Institute of Oceanology, Chinese Academy of Sciences
Article Information
- LI Xiaomin, XING Rong'e, XU Chaojie, LIU Song, QIN Yukun, LI Kecheng, YU Huahua, LI Pengcheng
- Immunostimulatory effect of quaternary degree and acetyl group of quaternized chitosan on macrophages RAW 264.7
- Journal of Oceanology and Limnology, 40(3): 1160-1170
- http://dx.doi.org/10.1007/s00343-021-1098-6
Article History
- Received Mar. 25, 2021
- accepted in principle May. 14, 2021
- accepted for publication Jun. 15, 2022
2 Laboratory for Marine Drugs and Bioproducts, Pilot National Laboratory for Marine Science and Technology (Qingdao), Qingdao 266237, China;
3 University of Chinese Academy of Sciences, Beijing 100049, China
Vaccination is the primary preventive treatment to prevent infections (Guimarães et al., 2015). With the promising effects it has achieved, the demand for vaccines in disease treatment has increased. However, the entire antigen used alone exhibits underestimated toxicity problems. While subunit vaccines have been developed with reduced toxicity, they sometimes have poor immunogenicity (Yüksel et al., 2020). Thus, the increasing demand for adjuvants is needed to help antigens improve the efficiency of immune responses and preserve antigen usage.
Adjuvants are defined as agents that are added to vaccines to induce enhanced immune responses (Petrovsky and Aguilar, 2004). Ideally, adjuvants should be nontoxic, inexpensive to produce and promote an appropriate immune response according to a specific antigen. There has been great progress in the development of adjuvants, but there are still some problems. Traditional oil emulsions can cause fever and granuloma (Aucouturier et al., 2001). The well-known alum adjuvant may induce weak cellular immunity and is difficult to degrade (Sun et al., 2018). Therefore, it is urgent to develop novel adjuvant materials that are characterized as safe, biodegradable, and effective stimuli in immunity.
Chitosan, a partial deacetylated product of chitin that is composed of glucosamine and N-acetylglucosamine units, has attracted wide attention in the pharmaceutical and immunization fields (Amidi and Hennink, 2010; Li et al., 2013). Surprisingly, chitosan possesses many ideal characteristics to perform as an adjuvant, such as being natural, biocompatible, biodegradable, and nonantigenic. The adjuvant properties of chitosan were proven as early as the 1980s (Suzuki et al., 1984). Furthermore, over 20 years ago, chitin derivatives, including chitosan, were found to enable potent immunological activities in activating macrophages and natural killer (NK) cells (Nishimura et al., 1984). Unfortunately, its poor water solubility has limited the application of chitosan in biomedical applications. The active sites on chitosan chains can be modified to various derivatives, such as carboxymethyl chitosan (Costain et al., 1997), quaternized chitosan (Thanou et al., 2002), sulfated chitosan (Dimassi et al., 2018). Among these modifications, quaternized chitosan displays superior water solubility over a broader spectrum of pH values, accompanied by brilliant biological activities. Guo et al. (2007) found that quaternized chitosan exhibits better antifungal activities than chitosan due to the introduction of quaternary ammonium groups. It has also been reported that N-2-hydroxypropyl trimethyl ammonium chloride chitosan can effectively provide potent immunization against porcine parvovirus (PPV) infection (Zhou et al., 2020). They also found that N-2-HACC showed higher immense potential for preventing Newcastle disease viruses (NDV) (Zhao et al., 2016). The obvious enhancements in activity produced by quaternary ammonium salts are due to the introduction of a positive charge bonded to quaternary ammonium groups. Obviously, the quaternary group plays a significant role in the activities of the quaternary ammonium salt (Hamman et al., 2002). However, few studies have reported the impact of quaternary ammonium levels on macrophages.
Macrophages, important members of the immune system involved in detection and phagocytosis, can also initiate inflammation by releasing active molecules. Therefore, the activation of macrophages is crucial in immune responses (Zhang et al., 2018). Activated macrophages have higher levels of nitrogen oxide (NO) and cytokines such as tumor necrosis factor (TNF)-α and interleukin (IL)-6. Yang et al. (2019) found that Hydroxypropyltrimethyl ammonium chloride chitosan (HACC) could effectively activate macrophages to trigger an immune response. In addition, N, N, N-trimethyl chitosan (TMC) was helpful to improve the immunogenicity of antigens on RAW264.7 cells (Liu et al., 2015).
The degree of quaternary ammonium does have an important impact on immunity (Kotzé et al., 1999a; Hamman et al., 2002). It was found that the quaternary ammonium degree was a significant factor influencing the adjuvant properties of HACC; and quaternized degrees of 41% and 60% were favorable to induce a higher immune response (Wang et al., 2016). The influences of the degree of quaternization of TMC were evaluated in Caco-2 cells to measure their permeability, and TMC-61% showed better absorption enhancement than TMC-12% (Kotzé et al., 1999a). However, the immunomodulatory effect of quaternized chitosan on RAW 264.7 macrophages has never been studied. In addition, the acetyl group is the most important activity index of chitosan. In this study, to determine the influence of the acetyl group on the quaternization of chitosan, we synthesized quaternary ammonium salts of chitosan and fully deacetylated chitosan quaternary ammonium salts with different degrees of substitution and studied their immunomodulatory activity.
