in June to September, it increased significantly and appeared small-scale cluster distribution, which may be related to patchy food available. Many sea cucumber species show cluster distribution due to food source, seasonal changes, and habitat types, such as Stichopus japonicus showed obvious aggregation in summer and relative dispersion in winter (Zhang, 2015). Besides, S. herrmanni with a homogenous distribution showed dense aggregates in some occasions (Eriksson et al., 2013).
4.3 Relationship between sea cucumber distribution and water depth gradientThe population distribution of the two species varied with water depth. It shows that H. edulis which preferred coral rubbles and sandy bottom likes living in a deeper water area, with the highest density of 2.65 inds./10 m2. However, water depth showed no remarkable impact on the distribution pattern of S. chloronotus, and they preferred patchy distribution. Dissanayake and Stefansson (2012) found that the distribution of H. edulis was greatly affected by depth, substrate type, and sediment. In our study, the area of sandy bottom and the sediment increased in deeper water, which is more attractive to H. edulis. It was found that most of the H. edulis individuals in deep water were large and directly exposed to the sandy bottom by the underwater video, and they need not hide because of the abundant food sources and stable water flow. Conand (1993) found that the size of S. herrmanni increased with depth and suggested this species migrated from shallow to deep water with growth, and so might the H. edulis. Lee et al. (2008) noted that the growth of H. atra has striking plasticity, and the body size increased by 300% after moving to areas with high food content. This may also be the reason for the larger size of H. edulis in sandy bottom with rich sediments. However, some scholars point out that few sea cucumbers are directly exposed to the open sand (Moriarty, 1982; Massin and Doumen, 1986), and we believe that it may relate to the variation of environmental factors in different study areas.
The patchy distribution pattern of S. chloronotus is the result of the interaction of multiple factors. Uthicke and Karez (1999) and Bellchambers et al. (2011) also found that the S. chloronotus in the coral reef of Cocos (Keeling) Islands distributed in patches. Many scholars have found that the wild sea cucumber population will show uneven patchy distribution due to the combined effects of biotic and abiotic factors (Woo et al., 2010; Morgan, 2011; Mmbaga, 2013; Navarro et al., 2013). Uthicke and Karez (1999) also pointed out that S. chloronotus would actively select sediments. The habitat types are scattered in three transects and S. chloronotus are distributed in patches, which may be related to its feeding selection.
In summary, the distribution of the two sea cucumber species with depth gradient may be the comprehensive influence of depth, sediment types together with food patches.
4.4 Reasons for habitat preference and seasonal changes of two speciesMany foreign scholars pointed out that different sea cucumber species have different habitat preferences (Conand and Mangion, 2002; Conand, 2008; Bellchambers et al., 2011). In the current study, the preferred habitat also showed a distinct speciesspecific character, and seasonal variation was obvious. Conand (2008) and Dissanayake and Stefansson (2012) found that the H. edulis had a distinct aggregation phenomenon in the coral reef and rocky reef area. In this study, the H. edulis also showed an evident preference for small rock (SR) and small coral reef (SCR), which may be related to the hidden behavior. According to underwater video and divers' observation, H. edulis in shallow water hides inside coral reefs or under reefs to avoid adverse sea conditions, such as the torrent surge in January. The hidden behavior has been confirmed in Holothuria difficilis and Apostichopus japonicus (Conand and Mangion, 2002; Zhang et al., 2015). In addition, H. edulis in this study has a clear preference for sand (S) habitat, which may be related to the feeding habits. H. edulis prefer to eat coarse-grained sediments (Dissanayake and Stefansson, 2012), while S habitat is rich in coral sand. The result of correlation analysis between H. edulis and six habitat variables is consistent with other scholars: H. edulis will live in hard habitats, while also like to ingest sediments in sandy bottom. It is worth noting that there was a strong negative correlation between H. edulis and live coral coverage (LC) in all three transects (P < 0.01), and the influence of LC is remarkably greater than that of the other five habitat factors, which indicates that the H. edulis will deliberately avoid the places with living corals, such as small coral reef (SCR) and big coral reef (BCR).
