Institute of Oceanology, Chinese Academy of Sciences
Article Information
- SUN Zhihua, WU Jiajia, ZHANG Dun, LI Ce, ZHU Liyang, LI Ee
- Influence of nitrate concentrations on EH40 steel corrosion affected by coexistence of Desulfovibrio desulfuricans and Pseudomonas aeruginosa bacteria
- Journal of Oceanology and Limnology, 40(4): 1448-1461
- http://dx.doi.org/10.1007/s00343-021-1247-y
Article History
- Received Aug. 11, 2021
- accepted in principle Sep. 7, 2021
- accepted for publication Oct. 21, 2021
2 Open Studio for Marine Corrosion and Protection, Pilot National Laboratory for Marine Science and Technology (Qingdao), Qingdao 266237, China;
3 Center for Ocean Mega-Science, Chinese Academic of Sciences, Qingdao 266071, China;
4 University of Chinese Academy of Sciences, Beijing 100049, China;
5 Key Laboratory of Marine Materials and Related Technologies, Ningbo Institute of Materials Technology & Engineering, Chinese Academy of Sciences, Ningbo 315201, China
Microbiologically influenced corrosion (MIC) is a common phenomenon in oil field industries (Lahme et al., 2019). To save cost, seawater is usually used as make-up water in produced water reinjection (PWRI) systems for offshore oil production (Shartau et al., 2010). SO42ˉ in seawater can aggravate the activity of sulfate reducing bacteria (SRB). Notoriously, SRB induce corrosion, and about half of MIC loss is believed to be associated with them (Oguzie et al., 2013; Chen et al., 2018; Jia et al., 2018; Wang et al., 2021). Various attempts have been made to retard the destructive effect of corrosion caused by SRB, including chemical biocides, physical scraping, and biological competition. The overuse of biocides is generally inefficient and causes environmental pollution (Pillay and Lin, 2014), and the biocides themselves may cause corrosion (Zuo et al., 2004). Although physical scraping (pigging) is more effective than bactericide in controlling MIC in seawater pipelines, it is difficult to practice this technique in long and narrow pipes (Costerton, 2007). Compared with the above two methods, bio-competitive exclusion (BCE) method shows unique advantages in cost and environments (Lai et al., 2020). The addition of nitrate is a common BCE method to mitigate corrosion in PWRI systems, which can affect microbial community compositions, especially nitrate reducing bacteria (NRB) and SRB, but its effectiveness is in controversy.
Numerous works have demonstrated that corrosion was inhibited by nitrate addition in PWRI systems. Schwermer et al. (2008) discovered that uniform corrosion in PWRI systems was clearly mitigated by the addition of 1-mmol/L NO3ˉ, and corrosion inhibition was due to the change in microbial communities. Compared to the community with low biomass but rich species without treatment, nitrate introduction increased biomass, reduced diversity, and stimulated the growth of NRB. The biological inhibition played by nitrate on the SRB growth has also been reported by Lai et al. (2020) and corrosion was inhibited accordingly. Moreover, Kebbouche-Gana and Gana (2012) discovered that the total amount of sulfide decreased in static tests with the SRB consortium obtained from the In Amenas injected water, when nitrate was dosed at 120 mg/L. However, the positive role of nitrate addition was doubted by some researchers, and they claimed that it promoted corrosion instead of inhibition. Chen et al. (2013) investigated the corrosion influence of nitrate added with different concentrations in the static experiment simulating Shengli oilfield PWRI system, and found that corrosion rate increased with the addition of 80-mg/L nitrate. Nemati et al. (2001) found that the addition of nitrate to the nitrate-reducing, sulfide-oxidizing bacterium (NR-SOB) and SRB consortium improved the average corrosion rate from 0.2 to 2.9 g/(m2·d). Hubert et al. (2005) discovered that nitrate (17.5 mmol/L) eliminated sulfide but brought pitting corrosion, and shifted the corrosion risk from the bioreactor outlet to the inlet (i.e., from production to injection wells). This corrosion acceleration by nitrate introduction might be related to the activity of NRB, which have been reported to promote steel corrosion by extracellular electron transfer (EET) (Gu et al., 2019). The conflicts in corrosion affected by nitrate addition might be closely associated with differences in nitrate concentration, environment, and microbial community feature, and microbial communities could also be regulated by nitrate concentrations (Reinsel et al., 1996; Hubert et al., 2005; Rodríguez-Gómez et al., 2005). Consequently, the concentration of nitrate is believed to play a significant role in the conflicts.
In the present work, nitrate was added at different concentrations into a microbiota consisting of Desulfovibrio vulgaris and Pseudomonas aeruginosa, which were typical strains of SRB and NRB. Desulfovibrio and Pseudomonas species are commonly found in seawater, and dominated in enriched produced water (Batmanghelicha et al., 2017). EH40 steel corrosion in the biotic systems was investigated, and the corrosion influence mechanism of nitration introduction was proposed.
2 EXPERIMENTAL 2.1 Material and specimenEH40 steel was manufactured by Nanjing Iron & Steel Co., Ltd., and its chemical composition (wt.%) was C 0.086, Si 0.24, Mn 1.49, P 0.011, Al 0.034, Nb 0.034, Cu 0.028, Cr 0.118, Ni 0.013, Ti 0.01, Mo 0.001 and Fe balance. Coupons with dimensions of 20 mm×10 mm×5 mm, 10 mm×10 mm×5 mm, and 5 mm×5 mm×3 mm were utilized for weight loss, electrochemical tests, and X-ray photoelectron spectroscope (XPS), respectively. To avoid crevice corrosion, the flanks of electrochemical coupons were coated with polytetrafluoroethylene. After copper wires were welded, the samples were embedded with epoxy resin, leaving an exposed area of 100 mm2. All coupons were polished with a series of abrasive papers (80, 240, 400, and 800 grit), washed with pure ethyl alcohol, and dried with warm air. Before corrosion tests, they were wrapped by aluminum foil and sterilized in an autoclave (LDZM-40KCS; Shanghai Shenan Medical Instrument Co., Ltd.) at 121 ℃ for 20 min. Our previous work has shown that the high-temperature sterilization has no effect on epoxy resin or coupons (Gao et al., 2018).
