Institute of Oceanology, Chinese Academy of Sciences
Article Information
- LIU Weixiang, SUN Chaomin
- C17-fengycin B, produced by deep-sea-derived Bacillus subtilis, possessing a strong antifungal activity against Fusarium solani
- Journal of Oceanology and Limnology, 39(5): 1938-1947
- http://dx.doi.org/10.1007/s00343-020-0215-2
Article History
- Received Jun. 2, 2020
- accepted in principle Sep. 22, 2020
- accepted for publication Oct. 8, 2020
2 Laboratory for Marine Biology and Biotechnology, Pilot National Laboratory for Marine Science and Technology (Qingdao), Qingdao 266237, China
Root rot is a kind of soil-borne disease which is common in the global economic crop production area (Egamberdieva et al., 2017). The first research on root rot was reported by von Schrenk in 1902 when he was studying apple tree diseases (Von Schrenk and Spaulding, 1902). Since then, root rot infections have been found in potato (Al-Mughrabi et al., 2013), soybean (Cui et al., 2016), cucumber (Wiggell and Simpson, 1969) and other crops. At present, root rot has become one of the main diseases of economic crops, which can reduce the yield by 25%–60% in serious cases (Coetzee et al., 2018). Root rot can be caused by a variety of soil microorganisms, among which Fusarium spp. is the main group (Egamberdieva et al., 2017; Chang et al., 2018). Fusarium spp. has a wide range of distribution, and Fusarium solani is the main pathogen of root rot because of its high isolation frequency and strong pathogenicity (Chittem et al., 2015; Schroers et al., 2016). At present, the control strategies of Fusarium mainly include chemical control and biological control (Patzke et al., 2017; D'Agostino et al., 2018). Among them, chemical control method has the advantages of low cost, good effect, quick effect and easy to use, which has been wildly used (Bonilla-Landa et al., 2018). However, excessive use of chemical pesticides will lead to soil pollution, food pesticide residues, and other problems, which brings a huge threat to environment and food safety (Lewis et al., 2016; Shugart, 2017; Nile et al., 2019). At the same time, long-term use of chemical pesticides will lead to drug resistance of pathogenic microorganisms (Sav et al., 2018; da Rosa et al., 2019). Therefore, it is an urgent task to find new effective and green biological pesticides to control root rot (Azizbekyan, 2019).
For decades, researchers have been studying marine natural products and found a series of compounds with significant biological activities (Blunt et al., 2018). Marine bacteria are an important branch of natural product research. It can be cultured by large-scale fermentation, which solves the problem of limited production of separated compounds and the sustainability of marine microbial resources (Kobayashi, 2016). In 2018 alone, 1 554 new marine derived compounds were reported, many of which have antibacterial, insecticidal, or antitumor activities (Carroll et al., 2020).
Bacillus is a kind of Gram-positive bacteria, which can adapt to a variety of extreme environments, even in the deep sea (Shafi et al., 2017). Research on the secondary metabolites of Bacillus from marine environment has been always a hot spot in the research of natural products (Zeigler and Nicholson, 2017). Many types of compounds, such as ester peptide, polypeptide, polyketone, and fatty acid, have been isolated from their metabolites. Meanwhile, these compounds also have antibacterial, antitumor, and other biological activities (Li et al., 2011; Xing et al., 2018). In this study, we purified C17-fengycin B from the metabolites of B. subtilis 2H11 (BS2H11) isolated from the sediment of deep-sea cold seep. The structure was analyzed by electrospray ionization mass spectrometry (ESI-MS) and tandem mass spectrometry (ESI-MS/MS). The antifungal property of C17-fengycin B against F. solani was evaluated. In addition, we also studied the effects of C17-fengycin B on spores and hyphae of F. solani by electron microscopy. The ester peptide derivative may be developed as an agricultural antifungal agent.