2 MATERIAL AND METHOD 2.1 MaterialChitosan, 1 820 kDa in mass with an 86% deacetylation degree (DD), was obtained from Qingdao Yunzhou Biochemical Corp (Qingdao, China). Acetic acid, hydrochloric acid, epoxypropyl trimethyl ammonium chloride (GTMAC), sodium hydroxide and hydrogen peroxide (30%) were purchased from Sinopharm Chemical Reagent Co. Ltd. (Shanghai, China). Roswell Park Memorial Institute (RPMI) 1640 medium was obtained from HyClone (Logan, UT, USA), fetal bovine serum (FBS) was acquired from Gibco in Australia, (4, 5-dimethylthiazol-2-yl)-3, 5-diphenyltetrazolium bromide (MTT) was supplied by Apexbio (Houston, TX, USA), an E.Z.N.A. Total RNA Kit was supplied by OMEGA Biotechnology Company (USA), and a Prime Script RT reagent Kit (include gDNA Eraser) and a SYBR Premix Ex Taq II (Tli RNaseH Plus) were purchased from Takla in Japan.
2.2 Preparation of fully deacetylated chitosanFully deacetylated chitosan was prepared by the method of He et al. (2016). Briefly, 100-g chitosan and 200-g NaOH were dispersed in 800-mL deionized water and heated at 120 ℃ under high-pressure conditions. After 2 h, it was washed with water until it was neutralized and was then filtered by suction. Finally, the product was dried in oven at 60 ℃ and pulverized into a powder to obtain chitosan with 100% DD.
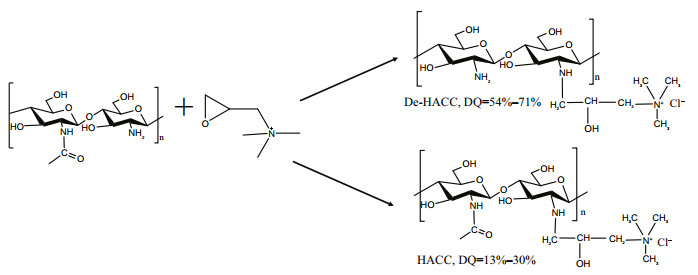
2.3 Preparation of HACC and De-HACC with different degrees of substitutionDifferent degrees of quaternization (DQ) HACCs and De-HACC were prepared using the modified method of Spinelli et al. (2004). Briefly, 5-g chitosan or fully deacetylated chitosan and a certain amount of glycidyl trimethylammonium chloride were added to 70-mL distilled water and then placed in a three-necked flask heated at 80 ℃. After 24 h, the reaction products were dialyzed on a 3 000-Da dialysis membrane for 72 h against distilled water, and finally, the product was concentrated and lyophilized to obtain quaternized chitosan or fully deacetylated chitosan with different degrees of substitution.
The ratio between chitosan and glycidyl trimethylammonium chloride was adjusted to obtain various quaternary degrees of chitosan or fully deacetylated chitosan quaternary ammonium salt. The synthetic route of the two samples is shown in Fig. 1.
|
| Fig.1 Synthetic schematic of HACC and De-HACC |
Fourier transform infrared (FTIR) spectra of chitosan, fully deacetylated chitosan, HACC and De-HACC with different degrees of substitution were obtained using a Thermo Scientific Nicolet iS10 FT-IR spectrometer with a scope of testing from 4 000 to 400 cm-1 and KBr discs.
Chitosan, fully deacetylated chitosan, HACC, and De-HACC with different degrees of substitution were dissolved in D2O to a concentration of 20 mg/mL and were then evaluated by a JEOL JNM-ECP600 spectrometer (JEOL Ltd., Tokyo, Japan) to record their hydrogen nuclear magnetic resonance (1HNMR) spectra.
2.5 Determination of the degree of quaternization (DQ)DQ was defined as the molar ratio of bonded GTMAC per mol of glucosamine, which was determined by the conductometric titration of introduced Cl–. Briefly, various HACCs and De-HACCs with different degrees of substitution (50 mg) were dissolved in ultrapure water (50 mL) to make a 1-mg/mL solution. Then, the above solution was conductometrically titrated with AgNO3 solution (0.01 mol/L) and monitored by a conductivity meter. The specific DQ was determined by the following formula:
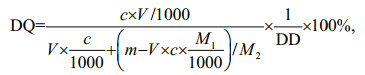
where V (mL) is the volume of AgNO3 solution used when the conductivity of the solution is the lowest; c (mol/L) is the concentration of AgNO3 solution; m (g) is the masses of HACC and De-HACC used with different degrees of substitution; DD is the deacetylated degree of chitosan; M1 (mol/g) is the molar mass of quaternized chitosan, and M2 (mol/g) is the molar mass of glucosamine.
2.6 Determination of solubilityA 0.5-g HACC sample was prepared into a supersaturated solution with water. Then, the solution was fully dissolved under stirring for 24 h. After that, the sample was centrifuged to remove the supernatant, and the undissolved sample was dried and weighed. The solubility was calculated by comparing the mass difference before and after dissolution.
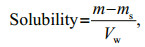
where m is the mass of quaternary chitosan, and ms is the mass of the undissolved sample after centrifugation and drying. Vw is the volume of distilled water.
2.7 Cell cultureRAW 264.7 macrophages were purchased from American Type Culture Collection (Manassas, VA, USA). RPMI 1640 supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin was used as the culture medium. The cells were incubated in a plastic culture bottle placed at 37 ℃ in an atmosphere of 5% CO2.
2.8 Cell viability testThe MTT method was used to determine the cytotoxicity of HACC and De-HACC with different degrees of substitution. Briefly, cells were adjusted to 105 cells/mL in a 96-well plate and preincubated for 24 h. Then, 100-μL aliquots of the samples at different concentrations were added. Then, the initial medium was replaced with 100-μL MTT solution (0.5 mg/ mL). Four hours later, 15-μL dimethyl sulfoxide (DMSO) was added to stop the MTT reaction. Immediately following this addition, the optical density (OD) OD550 was measured to determine the viability of cells using an Infinite M200 Pro spectrophotometer (Tecan, Männedorf, Switzerland).