2.2 Bacterial cultivation and experimental system organizationPseudomonas aeruginosa was cultivated in 2216E medium containing yeast extract 1 g, tryptone 5 g, and FePO4 0.01 g/L of natural seawater. D. vulgaris was cultured in a modified Postgate's medium with the composition of K2HPO4 0.65 g, NH4Cl 1.00 g, CaCl2 0.10 g, Na2SO4 3 g, yeast extract 1.00 g, and sodium lactate 4 mL in 1-L natural seawater. Resazurin was added as an anaerobic indicator before sterilization, and L-cysteine was adopted as an oxygen scavenger (Li et al., 2018).
Different amounts of NaNO3 were added into the modified Postgate's medium to give concentrations of 0, 0.5, 5, and 50 g/L. P. aeruginosa and D. vulgaris seed cultures with a volume of 4 mL in the logarithmic growth phase were inoculated after the autoclaved Postgate's media became oxygen-free in an anaerobic box (COY-8300600, COY Laboratory Product Inc.). All systems were operated in batch mode without the addition of nutrients over a 7-d experimental period, and were kept in the anaerobic box. The experimental procedure is shown in Fig. 1.
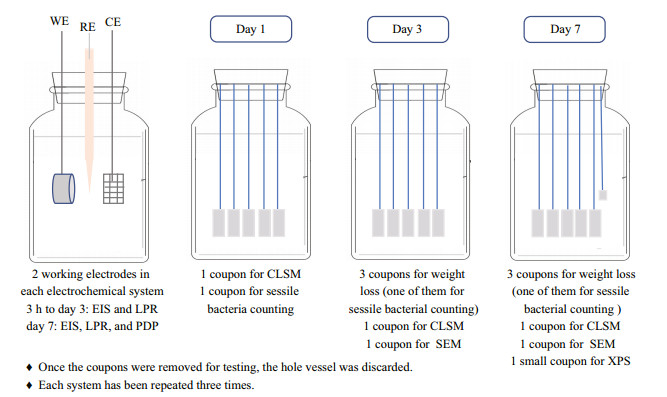
|
| Fig.1 The experimental procedure EIS: electrochemical impedance spectroscopy; LPR: linear polarization resistance; PDP: potentiodynamic polarization; CLSM: confocal laser scanning microscope; SEM: scanning electron microscopy. |
After immersion in different systems for 3 and 7 d, the coupons were taken out. Corrosion products were removed by Clark's solution (ASTM g1-03) in the ultrasonic cleaner for 20–40 s. Then they were washed with distilled water and absolute ethanol, dried with N2, and weighed. Corrosion rates were calculated by dividing weight loss by surface area. Three replicates were conducted for each system.
2.3.2 Electrochemical experimentsAn electrochemical workstation (Gamry 3000) was used to test the electrochemical corrosion behavior of coupons. A three-electrode system was used, where the EH40 steel specimen, a platinum mesh, and Ag/AgCl (KCl-sat) were used as working, counter, and reference electrodes, respectively. After open circuit potential (OCP) stabilized, the electrochemical impedance spectroscopy (EIS) was measured in the frequency range of 100 kHz to 10 MHz, applying a sinusoidal voltage signal of 10 mV, and the data were fitted with Zsimpwin software. Linear polarization resistance (LPR) measurements were carried out within the potential range of ±10 mV vs. OCP with the scan rate of 0.167 mV/s. In the range of -600 mV to +600 mV vs. OCP, the potentiodynamic polarization (PDP) curves were recorded at a potential sweep rate of 1 mV/s.
2.4 Biofilm observationA Live/Dead™ BacLight™ Bacterial Viability Kit (Molecular Probes, Texas, USA) was used to stain biofilms on coupons. The coupons immersed in different systems for 1, 3, and 7 d were taken out, rinsed gently with phosphate buffered saline (PBS) solution, and stained under dark conditions for 15 min. Coupons were observed under a confocal laser scanning microscope (CLSM, CZ microimaging GmbH). Biofilm thickness was measured using the 3D model.
2.5 Corrosion morphology and corrosion product characterizationsAfter immersed in different systems for 3 and 7 d, the coupons were taken out, and fixed with 2.5% glutaraldehyde for 1 h. Subsequently, they were dehydrated through a graded series of ethanol (30%, 50%, 70%, and 100%), and then dried and gold sprayed. The surface morphologies of the coupons before and after corrosion product removal were observed by scanning electron microscopy (SEM). The corrosion product compositions were measured with an XPS.
2.6 Environmental parameter measurements and cell countingA pH meter (PHS-3C) was used to measure the pH values of different solutions after 7 d. And the concentrations of HS–, SO42ˉ, NO3ˉ, and NO2ˉ ions in different systems were measured by ion chromatography. The number of sessile and planktonic cells of P. aeruginosa were determined on day 1, 3, and 7 using the plate count method with 2216E solid medium under aerobic conditions.
3 RESULT 3.1 Weight loss analysisFigure 2 displays corrosion weight losses (Fig. 2a) and rates (Fig. 2b) of EH40 steel in biotic systems with different concentrations of NaNO3 added. In the presence of mixed bacteria, the weight loss increased with NaNO3 concentrations added in the range of 0 to 5 g/L, and decreased from 5 to 50 g/L. The highest weight loss was achieved with an added NaNO3 concentration of 5 g/L. Corrosion rates of EH40 steel on day 3 were much larger than those on day 7 in biotic systems with NaNO3 concentrations added from 0 to 5 g/L, indicating that steel corroded faster at the early stage in these systems. In the biotic system with 50-g/L NaNO3 added, the corrosion rates on day 3 and 7 were similar, and EH40 steel corroded steadily. These corrosion discrepancies in different biotic systems might be relevant to the distinct growth status of bacteria, which will be evidenced below.
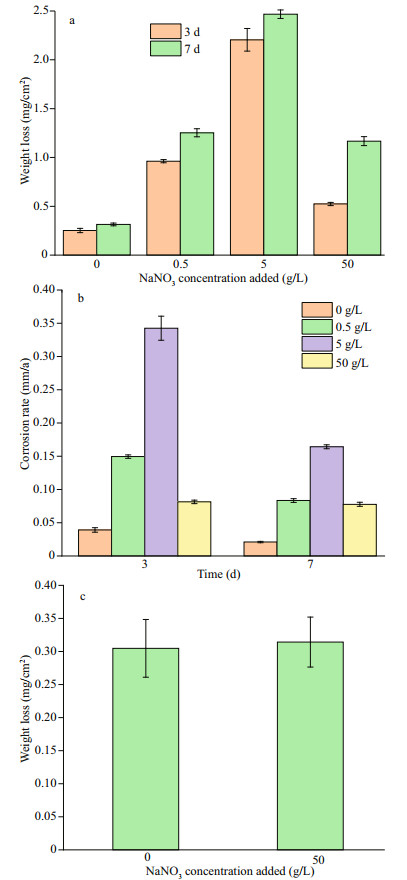
|
| Fig.2 Corrosion weight loss (a) and rate (b) of EH40 steel after 3 and 7 d of immersion in biotic systems with different concentrations of NaNO3; comparison of weight loss after 7-d immersion in sterile systems with and without 50-g/L NaNO3 added (c) |
In abiotic systems, the weight loss of EH40 steel was around 0.3 mg/cm2 no matter whether 50-g/L NaNO3 was added or not (Fig. 2c), and NaNO3 addition has no influence on corrosion under sterile conditions. Consequently, the impact of nitrate on EH40 steel corrosion was achieved by the action of bacteria.