Stichopus chloronotus prefers small rock (SR) and small coral reef (SCR) habitats in this study, which was consistent with the research conclusion of other scholars (Bellchambers et al., 2011; Zhang et al., 2015). In addition, the habitat preference of sea cucumbers is different in varying life history stages. Eriksson et al. (2012) found the juvenile S. chloronotus preferred soft-bottom habitat, while adult S. chloronotus preferred to live in hard bottom habitats such as rocks and coral reefs, which may be related to its feeding habits and avoidance of current. We found part of S. chloronotus likes feeding on the rocks and coral reefs (with excrement nearby) according to underwater video and divers' observation, while others hide in hard bottom habitat. Uthicke (1994) found the water flow is the main factor affecting the distribution of S. chloronotus, and they tend to live on reef flat with steady water flow. Hard habitats such as coral reefs and rocks are good attachment substrate and can effectively block part of the water flow, providing a mild living place for S. chloronotus. Moreover, Uthicke (1999) and Uthicke and Karez (1999) found that S. chloronotus would actively select food patches and ingest sediments with high microalgae content. There are abundant microalgae in SR and SCR habitats (Zhang et al., 1995), which is more attractive to S. chloronotus.
Both species showed different changing processes during the seasonal changes. Changes in environmental factors (Ocean current, sediment source, precipitation, and temperature) were not instantaneous, therefore, the change of sea cucumber's preferred habitat was also gradually migrated. The main habitat of the two species was S in January, and it gradually moved to SR and SCR after April, which may be related to avoiding hostile sea conditions and choosing food patches. In January, both species tended to migrate to deeper waters to seek shelter against violent waves. The individual size of S. chloronotus is large, and the ventral tube foot can make it firmly adsorb on the hard substrate. Therefore, S. chloronotus also showed a preference for SR habitat in January and April. The preference of sea cucumbers for reefs with microalgae was obvious. Uthicke and Karez (1999) found that S. chloronotus had a higher requirement on microalgae content in habitat patches. Zhang et al. (2015) further confirmed it in the behavior of A. japonicus. In our study area, abundant rainfall in the rainy season is conducive to the growth of algae, consequently bringing abundant algae-derived detritus food to the sediment. Therefore, the number of sea cucumbers in small rock (SR) and small coral reef (SCR) habitats gradually increased from June to September, especially the S. chloronotus. Comparatively, H. edulis still preferred sand (S) habitat for its feeding selection of coarse-grained sediments.
5 CONCLUSIONIn this study, the population density of H. edulis was significantly higher than that of S. chloronotus, and the density of H. edulis increased with the depth of water. In winter, sea cucumbers tended to migrate to deep water due to poor sea conditions, and then the density of two target sea cucumber species increased with the increase of temperature and abundant food source in rainy season. H. edulis preferred soft substrates with stable water flow, while S. chloronotus preferred hard substrates and distributed in small clusters in rainy season. The preferred habitats of H. edulis and S. chloronotus changed seasonally and gradually migrated. The shift from dry season to rainy season was the key factor affecting their habitat preference. The results of correlation analysis show that H. edulis could deliberately avoid the habitats with living corals, such as small coral reef (SCR) and big coral reef (BCR). These results are helpful to analyze the seasonal variation of the two sea cucumber species to restore a specific coral reef ecosystem, and to assess their importance as ecological restoration species.
6 DATA AVAILABILITY STATEMENTThe datasets generated and/or analyzed during the current study are not publicly available as the datasets also forms parts of an ongoing study, but are available from the corresponding author on reasonable request.
7 ACKNOWLEDGMENTThe authors are grateful to Fengguo WANG and Zhigang ZHOU from Wuzhizhou Island Tourism Company for their help in fieldwork.
Alvarado J J, Guzman H M, Breedy O. 2012. Distribution and diversity of echinoderms (Asteroidea, Echinoidea, Holothuroidea) in the islands of the Gulf of Chiriqui, Panama. Revista de Biologia Marina y Oceanografia, 47(1): 13-22.
DOI:10.4067/S0718-19572012000100002 |
Bellchambers L M, Meeuwig J J, Evans S N. 2011. Modelling habitat associations of 14 species of holothurians from an unfished coral atoll: implications for fisheries management. Aquatic Biology, 14(1): 57-66.