2.9 Determination of NOThe expression of NO was indirectly reflected bymeasuring the production of nitrite (NO2-) in the culture medium using Griess reagent as reported by Baek et al. (2015). We used seven kinds of HACC and De-HACC with different degrees of substitution ranging from 7% to 71% (HACC 7%, HACC 13%, HACC 23%, HACC 30% and De-HACC 54%, De-HACC 60%, De-HACC 71%) in solution to clarify the NO expression activity. Briefly, RAW 264.7 cells were cultured in 96-well plates at a concentration of 1×105 cells/mL and incubated with 100-μL RPMI 1640 medium per well. After 24 h, 100-μL HACC and De-HACC with different degrees of substitution diluted to 100 μg/mL with RPMI 1640 medium were added to the wells for 24 h of incubation. Wells containing 1 μg/mL lipopolysaccharides (LPS) or RPMI 1640 alone were included as controls. After 24 h, the cell supernatant was collected in a new 96-well plate and mixed with an equal volume of Griess reagent and incubated for 10 min at room temperature under dark conditions. Then, the absorbance at 540 nm was determined using an M200 Pro spectrophotometer. The expressed NO production was calculated by the NaNO2 standard curve.
2.10 RNA isolation and reverse transcription-polymerase chain reactionRAW 264.7 macrophages were seeded onto 6-well plates for 24 h at 1×106 cells/well. Then, the cells were stimulated with HACC and De-HACC with different degrees of substitution (100 μg/mL), and RPMI 1640 culture medium was used as the control group. After 24 h of incubation, the original suspension was discarded, and phosphate buffer saline (PBS) was added to wash the cells. After that, the cells were collected to extract total RNA using TRIzol reagent (E.Z.N.A. Total RNA Kit). For subsequent successful amplification of DNA, we also measured the RNA concentration using a Nanodrop spectrophotometer before reverse transcription. Meanwhile, the original mixed DNA was removed at 42 ℃ for 2 min. Single-stranded cDNA was synthesized using a reverse transcription kit according to the instructions. After synthesis, the cDNA was diluted 5–10 times using sterile water before the amplification experiment. Then, amplification was performed according to the introduction of the PCR procedure. Specific PCR primers were designed and are shown in Table 1. The relative mRNA levels of all tested genes were calculated by the real-time quantitative PCR 2-ΔΔCt method.
The given data are expressed as the mean±standard deviation (SD). The statistical analysis was performed by analysis of variance (ANOVA) via Statistical Product and Service Solutions software (SPSS). Values at P < 0.05 were considered significant.
3 RESULT 3.1 Characterization of fully deacetylated chitosanThe infrared spectra of chitosan and fully deacetylated chitosan are shown in Fig. 2. The differences indicate the removal of the acetyl group on fully deacetylated chitosan chains. In the spectrum, the peak at 1 644 cm-1 represents the C=O stretching vibration peak of the original chitosan. Comparatively, we found that the peak at 1 644 cm-1 in the fully deacetylated chitosan disappeared, accompanied by an increase in the bending vibration peak at 1 589 cm-1 of the free amino group. These results indicated the successful preparation of fully deacetylated chitosan. In the 1HNMR spectra, strong and sharp peaks at 1.7×10-6 represent the characteristic peaks of primary amines exposed by the successful removal of acetyl groups. Compared with raw chitosan, the higher deacetylated samples had stronger signals at 2.95×10-6, which was attributed to GlcN H-2. In addition, the molecular weight of raw chitosan did not change much after deacetylation, and the molecular weight of fully deacetylated chitosan was 1 780 kDa.
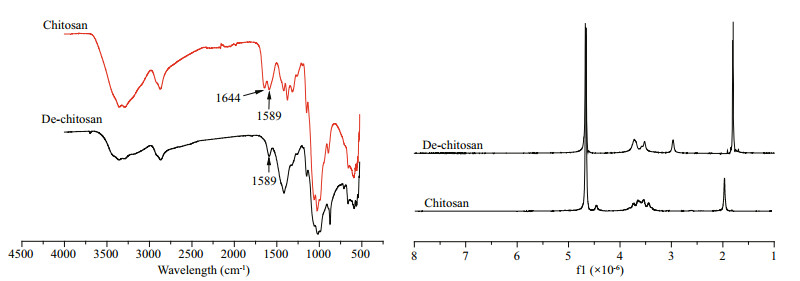
|
| Fig.2 Infrared and 1 HNMR spectra of raw chitosan and fully deacetylated chitosan |
HACC and De-HACC with different degrees of quaternization were prepared by altering the feeding ratio. To detect whether the quaternary ammonium salt groups were successfully introduced, the products were tested by FTIR and 1HNMR (Fig. 3a–d). In the FTIR spectra, the strong absorption peaks at 1 476 cm-1were attributed to C-H bending of methyl groups on the grafted trimethyl ammonium group. Moreover, the spectrum of HACC showed a broader absorption band at approximately 3 300 cm-1 for the increased number of hydroxyl groups with the continuous introduction of quaternary ammonium groups. In addition, the peaks in the range of 1 152–1 072 cm-1 did not change in HACC, indicating the successful introduction of quaternary ammonium groups on the -NH2 sites. In the 1HNMR spectra, the strongest peak at δ=3.09×10-6 was attributed to the protons of the methyl groups of HACC. The other signals at 4.18× 10-6, 3.29×10-6, and 2.65×10-6 were assigned to H8, H10, and H7 of the introduced quaternary ammonium groups, respectively. The signals at 4.45×10-6, 3.88×10-6, 3.65×10-6, 3.70×10-6, 3.55×10-6, and 2.47×10-6 were attributed to H1, H6, H4, H5, H3, and H2 of the chitosan chain, respectively.