3.2 Electrochemical characterizationsFigure 3 shows the Nyquist and Bode plots of EH40 steel in different biotic systems after immersion for different times. After 3 h of immersion (Fig. 3a, a'), all coupons shared similar plots, indicating that the initial surface conditions of all coupons were comparable. The diameters of capacitive loops in Nyquist plots of coupons exposed to the system without NaNO3 addition were stable in the initial stage and increased sharply from day 3 to 7 because of the accumulation of corrosion products. In the systems added with 0.5- and 5-g/L NaNO3, the diameter increased on the first day, and then decreased sharply. Meanwhile, the decrease of diameters in the system added with 5-g/L NaNO3 was more pronounced than that with 0.5-g/L NaNO3 added. When the medium was added with 50-g/L NaNO3, the diameters changed slightly in the whole experimental period. Before day 1 impedance modulus plots were close to each other in different systems, but distinct differences were observed on day 3. And coupons exposed to the system added with 5-g/L NaNO3 possessed the smallest values, indicating its worst corrosion resistance. Similarly, phase angle plots appeared divided into 3 groups from day 3. The plots from systems with 0.5- and 5-g/L NaNO3 added were similar, which were different from those recorded in the other 2 systems. The phase angle plots exhibited 2 peaks in the system added with 5-g/L NaNO3 on day 7, and the others gave one broad peak. Consequently, equivalent electrical circuit with 2 time constants shown in Fig. 4 was utilized to fit EIS data.
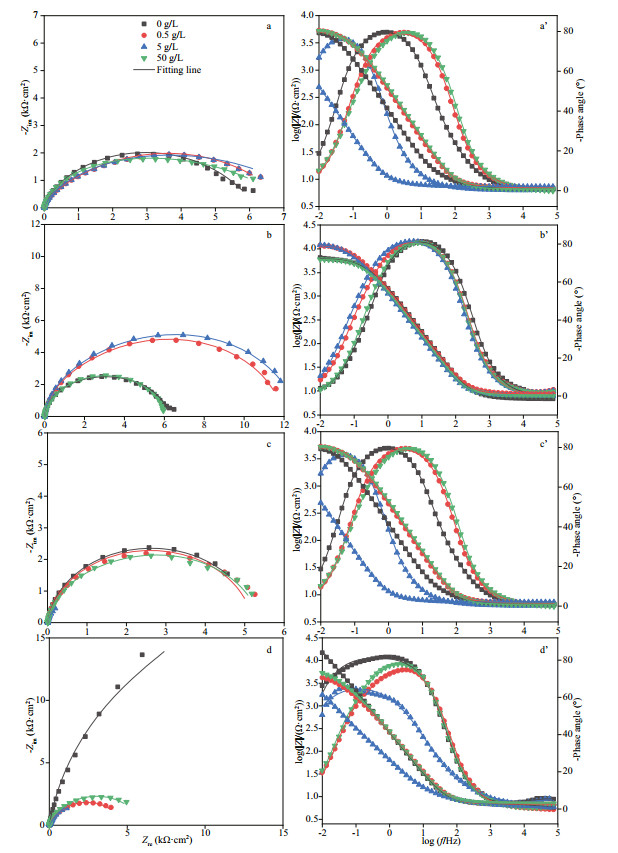
|
| Fig.3 Nyquist (a–d) and Bode plots (a'–d') of EH40 steel after immersed in biotic systems with different concentrations of NaNO3 added for different times a, a': 3 h; b, b': 1 d; c, c': 3 d; d, d': 7 d. |
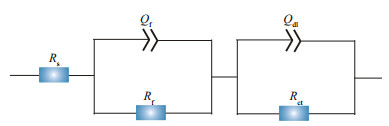
|
| Fig.4 The equivalent electrical circuit used for fitting EIS spectra Rp means linear polarization resistance; Ecorr means corrosion potential. |
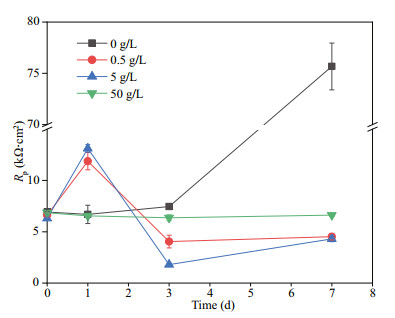
Rs, Rf, and Rct in the equivalent electrical circuit stood for the solution resistance, the resistance of the films, and the charge transfer resistance of the double-charge layers, respectively. The films were composed of corrosion products and biofilms. And capacitances of the surface films and electric double layers were denoted as Qf and Qdl, respectively. Variations of Rct and Rf with time are shown in Fig. 5. In all systems, the values of Rf were always smaller than those of Rct, demonstrating that corrosion was mainly controlled by charge transfer. Therefore, the corrosion rate was inversely proportional to Rct. When NaNO3 was not added, Rf fluctuated slightly before day 3, and then increased sharply, which might be due to the accumulation of corrosion products and biofilms shown below. There was an obvious increase in Rf on day 1 in systems added with 0.5- and 5-g/L NaNO3, and then it decreased. This could be associated with fast biofilm formation on day 1 and its later decay shown below. If 50-g/L NaNO3 was added, Rf increased slightly with time due to slow accumulation of corrosion products and biofilms. The time dependence of Rct was similar to that of Rf in all systems except the one added with 50-g/L NaNO3. Rct in systems added with different concentrations of NaNO3 increase in the order of 5 < 0.5 < 50 < 0 g/L on day 7, which was in agreement with weight loss results. Corrosion promotion by NaNO3 addition in the presence of D. vulgaris and P. aeruginosa mixed bacteria was clarified again, and the largest corrosion acceleration was achieved by NaNO3 addition with 5 g/L. Figure 6 displays time dependence of Rp in different systems. Rp variations were similar to those of Rct, and the smallest corrosion rate was recorded in the system without nitration addition.