DOI:10.3354/ab00381 |
Block W, Brennan L A. 1993. The Habitat Concept Theory and Applications. New York, Plenum Press.
|
Byrne M, Andrew N L, Worthington D G, Brett P A. 1998. Reproduction in the diadematoid sea urchin Centrostephanus rodgersii in contrasting habitats along the coast of New South Wales, Australia. Marine Biology, 132(2): 305-318.
DOI:10.1007/s002270050396 |
Conand C, Mangion P. 2002. Sea cucumbers on La Reunion Island fringing reefs: Diversity, distribution, abundance and structure of the populations. SPC Beche-de-mer Information Bulletin, 17: 27-33.
|
Conand C. 1993. Ecology and reproductive biology of Stichopus variegatus an Indo-Pacific coral reef sea cucumber (Echinodermata: Holothuroidea). Bulletin of Marine Science, 52(3): 970-981.
|
Conand C. 2008. Population status, fisheries and trade of sea cucumbers in Africa and the Indian Ocean. In: ToralGranda V, Lovatelli A, Vasconcellos M eds. Sea Cucumbers. A Global Review of Fisheries and Trade. FAO, Rome. p. 143-193.
|
Cowen R K, Sponaugle S. 2009. Larval dispersal and marine population connectivity. Annual Review of Marine Science, 1: 443-466.
DOI:10.1146/annurev.marine.010908.163757 |
Crowder L, Norse E. 2008. Essential ecological insights for marine ecosystem-based management and marine spatial planning. Marine Policy, 32(5): 772-778.
DOI:10.1016/j.marpol.2008.03.012 |
Dar M A, Ahmad H O. 2006. The feeding selectivity and ecological role of shallow water holothurians in the Red Sea. SPC Beche-de-mer Information Bulletin, 24: 11-21.
|
Dissanayake D C T, Stefansson G. 2010. Abundance and distribution of commercial sea cucumber species in the coastal waters of Sri Lanka. Aquatic Living Resources, 23(3): 303-313.
DOI:10.1051/alr/2010031 |
Dissanayake D C T, Stefansson G. 2012. Habitat preference of sea cucumbers: Holothuria atra and Holothuria edulis in the coastal waters of Sri Lanka. Journal of the Marine Biological Association of the United Kingdom, 92(3): 581-590.
DOI:10.1017/S0025315411000051 |
Ebert T A, Dixon J D, Schroeter S C, Kalvass P E, Richmond N T, Bradbury W A, Woodby D A. 1999. Growth and mortality of red sea urchins Strongylocentrotus franciscanus across a latitudinal gradient. Marine Ecology Progress Series, 190: 189-209.
DOI:10.3354/meps190189 |
Entrambasaguas L, Pérez-Ruzafa Á, García-Charton J A, Stobart B, Bacallado J J. 2008. Abundance, spatial distribution and habitat relationships of echinoderms in the Cabo Verde Archipelago (eastern Atlantic). Marine and Freshwater Research, 59(6): 477-488.
DOI:10.1071/MF07109 |
Eriksson B H, De La Torre-Castro M, Eklöf J, Jiddawi N. 2010. Resource degradation of the sea cucumber fishery in Zanzibar, Tanzania: a need for management reform. Aquatic Living Resources, 23(4): 387-398.
DOI:10.1051/alr/2011002 |
Eriksson H, Jamon A, Wickel J. 2012. Observations on habitat utilization by the sea cucumber Stichopus chloronotus. SPC Beche-de-mer Information Bulletin, 32: 39-42.
|
Eriksson H, Thorne B V, Byrne M. 2013. Population metrics in protected commercial sea cucumber populations(curryfish: Stichopus herrmanni) on one tree reef, Great Barrier Reef. Marine Ecology Progress Series, 473: 225-234.
DOI:10.3354/meps10054 |
Hermosillo-Nuñez B B, Rodríguez-Zaragoza F A, Ortiz M, Calderon-Aguilera L E, Cupul-Magaña A L. 2016. Influence of the coral reef assemblages on the spatial distribution of echinoderms in a gradient of human impacts along the tropical Mexican Pacific. Biodiversity and Conservation, 25(11): 2137-2152.
DOI:10.1007/s10531-016-1182-y |
Huang D J, Xu Q, Li X B, Xue Y L, Wu P L, Gao F. 2020. The community structure of echinoderms in sandy coral reef area in Wuzhizhou Island, Sanya, China. Oceanologia et Limnologia Sinica, 51(1): 103-113.