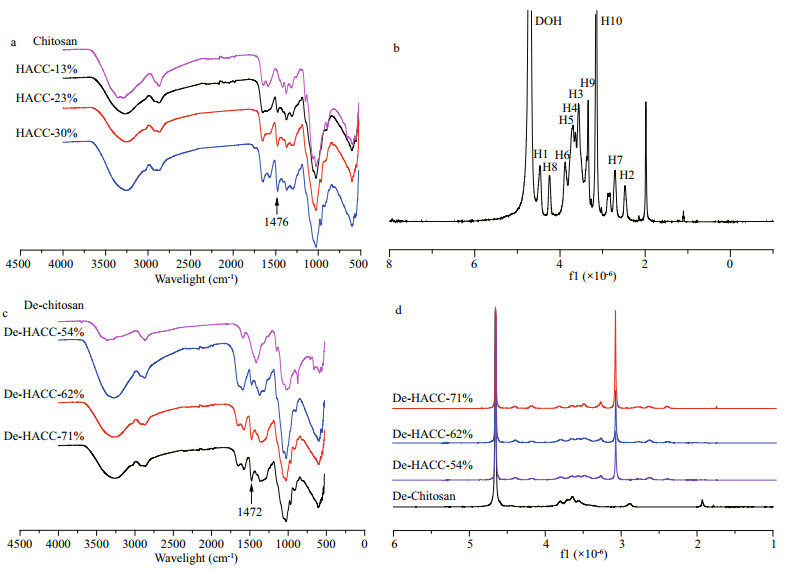
|
| Fig.3 Characterization analysis of HACC and De-HACC with different degrees of quaternization a. FTIR spectra of HACC; b. 1HNMR spectra of HACC (30% HACC, for example); c. FTIR spectra of De-HACC; d. 1HNMR spectra of De-HACC. |
De-HACC could be confirmed in Fig. 3c–d. In FTIR, a new peak of quaternary chitosan appeared at 1 472 cm-1, indicating that the quaternary ammonium group was successfully synthesized on chitosan. In the 1HNMR spectra, the most intense peak at 3.10×10-6 was assigned to the characteristic peak of the trimethyl group on the introduced quaternary ammonium group.
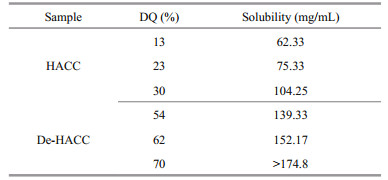
3.3 DQ characterizations of HACC and De-HACCThe DQs of HACC and De-HACC were measured by conductivity titration. As listed in Table 2, ranges of different quaternary degrees of HACC and De-HACC were successfully synthesized by altering the ratio of chitosan to GTMAC. As shown, the DQ of HACC increased from 13% to 30% when the ratio was increased from 1꞉1 to 1꞉5. This is because with increasing GTMAC concentration, the probability of GTMAC entering the chitosan chain improves, which makes the reaction between chitosan and GTMAC more thorough; as a result, the DQ also increases. However, further continuous addition of GTMAC to the chitosan solution resulted in an obvious decrease in the DQ (Fig. 4), which was probably caused by the intermolecular polymerization of GTMAC and the steric hindrance of chitosan. Therefore, after comprehensive consideration, we chose HACC 13%, HACC 23% and HACC 30% as the following test materials.
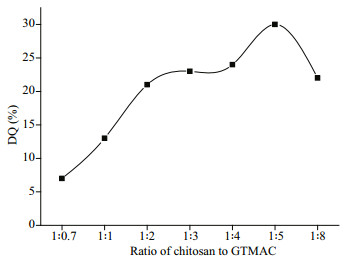
|
| Fig.4 Effect of different ratios of chitosan to GTMAC on DQ |
Fully deacetylated chitosan can more easily introduce quaternary ammonium groups. As shown in our results, although the reaction ratio of chitosan to GTMAC was only 1꞉1, surprisingly, a 54% degree of quaternization was obtained. Obviously, the removal of acetyl groups is helpful for the improvement of DQ. On the one hand, after the acetyl group is removed, the steric hindrance encountered by the introduced group is reduced, which increases the possibility for the quaternary ammonium group to bond. On the other hand, the -NH originally connected to the acetyl group of chitosan turns to -NH2 due to the removal of the acetyl group, resulting in more active sites.
3.4 Characterization of the solubility of HACC and De-HACCThe solubilities of HACC and De-HACC are summarized in Table 3. As the table shows, as the quaternary degree increased, the solubility of quaternary chitosan was enhanced. However, chitosan itself is insoluble in water due to strong hydrogen bonding. The introduction of quaternary ammonium groups into chitosan chain can greatly weaken the hydrogen bonds between chitosan molecules so that its water solubility can be improved. For De-HACC, its solubility is generally higher than that of HACC.
The RAW 264.7 cell cytotoxicities of various HACC and De-HACC formulations were measured by the MTT method. The results are shown in Fig. 5, and the data indicated that HACC and De-HACC were safe and nontoxic at concentrations of 200 and 250 μg/mL, respectively. However, high quaternary ammonium was accompanied by a certain degree of toxicity. This was especially obvious at a concentration of 500 μg/mL fully deacetylated quaternary ammonium salt. When the degree of quaternization was 54%, there was no damage to the cells, but 71% quaternized fully deacetylated chitosan produced obvious toxicity. Therefore, to eliminate toxic interference, we applied a concentration of 100 μg/ mL in the following experiments.