|
| Fig.5 Time dependence of Rf (a) and Rct (b) in biotic systems with different concentrations of NaNO3 added |
|
| Fig.6 Time dependence of Rp in biotic systems with different concentrations of NaNO3 added |
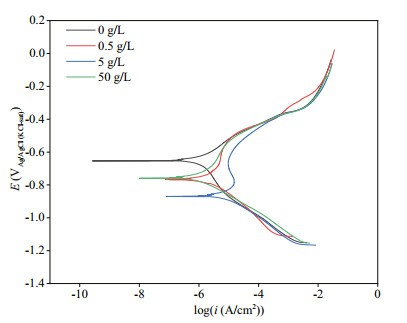
Figure 7 illustrates PDP curves of the coupons at the end of 7 d of immersion. It was obvious that Ecorr shifted negatively with the addition of NaNO3 (Fig. 7; Table 1), and the value in the system added with 5-g/L NaNO3 was the most negative. Both the acceleration in anodic reactions and inhibition in cathodic ones could result in negative shift of Ecorr, and depolarization of anodic reactions were observed and responsible for the Ecorr shift. A high corrosion current density (icorr) indicates a larger corrosion rate, and it could be concluded that coupons corroded at the highest rate when 5-g/L NaNO3 added owing to the largest icorr. Meanwhile, icorr was the smallest in the system without NaNO3 addition, and consequently coupons possessed the lowest corrosion rate. Furthermore, the lack of an evident passivity branch in the anodic curve in the system without NaNO3 addition demonstrated that the surface was free of passivation. But passivity appeared around -0.65 V vs. Ag/AgCl reference electrode with the addition of NaNO3 at 5-g/L. This passivation by NaNO3 addition might be associated with the accumulation of sessile P. aeruginosa corrosion products.
|
| Fig.7 PDP curves recorded after 7 d of immersion in biotic systems added with NaNO3 in different concentrations |
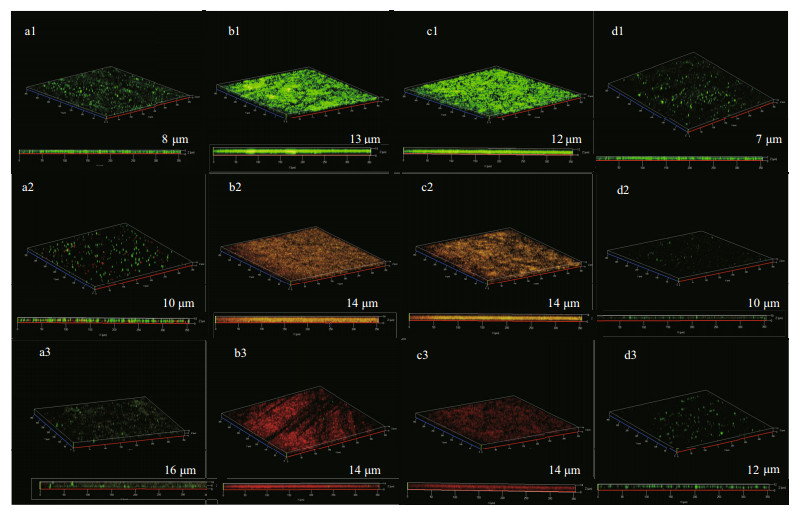
CLSM images of EH40 steel in biotic systems after different times of immersion are described in Fig. 8. Biofilm development varied in systems added with NaNO3 in different concentrations. On the first day, bacteria grew rapidly and formed a dense biofilm on the surface of the coupons in the systems with 0.5- and 5-g/L NaNO3 added. After 3 d of immersion, about half of bacteria in biofilms were live, and then most died at day 7. The decline of bacteria was caused by the depletion of nutrients. Compared with the former two systems, the number of bacteria was smaller in the system without NaNO3 addition, but most were live. Because there was no extra addition, NaNO3 was limited. D. vulgaris was expected to dominate, and it could sustain for a longer time due to its slower metabolic rate than P. aeruginosa. Besides, there were the least bacteria on coupons in the system added with 50-g/L NaNO3, suggesting that high concentration of nitrate hindered the growth of bacteria. But almost sessile bacteria were alive in this system, maybe determining the steadily increased weight loss.
|
| Fig.8 The CLSM images of EH40 steel obtained after 1 d (a1–d1), 3 d (a2–d2), and 7 d (a3–d3) of immersion in biotic systems added with NaNO3 in different concentrations a1–a3: 0 g/L; b1–b3: 0.5 g/L; c1–c3: 5 g/L; d1–d3: 50 g/L. |
Figure 9 displays the morphology of EH40 steel with/without corrosion product removal after 7 d of immersion in biotic systems with different concentrations of NaNO3 added. When the concentrations of NaNO3 added were 0.5 and 5 g/L, EH40 steel coupons were covered by a layer of corrosion products with more bacteria and extracellular polymeric substance (EPS). While in the other 2 systems, the corrosion product layer held fewer bacteria and EPS. After removing corrosion products, polishing scratches were still visible on coupons exposed to the system without NaNO3 addition, and consequently corrosion was slight. The addition of NaNO3 accelerated the corrosion of EH40 steel. When the added NaNO3 concentration was 5 g/L, EH40 steel suffered from most serious corrosion.

|
| Fig.9 The SEM images of EH40 steel without (a–d) and with (a'–d') corrosion product removal after 7 d of immersion in biotic systems added with NaNO3 in different concentrations a, a': 0 g/L; b, b': 0.5 g/L; c, c': 5 g/L; d, d': 50 g/L. |
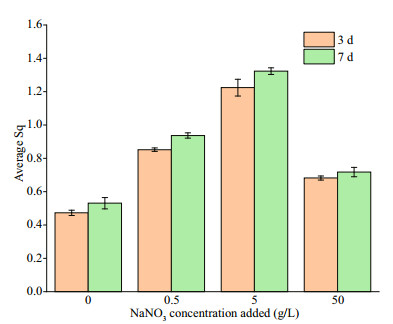
Surface roughness was further measured to check corrosion differences among different systems, and the result are shown in Fig. 10. The surface roughness increased with time, and the surface roughness of coupons increased in the order of 0- < 50- < 0.5- < 5-g/L NaNO3 added no matter on the 3th or 7th day. When the NaNO3 concentration added was 5 g/L, the surface roughness was the largest (1.323), followed by 0.5 g/L. Evident rougher surfaces were observed when NaNO3 was added, indicating acceleration in corrosion again.