(in Chinese with English abstract) |
Hutto R L. 1985. Habitat selection by nonbreeding, migratory land birds. In: Cody M ed. Habitat Selection in Birds. Academic Press, New York. p. 455-476.
|
Lee J, Byrne M, Uthicke S. 2008. The influence of population density on fission and growth of Holothuria atra in natural mesocosms. Journal of Experimental Marine Biology and Ecology, 365(2): 126-135.
DOI:10.1016/j.jembe.2008.08.003 |
Li J H, Huang L L, Zou L M, Kano Y C, Sato T, Yahara T. 2012. Spatial and temporal variation of fish assemblages and their associations to habitat variables in a mountain stream of north Tiaoxi River, China. Environmental Biology of Fishes, 93(3): 403-417.
DOI:10.1007/s10641-011-9928-6 |
Li X B, Li Y C, Xu Q. 2019. Current Situation, Ecological Restoration and Protection Countermeasures of Coral Reefs in Wuzhizhou Island, Sanya. Science Press, Beijing. p.32-39.
(in Chinese)
|
Li Y C, Yang Y, Zheng X Q, Chen S Q. 2015. Health status and influencing factors of coral reef ecosystems in Houhai waters, Sanya, Hainan Province. Chinese Journal of Ecology, 34(4): 1105-1112.
(in Chinese with English abstract) |
Liao Y L. 1997. Fauna Sinica: Echinodermata. Science Press, Beijing. 334p.
(in Chinese)
|
Mangion P D, Taddei D, Frouin P, Conand C. 2004. Feeding rate and impact of sediment reworking by two deposit feeders Holothuria leucospilota and Holothuria atra on fringing reef (Reunion Island, Indian Ocean). In: Heinzeller T, Nebelsick J H eds. Echinoderms: München. Taylor and Francis, London. p. 311-317.
|
Massin C, Doumen C. 1986. Distribution and feeding of epibenthic holothuroids on the reef flat of Laing Island(Papua New Guinea). Marine Ecology Progress Series, 31: 185-195.
DOI:10.3354/meps031185 |
Mercier A, Battaglene S C, Hamel J F. 1999. Daily burrowing cycle and feeding activity of juvenile sea cucumbers Holothuria scabra in response to environmental factors. Journal of Experimental Marine Biology and Ecology, 239(1): 125-156.
DOI:10.1016/S0022-0981(99)00034-9 |
Mmbaga T K. 2013. The effect of fishing on the ecology of sea cucumber (Holothuroidea: Echinodermata) Holothuria scabra and Holothuria nobilis in Tanzanian sea water. International Journal of Development and Sustainability, 2(2): 1099-1126.
|
Morgan A D. 2011. Patterns of distribution and abundance of the temperate sea cucumber Australostichopus mollis on a rocky subtidal reef. New Zealand Journal of Zoology, 38(3): 195-206.
DOI:10.1080/03014223.2011.563426 |
Moriarty D J W. 1982. Feeding of Holothuria atra and Stichopus chloronotus on bacteria, organic carbon and organic nitrogen in sediments of the Great Barrier Reef. Marine & Freshwater Research, 33(2): 255-263.
|
Navarro P G, Garcia-Sanz S, Barrio J M, Tuya F. 2013. Feeding and movement patterns of the sea cucumber Holothuria sanctori. Marine Biology, 160(11): 2957-2966.
DOI:10.1007/s00227-013-2286-5 |
Parma A, Jerez G, Barahona N, Montecinos M, Elias I, Orensanz J. 2005. What are the key elements for the sustainability of "S-fisheries"? Insights from South America. Bulletin of Marine Science, 76(2): 527-556.
|
Purcell S W, Gossuin H, Agudo N S. 2009. Status and Management of the Sea Cucumber Fishery of La Grande Terre, New Caledonia. The WorldFish Center, Penang, Malaysia. 135p.
|
Purcell S W. 2004. Criteria for release strategies and evaluating the restocking of sea cucumbers. In: Lovatelli A, Conand C, Purcell SW, Uthicke S, Hamel J F, Mercier A eds. Advances in Sea Cucumber Aquaculture and Management. FAO, Dalian, China. p. 181-191.
|
Purcell S, Conand C, Uthicke S, Byrne M. 2016. Ecological roles of exploited sea cucumbers. Oceanography and Marine Biology: An Annual Review, 54: 367-386.
|
Roberts D. 1979. Deposit-feeding mechanisms and resource partitioning in tropical holothurians. Journal of Experimental Marine Biology and Ecology, 37(1): 43-56.