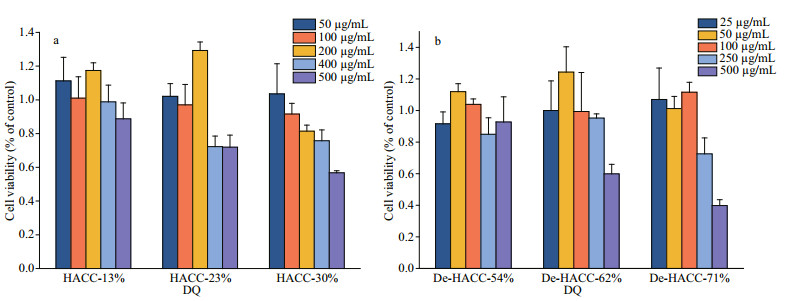
|
| Fig.5 The effects of HACC (a) and De-HACC (b) on cell viability |
NO is an important signaling molecule for the immune activity of macrophages. To determine whether the immunostimulatory activities of HACC and De-HACC were related to the degree of quaternization, the NO-promoting expression levels of HACC and De-HACC with different quaternization degrees were compared. Figure 6 shows the effects of HACC and De-HACC with different quaternary degrees on the expression of NO in RAW 264.7 cells. The results show that compared with the control group, HACC and De-HACC effectively promoted the production of NO, indicating that quaternary ammonium salt is an effective immunostimulant. Furthermore, the effects of HACC and De-HACC far exceeded those of chitosan and De-chitosan respectively. This effect is related to the degree of quaternization but is not linearly proportional. As shown in Fig. 6, as the degree of quaternization of HACC increased, the promoting effect of NO was enhanced, and 30% quaternization was the best, at 10 times that of the control group. However, De-HACC exhibited the opposite trend: as the quaternary ammonium degree increased, the NO-promoting effect decreased, and the 54% quaternization effect was superior, at 20 times that of the control group. In comparison, De-HACC 54% displayed the best promotion effect.
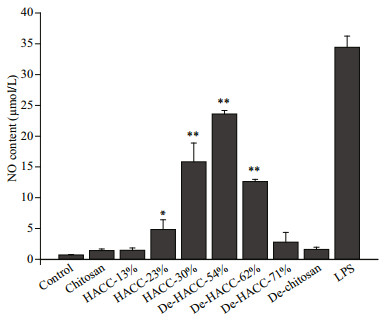
|
| Fig.6 The effects of HACC and De-HACC on NO expression levels HACC-13%: 13% quaternized hydroxypropyltrimethyl ammonium chloride chitosan; HACC-23%: 23% quaternized hydroxypropyltrimethyl ammonium chloride chitosan; HACC-30%: 30% quaternized hydroxypropyltrimethyl ammonium chloride chitosan; De-HACC-54%: 54% quaternized fully deacetylated hydroxypropyltrimethyl ammonium chloride chitosan; De-HACC-62%: 62% quaternized fully deacetylated hydroxypropyltrimethyl ammonium chloride chitosan; De-HACC-71%: 71% quaternized fully deacetylated hydroxypropyltrimethyl ammonium chloride chitosan. HACC versus control and De-HACC versus control.*: P < 0.05; **: P < 0.01. |
IL-6 and TNF-α are both important proinflammatory cytokines that are involved in immune responses. To test and compare various HACC and De-HACC formulations on the effect of gene expression, the RNA extracted from RAW 264.7 cells was subjected to quantitative PCR after reverse transcription to test the expression levels of selected genes, and the results are shown in Fig. 7. The gene expression of IL-6 and TNF-α was calculated by the 2-ΔΔCt method. This study mainly evaluated the effects of different quaternary degrees on quaternary chitosan, the results of IL-6 and TNF-α caused by chitosan deacetylation and chitosan were not measured.
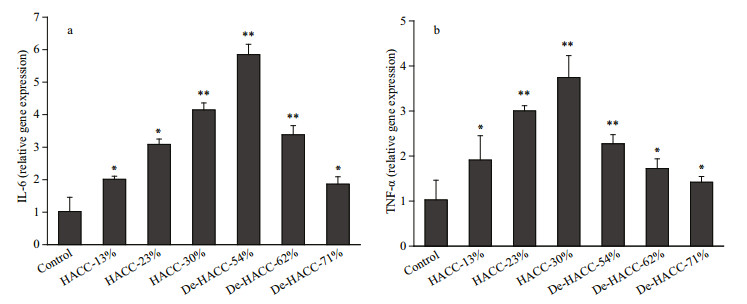
|
| Fig.7 Gene expression levels of two types of quaternary chitosan a. IL-6 expression on RAW 264.7 cells of HACC and De-HACC; b. TNF-α expression on RAW 264.7 cells of HACC and De-HACC. The values are given as means±SD (n=3). HACC versus control and De-HACC versus control. *: P < 0.05; **: P < 0.01. |
The results show that HACC and De-HACC displayed upregulation of the expression levels of immune factor genes and were affected by the quaternary degree. More specifically, among the HACC group, the 30% degree of quaternization had better effects on the gene expression levels of the cytokines IL-6 and TNF-α. However, for De-HACC, the promotion level decreased with increasing quaternary degree, and a 54% quaternization degree showed higher expression levels, with 6-fold upregulation of IL-6 compared with the control group.
4 DISCUSSIONImmune adjuvants have already become an indispensable part of improving vaccine effectiveness. The ideal adjuvant should be broad-spectrum, have no side effects, be nontoxic, have good biocompatibility, be effective in activating the immune system, and be easy to produce and use (Portuondo et al., 2015; Trier et al., 2019).