|
| Fig.10 The surface roughness of EH40 steel after 3 and 7 d of immersion in biotic systems added with NaNO3 in different concentrations |
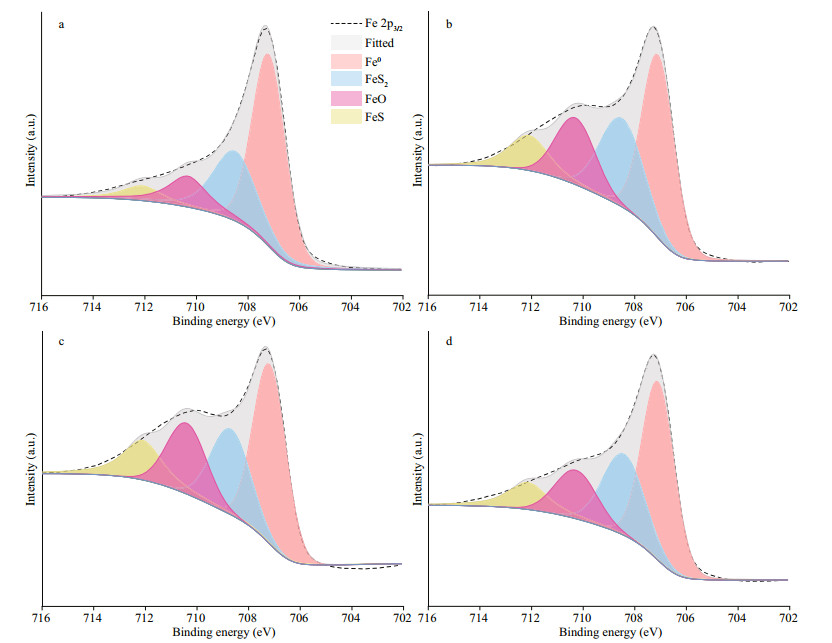
The Fe 2p3/2 spectra of the EH40 steel after immersed in biotic systems for 7 d are depicted in Fig. 11. The components of products in different systems were identical, all consisted of FeS2, FeO, and FeS, and their peaks were located at around 708.5, 710.3, and 712.1 eV, respectively (Binder, 1973; Allen et al., 1974; Best et al., 1977; Hawn and Dekoven, 1987). The growth differences of two species in different systems did not affect the composition of corrosion products due to the fact that metabolites of P. aeruginosa could not form depositions with Fe.
|
| Fig.11 The Fe 2p3/2 spectra of corrosion products on EH40 steel after 7 d of immersion in biotic systems added with NaNO3 in different concentrations a: 0 g/L; b: 0.5 g/L; c: 5 g/L; d: 50 g/L. |
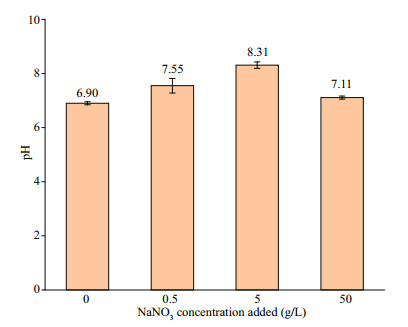
After 7 d of incubation, pH values of the biotic systems with the addition of different NaNO3 concentrations are shown in Fig. 12. When NaNO3 in concentration of 5 g/L was added, pH was the highest (8.39), and followed by 0.5 g/L (7.74). In the systems without NaNO3 added and with the addition at 50 g/L, the change of pH was rather slight compared with the initial pH (7.10). The high pH in systems with 0.5- and 5-g/L NaNO3 indicated that P. aeruginosa was stimulated, and it grew better in the system with 5-g/L NaNO3 added. Furthermore, the promotion of nitrate to corrosion was not achieved by organic acids.
|
| Fig.12 The pH in biotic systems added with NaNO3 in different concentrations on day 7 |
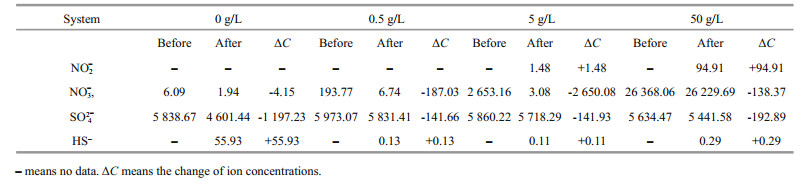
Table 2 displays the concentrations of HS–, SO42ˉ, NO3ˉ, and NO2ˉ in different systems before and after 7 d of culture. In the system without NaNO3 added, a large amount of SO42ˉ (1 197×10-6) was consumed and HS– (56×10-6) was produced. Instead, only 4×10-6 NO3ˉ was utilized. The result indicated that D. vulgaris played a dominant role in the system without nitrate added, and it reduced sulfate to sulfide by metabolism. In the systems with 0.5- and 5-g/L NaNO3 added, less SO42ˉ was consumed and almost no HS– was detected after 7 d of culture, suggesting that quite few D. vulgaris grew in these systems. While there was almost no residual NO3ˉ, and 96%–99% of NO3ˉ was consumed. Because of the addition of NaNO3, P. aeruginosa was dominant by utilizing NO3ˉ as the electron acceptor for metabolism. Besides, there was almost no NO2ˉ produced, which might be due to the strong metabolic activity and direct reduction of NO3ˉ to NH4+ of P. aeruginosa under optimum conditions.
|
Around 95×10-6 NO2ˉ was accumulated in the system with 50-g/L NaNO3 added, which might be due to the decline of bacterial metabolic activity by high nitration concentration. Rodríguez-Gómez et al. (2005) discovered that the denitrification was incomplete when the high nitrate concentration of nitrate was provided. Compared with the other two systems with the addition of 0.5- and 5-g/L NaNO3, NO3ˉ consumption was inhibited sharply by the addition of 50-g/L NaNO3.