DOI:10.1016/0022-0981(79)90025-X |
Schneider K, Silverman J, Woolsey E, Eriksson H, Byrne M, Caldeira K. 2011. Potential influence of sea cucumbers on coral reef CaCO3 budget: a case study at one tree reef. Journal of Geophysical Research: Biogeosciences, 116(G4): G04032.
|
Shiell G R, Knott B. 2010. Aggregations and temporal changes in the activity and bioturbation contribution of the sea cucumber Holothuria whitmaei (Echinodermata: Holothuroidea). Marine Ecology Progress Series, 415: 127-139.
DOI:10.3354/meps08685 |
Slater M J, Jeffs A G. 2010. Do benthic sediment characteristics explain the distribution of juveniles of the deposit-feeding sea cucumber Australostichopus mollis?. Journal of Sea Research, 64(3): 241-249.
DOI:10.1016/j.seares.2010.03.005 |
Sloan N A, Von Bodungen B. 1980. Distribution and feeding of the sea cucumber Isostichopus badionotus in Relation to shelter and sediment criteria of the Bermuda platform. Marine Ecology Progress Series, 2: 257-264.
DOI:10.3354/meps002257 |
Uthicke S, Karez R. 1999. Sediment patch selectivity in tropical sea cucumbers (Holothurioidea: Aspidochirotida) analysed with multiple choice experiments. Journal of Experimental Marine Biology and Ecology, 236(1): 69-87.
DOI:10.1016/S0022-0981(98)00190-7 |
Uthicke S. 1994. Distribution patterns and growth of two reef flat holothurians, Holothuria atra and Stichopus chloronotus. In: David B, Guille A, Feral J P, Roux M eds. Echinoderms Through Time. CRC Press, Rotterdam, Netherlands. p. 569-576.
|
Uthicke S. 1999. Sediment bioturbation and impact of feeding activity of Holothuria (Halodeima) atra and Stichopus chloronotus, two sediment feeding holothurians, at Lizard Island, Great Barrier Reef. Bulletin of Marine Science, 64(1): 129-141.
|
Wiedemeyer W L. 1994. Biology of small juveniles of the tropical holothurian Actinopyga echinites: growth, mortality, and habitat preferences. Marine Biology, 120(1): 81-93.
DOI:10.1007/BF00381944 |
Woo S P, Zulfigar Y, Norhanis M R, Teh C P, Tan S H. 2010. The diversity of sea cucumbers (Echinodermata: Holothuroidea) in five reefs of the South China Sea. Malayan Nature Journal, 62(4): 371-377.
|
Yamana Y, Hamano T, Goshima S. 2010. Natural growth of juveniles of the sea cucumber Apostichopus japonicus: studying juveniles in the intertidal habitat in Hirao Bay, eastern Yamaguchi Prefecture, Japan. Fisheries Science, 76(4): 585-593.
DOI:10.1007/s12562-010-0245-3 |
Zhang B L, Sun D Y, Wu Y Q. 1995. Preliminary analysis on the feeding habit of Apostichopus japonicus in the rochy coast waters off Lingshan Island. Marine Sciences, 19(3): 11-13.
(in Chinese with English abstract) |
Zhang H Y, Xu Q, Liu H, Yang H S. 2015. Preliminary study on the property of bottom mariculture sea cucumber(Apostichopus japonicus) group in Qiansan Islands, Haizhou Bay. Marine Sciences, 39(6): 1-7.
(in Chinese with English abstract) |
Zhang H Y. 2015. Ecology, Distribution and Feeding Habitats of Bottom Sowing Culture Apostichopus japonicus in Qiansan Island, Haizhou Bay. The Institute of Oceanology, Chinese Academy of Sciences, Qingdao. (in Chinese with English abstract)
|
Zhang M H, Li Y K. 2005. The temporal and spatial scales in animal habitat selection research. Acta Theriologica Sinica, 25(4): 395-401.
(in Chinese with English abstract) |
 2022, Vol. 40
2022, Vol. 40