Chitosan quaternary ammonium salt is a positively charged chitosan derivative, and previous studies have shown that it is nontoxic and has good biocompatibility and good immunostimulatory properties (Jin et al., 2013). The degree of substitution of the quaternary ammonium group has a significant impact on the performance of immune activity (Hamman et al., 2002). However, the effects of the optimum quaternary ammonium degree and the acetylation degree of chitosan on immune activity have not been reported. In this paper, quaternized chitosan (HACC) and fully deacetylated chitosan (De-HACC) with different degrees of substitution were successfully synthesized. During the synthesis process, it was found that changing the ratio of chitosan to GTMAC can effectively increase the degree of substitution, but under the influence of steric hindrance, the quaternary ammonium degree will decrease. In contrast, the removal of the acetyl group of chitosan reduces the steric hindrance and increases the exposure of the active sites. Therefore, the degree of quaternary ammonium is higher under the same ratio. In other words, the acetyl group in chitosan has a significant effect on the degree of substitution of quaternary ammonium salt of chitosan. To obtain a high degree of substitution of chitosan quaternary ammonium salt products, removing the acetyl group of chitosan is effective. At the same time, in this study, we found that the immunostimulatory effect of HACC on RAW 264.7 cells increased with increasing quaternary ammonium levels. However, due to the high substitution degree of De-HACC, the immunostimulatory effect on RAW 264.7 cells decreased with increasing quaternary ammonium levels.
Macrophages play a crucial role in the immune system. They release a large number of anti-inflammatory factors to provoke the immune response when activated effectively, among which NO is an important active signaling molecule. Thus, in this study, NO expression in RAW 264.7 cells evoked by HACC and De-HACC with various quaternary degrees was first determined. The results showed that the bioactivity of HACC was related to the degree of quaternization, which was also dependent on structure. In the HACC, a higher quaternary degree (30%) showed better stimulation of NO expression than the other DQs. In contrast, in De-HACC, De-HACC with a DQ of 54% had a more obvious promotion effect on NO production than other higher DQs. Moreover, for De-HACC with a DQ of 54%, the promotion effect on NO was better than HACC with a DQ of 30%, which indicates that the acetyl group plays an important role in the quaternary ammonium salt of chitosan. Further research is needed to determine how these differences influence function.
Cytokines are secreted by stimulated immune cells and act as signaling molecules in mediating immune responses, including innate and adaptive immunity (Devi et al., 2019). The secretion level of cytokines can be used as an important indicator to reflect the effect of the immune response. IL-6 enables activating lymphocytes to influence a specific immune response (Fan et al., 2015). TNF-α plays a significant role in triggering an inflammatory response and activating adaptive immunity against viral infection (Guo et al., 2018). It was reported that upregulation of IL-6 and TNF-α was enhanced after macrophages were exposed to chitosan (Koppolu and Zaharoff, 2013). To further investigate the effects of quaternary degrees of HACC and De-HACC on macrophages, gene expression levels of IL-6 and TNF-α were measured. We found that the substitution degree of quaternary chitosan vitally influenced the expression of immune factors. Specifically, 30%-HACC introduced higher gene expression levels of both IL-6 and TNF-α. Similarly, in De-HACC, obvious differences in IL-6 and TNF-α were observed between different quaternary degrees. Among them, 54% De-HACC performed better in promoting the production of these proinflammatory cytokines. Our results are consistent with many reported studies. The DQ of quaternized chitosan profoundly affected their bioactivities. Malik et al. (2018) reported that a degree of quaternization between 40%–50% was effective in drug delivery systems. Peng et al. (2010) found that the antibacterial activities of HACC were substitution-dependent. Hamman et al. (2002) concluded that the degree of quaternization (> 36%) of TMC displayed enhanced absorption properties across nasal epithelia. In addition, Kotzé et al. (1999b) found that the degree of quaternization played a vital role in the penetration enhancement of Caco-2 cells. Moreover, an exaggerated high quaternary degree will cause toxic effects and bring negative effects. This can fully demonstrate the importance of the quaternary degree for studying the biological activity of quaternary ammonium salts.
In this preliminary study, we found that the presence of acetyl groups affected the degree of substitution of quaternary chitosan. Once the acetyl group was removed, the degree of substitution was effectively improved. Furthermore, we also thought that the immunostimulatory effect of quaternary ammonium was deeply DQ-related. Previous research by our team found that the quaternary ammonium salt of chitosan could activate RAW 264.7 macrophages through the mitogen-activated protein kinase (MAPK) and janus kinase/signal transducer and activator of transcription (JAK-STAT) signaling pathways (Yang et al., 2019). A possible explanation for the activation of these immune pathways is that ammonium salts can be recognized by toll-like receptors (TLRs) such as TLR4 (Zhang et al., 2014). Moreover, the immune function of macrophages is inseparable from the supply of energy. Therefore, the effector function of immune cells is deeply affected by metabolism. M1 macrophage energy is supplied mainly through glycolysis (Jha et al., 2015). Therefore, after the influence of quaternary ammonium degree and the acetyl group was determined, whether the improvement of the immune level of macrophages by chitosan quaternary ammonium salt is achieved by regulating metabolism has become an interest point of our follow-up research, with the hope of providing more directional significance for the development of vaccine adjuvants.
5 CONCLUSIONIn this study, we successfully synthesized 6 kinds of chitosan quaternary ammonium salts, including HACC and De-HACC, with different substitutions. We also explored the immunostimulatory influence of different DQs on RAW 264.7 macrophages. The results show that removal of acetyl groups is beneficial to the improvement of the degree of substitution of chitosan. HACC and De-HACC can effectively promote the production of NO, particularly 30% HACC and 54% De-HACC. In addition, HACC and De-HACC displayed immune-boosting functions by improving the expression of proinflammatory mediators, including TNF-α and IL-6. Moreover, the immunostimulatory activity of 54% De-HACC was better than that of 30% HACC. All together, these results confirm that the immunostimulatory activity of HACC and De-HACC was DQ-dependent and was influenced by the acetyl group.
6 DATA AVAILABILITY STATEMENTThe data generated and/or analyzed during the current study are available from the corresponding author Rong'e XING on reasonable request.
Amidi M, Hennink W E. 2010. Chitosan-based formulations of drugs, imaging agents and biotherapeutics. Advanced Drug Delivery Reviews, 62(1): 1-2.