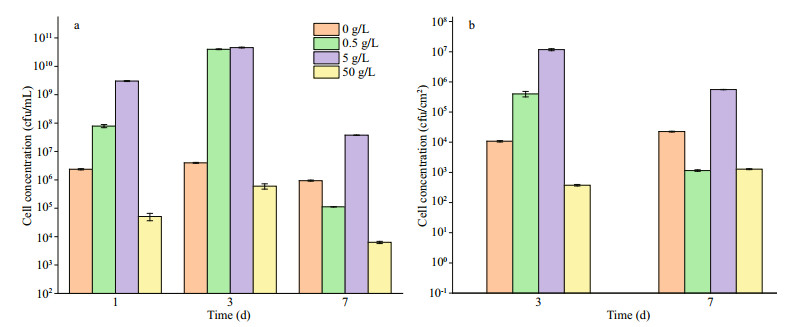
3.6.3 Quantification of P. aeruginosaThe planktonic and sessile cell numbers of P. aeruginosa in systems with different NaNO3 concentrations added are displayed in Fig. 13. The quantity of planktonic cells was close to the initial inoculation (ca. 106 cfu/mL) in the whole period when NaNO3 was not added, indicating propagation was limited due to the lack of electron acceptors for P. aeruginosa. The sessile quantity was around 104 cfu/cm2 on days 3 and 7. In the systems with 0.5- and 5-g/L NaNO3 added, P. aeruginosa reproduced rapidly in the first 3 days due to substantial electron acceptors available, and more than 1010 cfu/mL cells were recorded on day 3. There was a sharp decrease of the planktonic quantity on day 7, which could be associated with the depletion of NO3ˉ (Table 2). Similarly, the numbers of sessile P. aeruginosa cells decreased significantly from day 3 to day 7. Compared with the system without NaNO3 addition, the numbers of sessile P. aeruginosa cells increased by 1.5 and 4 orders of magnitude in systems added with 0.5- and 5-g/L NaNO3 on day 3, respectively, but it decreased by 1 order of magnitude in the system with 0.5-g/L NaNO3 added on day 7. The numbers of planktonic and sessile P. aeruginosa cells in the system added with 5-g/L NaNO3 were more than 1 order of magnitude higher than those in the system without NaNO3 addition on day 7. The quantity of both planktonic and sessile P. aeruginosa cells was the smallest in the system added with 50-g/L NaNO3. Corrosion rate is not always linear with sessile bacterial number, and corrosion activity of cells play a key role. Jia et al. (2017) reported that the number of sessile P. aeruginosa decreased under carbon starvation, but the weight loss increased. The quantity of planktonic cells was close to the initial inoculation in the whole period when NaNO3 was not added, indicating propagation was limited due to the lack of electron acceptors for P. aeruginosa. In the system added with 50-g/L NaNO3, the number of sessile P. aeruginosa was smaller than that in the system without NaNO3 addition due to high osmotic pressure. But the bacteria tended to aggregate (Fig. 9). It was difficult for bacteria at the bottom of aggregates to obtain organics from solution, so they were expected to attack iron to obtain electrons. In addition, the presence of nitrite might affect corrosion, and it was found that corrosion rate increased from 0.02 mm/a in the system without NO2ˉ addition to 0.06 and 0.1 mm/a when 0.07- and 0.14-g/L NaNO2 was added (Fig. 14). However, the specific mechanism was not clear.
|
| Fig.13 The planktonic (a) and sessile (b) cell numbers of P. aeruginosa in systems added with NaNO3 in different concentrations |
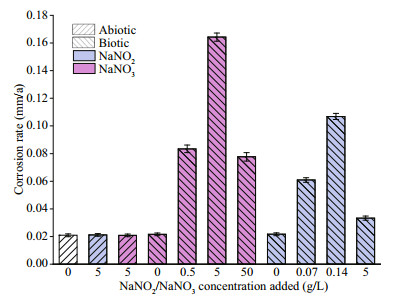
|
| Fig.14 Corrosion rate of EH40 steel immersed in different systems for 7 d |
The mixed bacteria had little effect on EH40 steel corrosion in the systems without NaNO3 addition. After 7 d of immersion, the weight loss in biotic media was close to that in the sterile media (Fig. 2). According to the results of ion concentrations (Table 2), D. vulgaris dominated in the mixed bacteria. Nutrients were rich in the present media, and biofilms were relatively thin (Fig. 8). Consequently, EH40 steel corrosion acceleration by EET under carbon starvation did not play the leading role, which was in good agreement with our previous work on Q235 carbon steel corrosion affect by Desulfovibrio sp. Huiquan2017 (Li et al., 2021). On the other hand, corrosion by D. vulgaris might be hampered by P. aeruginosa. Liu and Cheng (2020) claimed that the presence of P. aeruginosa could promote D. vulgaris cells to be surrounded by corrosion products, leading to weaker corrosion than the system with D. vulgaris only.
EH40 steel corrosion was promoted with nitrate addition in the presence of mixed bacteria, but no corrosion differences were observed in sterile systems with and without the addition of 50-g/L NaNO3. Subsequently, the corrosion acceleration of nitrate addition was achieved by bacteria. Meanwhile, corrosion promotion was dependent on the addition of nitrate concentrations, which increased from 0 to 5 g/L and decreased from 5 to 50 g/L. However, Schwermer et al. (2008) discovered that the addition of 1-mmol/L NO3ˉ mitigated the uniform corrosion in PWRI systems. This difference was believed to be closely associated with different states of bacterial growth and biofilms in different systems. In the systems with 0.5- and 5-g/L NaNO3 added, bacteria grew rapidly and formed dense biofilms on the coupons in the early state (Fig. 8), which resulted in increased Rf values and protected EH40 steel. Moreover, dense biofilms hindered the electron transfer and the values of Rct increased in the early stage of the experiment. Furthermore, EET did not play a major role due to abundant nutrients and the decreased corrosion rate. In the later stage of the trial, bacteria decayed rapidly due to the depletion of organic matter, and the protective effect of biofilms was weakened. These led to decreased Rf and Rct. Bacteria began to attack EH40 steel in the bottom of biofilms to obtain electrons, and EET played a key role. According to the results of ion concentration, the addition of nitrate promoted the growth of P. aeruginosa, which utilized NO3ˉ as electron acceptor. The voltage difference between NO3ˉ/NH4+(Eh pH=7.0=+358 mV vs. SCE) and Fe/Fe2+ (Eh pH=7.0=-447 mV vs. SCE) was larger than that between SO42ˉ/HS– (Eh pH=7.0=-217 mV vs. SCE) and Fe/Fe2+, and more energy was obtained from NO3ˉ reduction (Eckford and Fedorak, 2002; Jia et al., 2017; Gu et al., 2021). This proved thermodynamic feasibility. Accordingly, high corrosion activity of P. aeruginosa was expected from the perspective of energetics. Meanwhile, compared with the system added with 0.5-g/L NaNO3, the one added with 5 g/L supported more sessile P. aeruginosa cells in the later stage of the experiment (Fig. 13), and the corrosion promotion was stronger. Nevertheless, despite the favorable thermodynamics, corrosion rates are determined by kinetic limits. Hence, taking into account the complexity of the long-term denitrification process, the central role of the biofilm's catalytic activity was crucial (Jia et al., 2017).