2 MATERIAL AND METHOD 2.1 Isolation, identification, and culture conditions of bacterial strainsMarine sediment was collected by RV Kexue (Science in Chinese) from the cold seep in the South China Sea (119°17'04.956"E, 22°06'58.384"N) at a depth of approximately 1 143 m in September 2017. The marine bacterial strain used in this study was isolated from the above-described samples by dilution method and cultured in the improved ZoBell 2216E broth (5-g/L tryptone, 1-g/L yeast extract, 1-L filtered seawater, pH adjusted to 7.4 to 7.6), with the temperature of 28 ℃ (Schut et al., 1993). To determine the phylogenetic position of the bacterial strain, universal primers 27F (5'-AGAGTTTGATCCTGG-CTCAG-3') and 1492R (5'-TACGGCTACCTTGT-TACGACTT-3') specific for bacterial 16S rRNA genes were used to amplify the corresponding gene. Then, NCBI-BLAST (see http://www.ncbi.nlm.nih.gov/BLAST) and the phylogenetic analysis program MEGA6 were used to compare the 16S rRNA gene sequence with related sequence in public databases (Tamura et al., 2013).
2.2 Screening of bacteria inhibiting the growth of F. solaniIn this study, F. solani, the main pathogen of root rot, was taken as the target fungus. To determine the antifungal activity of the bacteria strain, the surface plating method was used. The test method was as previously described in Zhang and Sun (2018). The bacterial suspension was incubated overnight and adjusted to 0.2 at 600 nm (OD600). F. solani was placed on potato glucose agar (PDA) plate and cultured at 28 ℃ for 5 d. The hyphal plug (0.5 cm in diameter) removed from the edge of the F. solani culture was placed in the center of the new plate. Ten microliters of different bacteria were inoculated 3 cm from the plug edge of F. solani. Plates containing only F. solani, or plates containing different bacteria and F. solani, were incubated at 28 ℃ for another 3 days, and then the growth area of fungi was measured. Then, the results were obtained by observing the presence (growth) or absence (non-growth) of fungi.
2.3 Isolation, purification, and identification of antifungal compound from B. subtilis 2H11The single colony of B. subtilis 2H11 strain was inoculated into a 250-mL flask containing 100-mL of Luria-Bertani (LB) medium and cultured at 180 r/min for 24 h at 28 ℃. Then 10 mL of this seed culture was inoculated into a 3-L flask containing 1 L of LB medium and cultured at 180 r/min for another 24 h at 28 ℃. The supernatant was obtained by centrifuging the fermentation broth (8 000×g, 4 ℃, 10 min). The pH of the supernatant was adjusted to 2.5 with 6 mol/L HCl at 4 ℃. When the solution turned clear, the precipitate was obtained by centrifugation (8 000×g, 4 ℃, 10 min), washed with 0.1 mol/L HCl and then extracted with methanol filtered through a 0.22-μm membrane and concentrated under reduced pressure. The supernatant was then passed through a Sephadex LH-20 column for fractionation and eluted with methanol as the mobile phase (Ramos and Prohaska, 1981). The eluted fraction was concentrated with a vacuum rotary evaporator and determined for antifungal activity using the paper disc method (Kordali et al., 2008). The eluted fraction with antifungal activity was filtered through a 0.22-μm membrane filter and further purified by reversed-phase high performance liquid chromatography (RP-HPLC) (Agilent 1260) equipped with an Eclipse XDB-C18 column (5 μm, 9.4×250 mm). The mobile phase A consisting of water and methanol (20:80, v/v) and the mobile phase B was methanol. The eluting strategy was as follows: 0–20 min, 0% B; 20–60 min, 0% B to 100% B and 60–80 min, 100% B. The flow rate was 2 mL/min and the elution was monitored using a UV detector set at 210 nm. Each elution peak was collected and the antifungal activity was determined. The stability of the active compound(s) was determined by HPLC column twice.
Mass spectra of active antifungal substances was analyzed by linear ion trap Orbitrap spectrometer (LTQ Orbitrap XL, Thermo fisher) using high-energy collision induced dissociation (HCD), which was a new mass spectrometry pyrolysis technology and could provide abundant fragmentation information. Data from HCD-MS/MS were acquired under the following conditions: electrospray ion source (ESI), spray voltage 3 kV, ion transfer capillary temperature 275 ℃; the dry gas was nitrogen gas, and the pressure was 0.05 MPa; HCD collision gas was helium, anion pattern detection; the collision energy of HCD is 45–60 eV. And then the results were analyzed by Xcalibur 2.1 (Thermo Fisher Scientific Inc, Waltham, MA).