DOI:10.1016/j.addr.2009.12.006 |
Aucouturier J, Dupuis L, Ganne V. 2001. Adjuvants designed for veterinary and human vaccines. Vaccine, 19(17-19): 2666-2672.
DOI:10.1016/s0264-410x(00)00498-9 |
Baek K S, Hong Y D, Kim Y, Sung N Y, Yang S, Lee K M, Park J Y, Park J S, Rho H S, Shin S S, Cho J Y. 2015. Antiinflammatory activity of AP-SF, a ginsenoside-enriched fraction, from Korean ginseng. Journal of Ginseng Research, 39(2): 155-161.
DOI:10.1016/j.jgr.2014.10.004 |
Costain D J, Kennedy R, Ciona C, McAlister V C, Lee T D G. 1997. Prevention of postsurgical adhesions with N-O, carboxymethyl chitosan: examination of the most efficacious preparation and the effect of N-O-carboxymethyl chitosan on postsurgical healing. Surgery, 121(3): 314-319.
DOI:10.1016/S0039-6060(97)90360-3 |
Devi G, Harikrishnan R, Paray B A, Al-Sadoon M K, Hoseinifar S H. 2019. Effects of aloe-emodin on innate immunity, antioxidant and immune cytokines mechanisms in the head kidney leucocytes of Labeo rohita against Aphanomyces invadans. Fish & Shellfish Immunology, 87: 669-678.
DOI:10.1016/j.fsi.2019.02.006 |
Dimassi S, Tabary N, Chai F, Blanchemain N, Martel B. 2018. Sulfonated and sulfated chitosan derivatives for biomedical applications: a review. Carbohydrate Polymers, 202: 382-396.
DOI:10.1016/j.carbpol.2018.09.011 |
Fan Y P, Ma X, Zhang J, Ma L, Gao Y Y, Zhang W M, Song X P, Hou W F, Guo C, Tong D W. 2015. Ophiopogon polysaccharide liposome can enhance the non-specific and specific immune response in chickens. Carbohydrate Polymers, 119: 219-227.
DOI:10.1016/j.carbpol.2014.11.048 |
Guimaräes L E, Baker B, Perricone C, Shoenfeld Y. 2015. Vaccines, adjuvants and autoimmunity. Pharmacological Research, 100: 190-209.
DOI:10.1016/j.phrs.2015.08.003 |
Guo M, Tang X Q, Sheng X Z, Xing J, Zhan W B. 2018. The effects of IL-1β, IL-8, G-CSF and TNF-α as molecular adjuvant on the immune response to an E. tarda subunit vaccine in flounder (Paralichthys olivaceus). Fish & Shellfish Immunology, 77: 374-384.
DOI:10.1016/j.fsi.2018.04.009 |
Guo Z Y, Xing R E, Liu S, Zhong Z M, Ji X, Wang L, Li P C. 2007. The influence of the cationic of quaternized chitosan on antifungal activity. International Journal of Food Microbiology, 118(2): 214-217.
DOI:10.1016/j.ijfoodmicro.2007.07.003 |
Hamman J H, Stander M, Kotzé A F. 2002. Effect of the degree of quaternisation of N-trimethyl chitosan chloride on absorption enhancement: in vivo evaluation in rat nasal epithelia. International Journal of Pharmaceutics, 232(1-2): 235-242.
DOI:10.1016/s0378-5173(01)00914-0 |
He X F, Li K C, Xing R E, Liu S, Hu L F, Li P C. 2016. The production of fully deacetylated chitosan by compression method. The Egyptian Journal of Aquatic Research, 42(1): 75-81.
DOI:10.1016/j.ejar.2015.09.003 |
Jha A K, Huang S C C, Sergushichev A, Lampropoulou VIvanova Y, Loginicheva E, Chmielewski K, Stewart K, MAshall J, Everts B, Pearce E J, Driggers E M, Artyomov M N. 2015. Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity, 42(3): 419-430.
DOI:10.1016/j.immuni.2015.02.005 |
Jin Z, Li W, Cao H W, Zhang X, Chen G, Wu H, Guo C, Zhang Y, Kang H, Wang Y F, Zhao K. 2013. Antimicrobial activity and cytotoxicity of N-2-HACC and characterization of nanoparticles with N-2-HACC and CMC as a vaccine carrier. Chemical Engineering Journal, 221: 331-341.
DOI:10.1016/j.cej.2013.02.011 |
Koppolu B, Zaharoff D A. 2013. The effect of antigen encapsulation in chitosan particles on uptake, activation and presentation by antigen presenting cells. Biomaterials, 34(9): 2359-2369.
DOI:10.1016/j.biomaterials.2012.11.066 |
Kotzé A F, Thanou M M, Lueben H L, De Boer A G, Verhoef J C, Junginger H E. 1999a. Enhancement of paracellular drug transport with highly quaternized iV-trimethyl chitosan chloride in neutral environments: in vitro evaluation in intestinal epithelial cells (Caco-2). Journal of Pharmaceutical Sciences, 88(2): 253-257.
DOI:10.1021/js980233c |
Kotzé A F, Thanou M M, Lueßen H L, de Boer B G, Verhoef J C, Junginger H E. 1999b. Effect of the degree of quaternization of N-trimethyl chitosan chloride on the permeability of intestinal epithelial cells (Caco-2). European Journal of Pharmaceutics and Biopharmaceutics, 47: 269-274.
DOI:10.1016/s0939-6411(99)00006-5 |
Li X S, Min M, Du N, Gu Y, Hode T, Naylor M, Chen D J, Nordquist R E, Chen W R. 2013. Chitin, chitosan, and glycated chitosan regulate immune responses: the novel adjuvants for cancer vaccine. Clinical and Developmental Immunology, 2013: 387023.