In the system with 50-g/L NaNO3 added, the growth of both bacteria was inhibited, and the bacterial activity decreased due to high osmotic pressure. The high concentration of nitrate influenced the denitrification and around 100×10-6 NO2ˉ was detected after 7 d of inoculation. The impact of NO2ˉ intermediate on corrosion has been investigated, and the result is shown in Fig. 14. Corrosion rate increased from 0.02 mm/a in the system without NO2ˉ addition to 0.06 and 0.1 mm/a when 0.07- and 0.14-g/L NaNO2 was added, and then decreased to 0.03 mm/a with the addition of 5-g/L NaNO2. The acceleration in corrosion rate by NaNO2 addition with low concentrations might be assowith additional electron acceptors provided by NO2ˉ and other unknown mechanisms. Moreover, the decrease by the high concentration could be due to its toxicity (Hubert et al., 2005). Lee et al. (2012) found that the addition of 200×10-6 nitrite decreased the corrosion rate of a carbon steel pipeline in deoxidized synthetic tap water by forming a passive film which was composed mainly of maghemite (γ-Fe2O3). But passivation effect of NaNO2 added with 5 g/L was not observed in abiotic media in the present work, which might be due to the difference in media.
The inhibition of nitrate addition on SRB growth and metabolism is in good agreement with previous reports. For example, Kebbouche-Gana and Gana (2012) discovered that the total amount of sulfide decreased in static tests with the SRB consortium obtained from the In Amenas injected water, when nitrate was dosed at 120 mg/L. In addition, Einarsen et al. (2000) discovered that nitrate prevented the formation of H2S and other odorous compounds. However, the inhibition on SRB did not inhibit corrosion in the present study, on the contrary, it promoted corrosion of EH40 steel. This was due to the stimulation on NRB with high corrosive activity. Therefore, care must be taken to BCE method with nitrate when NRB with high corrosive activity were present in the environments.
5 CONCLUSIONThis study identified that only mixed bacteria of D. vulgaris and P. aeruginosa or nitrate had little effect on EH40 steel corrosion, but nitrate could accelerate the corrosion of EH40 steel through the action of microorganisms. D. vulgaris was dominant without nitrate added, and its effect on EH40 steel by EET was weak in the case of sufficient nutrients. The corrosion promotion of nitrate addition in the presence of D. vulgaris and P. aeruginosa was dependent on its concentrations, and it increased from 0 to 5 g/L and decreased from 5 to 50 g/L. These differences might be attributed to the regulation of nitrate in the growth of bacteria and biofilms. NaNO3 added with 0.5 and 5 g/L promoted the growth of P. aeruginosa with high corrosive activity, and accordingly accelerated corrosion. Meanwhile, 5-g/L NaNO3 supported more sessile P. aeruginosa cells than 0.5 g/L in the later stage, and severer corrosion was achieved by EET. Further increase in NaNO3 concentration added to 50-g/L weakened corrosion promotion of EH40 steel due to low bacterial quantity and activity by high osmotic pressure. Although nitrate is commonly used as a corrosion inhibitor in PWRI system, it induces more serious corrosion when NRB with high corrosive activity are present in the environments. Therefore, sodium nitrate should be classified as a dangerous inhibitor and carefully adopted according to the specific environments.
6 DATA AVAILABILITY STATEMENTThe datasets generated during the current study are available from the corresponding author on reasonable request.
Allen G C, Curtis M T, Hooper A J, Tucker P M. 1974. X-ray photoelectron spectroscopy of iron-oxygen systems. Dalton Transactions, (14): 1525-1530.
|
Batmanghelich F, Li L, Seo Y. 2017. Influence of multispecies biofilms of Pseudomonas aeruginosa and Desulfovibrio vulgaris on the corrosion of cast iron. Corrosion Science, 121: 94-104.
DOI:10.1016/j.corsci.2017.03.008 |
Best S A, Brant P, Feltham R D, Rauchfuss T B, Roundhill D M, Walton R A. 1977. X-Ray photoelectron spectra of inorganic molecules. 18. Observations on sulfur 2P binding energies in transition metal complexes of sulfur-containing ligands. Inorganic Chemistry, 16(8): 1976-1979.
DOI:10.1021/ic50174a030 |
Binder H. 1973. Investigations on nature of chemical bonds in iron-sulfur compounds using X-ray photoelectron spectroscopy. Zeitschrift Fur Naturforschung Section B-A Journal of Chemical Sciences B, 28(5-6): 255-262.
|
Chen C, Wu J J, Zhang D. 2018. Effects of sulfate-reducing bacteria on marine corrosion of weld joints of EH40. Equipment Environmental Engineering, 15(10): 51-59.
(in Chinese with English abstract) |
Chen H Y, Wang W D, Du C A, Wang X, Zhang X X. 2013. Study on the microbiological corrosion in oilfield reinjection water system inhibited by biological competition technology. Industrial Water Treatment, 33(6): 79-81.
(in Chinese with English abstract) |
Costerton J W. 2007. The predominance of biofilms in natural and engineered ecosystems. In: Costerton J W ed. The Biofilm Primer. Springer, Berlin Heidelberg. p. 5-13.
|
Eckford R E, Fedorak P M. 2002. Planktonic nitrate-reducing bacteria and sulfate-reducing bacteria in some western Canadian oil field waters. Journal of Industrial Microbiology and Biotechnology, 29(2): 83-92.
DOI:10.1038/sj.jim.7000274 |
Einarsen A M, Æsøy A, Rasmussen A I, Bungum S, Sveberg M. 2000. Biological prevention and removal of hydrogen sulphide in sludge at Lillehammer Wastewater Treatment Plant. Water Science & Technology, 41(6): 175-187.
|
Gao J Y, Wu J J, Zhang D. 2018. Impact of Pseudoalteromonas sp. on galvanic corrosion between B10 and EH40. Equipment Environmental Engineering, 15(10): 77-83.
(in Chinese with English abstract) |
Gu T Y, Jia R, Unsal T, Xu D K. 2019. Toward a better understanding of microbiologically influenced corrosion caused by sulfate reducing bacteria. Journal of Materials Science & Technology, 35(4): 631-636.
|
Gu T Y, Wang D, Lekbach Y, Xu D K. 2021. Extracellular electron transfer in microbial biocorrosion. Current Opinion in Electrochemistry, 29: 100763.
DOI:10.1016/j.coelec.2021.100763 |
Hawn D D, Dekoven B M. 1987. Deconvolution as a correction for photoelectron inelastic energy losses in the core level XPS spectra of iron oxides. Surface and Interface Analysis, 10(2-3): 63-74.
DOI:10.1002/sia.740100203 |
Hubert C, Nemati M, Jenneman G, Voordouw G. 2005. Corrosion risk associated with microbial souring control using nitrate or nitrite. Applied Microbiology and Biotechnology, 68(2): 272-282.
DOI:10.1007/s00253-005-1897-2 |
Jia R, Tan J L, Jin P, Blackwood D J, Xu D K, Gu T Y. 2018. Effects of biogenic H2S on the microbiologically influenced corrosion of C1018 carbon steel by sulfate reducing Desulfovibrio vulgaris biofilm. Corrosion Science, 130: 1-11.
DOI:10.1016/j.corsci.2017.10.023 |
Jia R, Yang D Q, Xu J, Xu D K, Gu T Y. 2017. Microbiologically influenced corrosion of C1018 carbon steel by nitrate reducing Pseudomonas aeruginosa biofilm under organic carbon starvation. Corrosion Science, 127: 1-9.
DOI:10.1016/j.corsci.2017.08.007 |
Kebbouche-Gana S, Gana M L. 2012. Biocorrosion of carbon steel by a nitrate-utilizing consortium of sulfate-reducing bacteria obtained from an Algerian oil field. Annals of Microbiology, 62(1): 203-210.
DOI:10.1007/s13213-011-0247-0 |
Lahme S, Enning D, Callbeck C M, Vega D M, Curtis T P, Head I M, Hubert C R J. 2019. Metabolites of an oil field sulfide-oxidizing, nitrate-reducing Sulfurimonas sp. cause severe corrosion. Applied and Environmental Microbiology, 85(3): e01891-18.
|
Lai R Q, Li Q, Cheng C K, Shen H, Liu S Q, Luo Y J, Zhang Z Z, Sun S S. 2020. Bio-competitive exclusion of sulfate-reducing bacteria and its anticorrosion property. Journal of Petroleum Science and Engineering, 194: 107480.
DOI:10.1016/j.petrol.2020.107480 |
Lee D Y, Kim W C, Kim J G. 2012. Effect of nitrite concentration on the corrosion behaviour of carbon steel pipelines in synthetic tap water. Corrosion Science, 64: 105-114.
DOI:10.1016/j.corsci.2012.07.005 |
Li E E, Wu J J, Zhang D, Sun Y, Chen J N. 2018. D-phenylalanine inhibits the corrosion of Q235 carbon steel caused by Desulfovibrio sp. International Biodeterioration & Biodegradation, 127: 178-184.
|
Li E E, Wu J J, Zhang D, Wang P, Wang Y, Xu M, Li C, Sun Z H, Zhu L Y. 2021. Electron donor dependent inhibition mechanisms of D-phenylalanine on corrosion of Q235 carbon steel caused by Desulfovibrio sp. Huiquan2017. Corrosion Science, 188: 109493.
DOI:10.1016/j.corsci.2021.109493 |
Liu H W, Cheng Y F. 2020. Corrosion of X52 pipeline steel in a simulated soil solution with coexistence of Desulfovibrio desulfuricans and Pseudomonas aeruginosa bacteria. Corrosion Science, 173: 108753.
DOI:10.1016/j.corsci.2020.108753 |
Nemati M, Jenneman G E, Voordouw G. 2001. Impact of nitrate-mediated microbial control of souring in oil reservoirs on the extent of corrosion. Biotechnology Progress, 17(5): 852-859.
DOI:10.1021/bp010084v |
Oguzie E E, Oguzie K L, Akalezi C O, Udeze I O, Ogbulie J N, Njoku V O. 2013. Natural products for materials protection: corrosion and microbial growth inhibition using Capsicum frutescens biomass extracts. ACS Sustainable Chemistry & Engineering, 1(2): 214-225.
|
Pillay C, Lin J. 2014. The impact of additional nitrates in mild steel corrosion in a seawater/sediment system. Corrosion Science, 80: 416-426.
DOI:10.1016/j.corsci.2013.11.047 |
Reinsel M A, Sears J T, Stewart P S, McInerney M J. 1996. Control of microbial souring by nitrate, nitrite or glutaraldehyde injection in a sandstone column. Journal of Industrial Microbiology and Biotechnology, 17(2): 128-136.
DOI:10.1007/BF01570056 |
Rodríguez-Gómez L E, Delgado S, álvarez M, Elmaleh S. 2005. Inhibition of sulfide generation in a reclaimed wastewater pipe by nitrate dosage and denitrification kinetics. Water Environment Research, 77(2): 193-198.
DOI:10.2175/106143005X41762 |
Schwermer C U, Lavik G, Abed R M M, Dunsmore B, Ferdelman T G, Stoodley P, Gieseke A, de Beer D. 2008. Impact of nitrate on the structure and function of bacterial biofilm communities in pipelines used for injection of seawater into oil fields. Applied and Environmental Microbiology, 74(9): 2841-2851.
DOI:10.1128/AEM.02027-07 |
Shartau S L C, Yurkiw M, Lin S P, Grigoryan A A, Lambo A, Park H S, Lomans B P, van der Biezen E, Jetten M S M, Voordouw G. 2010. Ammonium concentrations in produced waters from a mesothermic oil field subjected to nitrate injection decrease through formation of denitrifying biomass and anammox activity. Applied and Environmental Microbiology, 76(15): 4977-4987.
DOI:10.1128/AEM.00596-10 |
Wang Y, Wu J J, Sun L P, Zhang D, Li E E, Xu M, Cai H Y. 2021. Corrosion of EH40 steel affected by Halomonas titanicae dependent on electron acceptors utilized. Corrosion Science, 182: 109263.
DOI:10.1016/j.corsci.2021.109263 |
Zuo R, Örnek D, Syrett B C, Green R M, Hsu C H, Mansfeld F B, Wood T K. 2004. Inhibiting mild steel corrosion from sulfate-reducing bacteria using antimicrobial-producing biofilms in Three-Mile-Island process water. Applied Microbiology and Biotechnology, 64(2): 275-283.
DOI:10.1007/s00253-003-1403-7 |
 2022, Vol. 40
2022, Vol. 40