DOI:10.1155/2013/387023 |
Liu Q F, Zheng X Y, Zhang C, Shao X Y, Zhang X, Zhang Q Z, Jiang X G. 2015. Conjugating influenza a (H1N1) antigen to n-trimethylaminoethylmethacrylate chitosan nanoparticles improves the immunogenicity of the antigen after nasal administration. Journal of Medical Virology, 87(11): 1807-1815.
DOI:10.1002/jmv.24253 |
Malik A, Gupta M, Gupta V, Gogoi H, Bhatnagar R. 2018. Novel application of trimethyl chitosan as an adjuvant in vaccine delivery. International Journal of Vanomedicine Volume, 13: 7959-7970.
DOI:10.2147/ijn.s165876 |
Nishimura K, Nishimura S, Nishi N, Saiki I, Tokura S, Azuma I. 1984. Immunological activity of chitin derivatives. Vaccine, 2(1): 93-99.
DOI:10.1016/S0264410X(98)90039-1 |
Peng Z X, Wang L, Du L, Guo S R, Wang X Q, Tang T T. 2010. Adjustment of the antibacterial activity and biocompatibility of hydroxypropyltrimethyl ammonium chloride chitosan by varying the degree of substitution of quaternary ammonium. Carbohydrate Polymers, 81(2): 275-283.
DOI:10.1016/j.carbpol.2010.02.008 |
Petrovsky N, Aguilar J C. 2004. Vaccine adjuvants: current state and future trends. Immunology & Cell Biology, 82(5): 488-496.
DOI:10.1111/j.0818-9641.2004.01272.x |
Portuondo D L F, Ferreira L S, Urbaczek A C, Batista-Duharte A, Carlos I Z. 2015. Adjuvants and delivery systems for antifungal vaccines: current state and future developments. Medical Mycology, 53(1): 69-89.
DOI:10.1093/mmy/myu045 |
Spinelli V A, Laranjeira M C M, Fávere V T. 2004. Preparation and characterization of quaternary chitosan salt: adsorption equilibrium of chromium(VI) ion. Reactive and Functional Polymers, 61(3): 347-352.
DOI:10.1016/j.reactfunctpolym.2004.06.010 |
Sun B N, Yu S, Zhao D Y, Guo S H, Wang X H, Zhao K. 2018. Polysaccharides as vaccine adjuvants. Vaccine, 36(35): 5226-5234.
DOI:10.1016/j.vaccine.2018.07.040 |
Suzuki K, Okawa Y, Hashimoto K, Suzuki S, Suzuki M. 1984. Protecting effect of chitin and chitosan on experimentally induced murine candidiasis. Microbiology and Immunology, 28(8): 903-912.
DOI:10.1111/j.1348-0421.1984.tb00746.x |
Thanou M, Florea B I, Geldof M, Junginger H E, Borchard G. 2002. Quaternized chitosan oligomers as novel gene delivery vectors in epithelial cell lines. Biomaterials, 23(1): 153-159.
DOI:10.1016/S0142-9612(01)00090-4 |
Trier N H, Güven E, Skogstrand K, Ciplys E, Slibinskas R, Houen G. 2019. Comparison of immunological adjuvants. APMS, 127(9): 635-641.
DOI:10.1111/apm.12976 |
Wang Y Q, Fan Q Z, Liu Y, Yue H, Ma X W, Wu J, Ma G H, Su Z G. 2016. Improving adjuvanticity of quaternized chitosan-based microgels for H5N1 split vaccine by tailoring the particle properties to achieve antigen dose sparing effect. International Journal of Pharmaceutics, 515(1-2): 84-93.
DOI:10.1016/j.ijpharm.2016.09.082 |
Yang Y, Xing R E, Liu S, Qin Y K, Li K C, Yu H H, Li P C. 2019. Hydroxypropyltrimethyl ammonium chloride chitosan activates RAW 264.7 macrophages through the MAPK and JAK-STAT signaling pathways. Carbohydrate Polymers, 205: 401-409.
DOI:10.1016/j.carbpol.2018.10.101 |
Yüksel S, Pekcan M, Puralı N, Esendağlı G, Tavukçuoğlu E, Rivero-Arredondo V, Ontiveros-Padilla L, López-Macías C, Şenel S. 2020. Development and in vitro evaluation of a new adjuvant system containing salmonella Typhi porins and chitosan. International Journal of Pharmaceutics, 578: 119129.
DOI:10.1016/j.ijpharm.2020.119129 |
Zhang G Q, Jia P Y, Liu H T, Hu T, Du Y G. 2018. Conjugation of chitosan oligosaccharides enhances immune response to porcine circovirus vaccine by activating macrophages. Immunobiology, 223(11): 663-670.
DOI:10.1016/j.imbio.2018.07.012 |
Zhang P, Liu W Z, Peng Y F, Han B Q, Yang Y. 2014. Toll like receptor 4 (TLR4) mediates the stimulating activities of chitosan oligosaccharide on macrophages. International Immunopharmacology, 23(1): 254-261.
DOI:10.1016/j.intimp.2014.09.007 |
Zhao K, Sun Y W, Chen G, Rong G Y, Kang H, Jin Z, Wang X H. 2016. Biological evaluation of V-2-hydroxypropyl trimethyl ammonium chloride chitosan as a carrier for the delivery of live Newcastle disease vaccine. Carbohydrate Polymers, 149: 28-39.
DOI:10.1016/j.carbpol.2016.04.085 |
Zhou M, Qu W Y, Sun Y W, Liang L, Jin Z, Cui S J, Zhao K. 2020. Water-soluble V-2-Hydroxypropyl trimethyl ammonium chloride chitosan enhanced the immunogenicity of inactivated porcine parvovirus vaccine vaccination on sows against porcine parvovirus infection. Immunology Letters, 223: 26-32.
DOI:10.1016/j.imlet.2020.04.014 |
 2022, Vol. 40
2022, Vol. 40