2.4 Antifungal assay (in vitro)The antifungal activity was evaluated in-vitro against F. solani by a mycelium growth rate test. Data were collected as previously described in literature (de Rodríguez et al., 2005). The antifungal activity was detected at the concentrations of 0.05, 0.10, and 0.20 mg/mL, respectively. The plates inoculated with mycelia of F. solani was cultured at 27 ℃. When the mycelium reached the edge of the negative control plate (no sample was added), the antifungal index was calculated by the following formula:
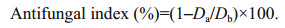
Da and Db are the diameter of hyphae in the test plate and negative control plate respectively. Each test was repeated three times, and the results were averaged.
2.5 Protective and curative Activity (in vivo)In this study, in-vivo antifungal activity was measured by pot experiment, and the protective and curative activity of the samples were measured. Because of the large amount of samples needed in the pot experiment, the crude extract was used. Data were collected as previously described in Benaouali et al. (2014). The experiment was carried out when tomato seedlings were cultured to at least 8 true leaves. For protective activity, the seedlings were treated with 10-mL test reagents of 0.40 and 0.80 mg/mL, respectively. 24 hours later, the spore suspension was adjusted to 3.5×103 spores/mL, and 2 mL of spore suspension was used to irrigate the seedlings. For curative activity, the sample treatment and spore suspension treatment time were exchanged. Tomato seedlings were treated with spore suspension at first, and then treated with sample after 24 h. Then the seedlings were cultured at 25±3 ℃ and 85% humidity. Each treatment was repeated in 3 groups. Seven days later, the disease index and control efficacy were calculated.
2.6 Morphology changes of F. solani hyphae following C17-fengycin B treatmentScanning electron microscope (SEM) was used to determine the effects of C17-fengycin B on hyphae of F. solani at the ultrastructural level. C17-fengycin B was spotted on a small sterilized filter paper and placed 2 cm away from the edge of the margin of freshly grown F. solani. The hyphae with or without C17-fengycin B treatment was prefixed with 2.5% glutaraldehyde. Fixed cells were rinsed three times for 10 min with 10 mmol/L Phosphate buffer solution (PBS) (pH 7.2 to 7.4). The samples were dehydrated through an ethanol gradient and coated with gold. Hitachi S-3400N scanning electron microscope was used for analysis (Hitachi, Tokyo, Japan).
2.7 Morphology changes of F. solani spores following C17-fengycin B treatmentTo induce the formation of sporangium, six hyphal plugs were cut from a 1–2 week culture dish, covered with sterile distilled water and placed in a growth chamber for 48 h at 25 ℃, and the light intensity was 1 400 lx (Matheron and Porchas, 2000). Then the culture dish was placed at 4 ℃ for 1 h to promote the release of spores. The spores were washed with distilled water and the concentration of spores was checked under microscope. After spores were collected, C17-fengycin B was added into spore suspension (1×105 spores/mL) of equal volume. The final concentration was 0, 0.10, and 0.20 mg/mL, respectively. The spores were incubated at 25 ℃ in dark for 1 h. The germination of spores was examined by transmission electron microscopy. The germination of spores was examined by transmission electron microscopy (TEM) (HT7700 Hitachi).
2.8 Cytotoxicity assayIn-vitro cytotoxicity of C17-fengycin B on LO2 cell line was evaluated by methyl thiazolyl tetrazolium (MTT) assay (Hu et al., 2014). LO2 cells (1×105 cells/well) were inoculated in 96-well flat-bottom culture plate and cultured in incubator for 24 h, and then introduced C17-fengycin B, the concentrations were 0.05, 0.10, and 0.20 mg/mL, individually. 24 hours later, the supernatants were removed, and 100-μL MTT working solution were added into each well. After incubation for 4 h, the formazan crystals were dissolved by MTT stopping buffer for 18 h and determined spectrophotometrically at 550 nm by a microplate reader (Infinite M1000 Pro, TECAN, Mannedorf, Switzerland).
2.9 Statistical analysisRepeat each test three times and take the average result. All the data were expressed by the mean±SD of four independent measurements, and ANOVA was performed by SPSS software (version 18.0 for windows, SPSS, Inc., Chicago, IL, USA). The mean comparison was performed by Duncan's multiple comparison test. Statistical differences were significant at P < 0.05.
2.10 Accession numberThe GenBank accession number for the 16S rRNA gene of B. subtilis 2H11 is MT211278.
3 RESULT AND DISCUSSION 3.1 Antifungal activity of marine bacterium B. subtilis 2H11 against F. solaniIn this study, more than 100 marine cold seep bacteria were screened through antagonistic experiments, and their ability to inhibit the growth of F. solani was evaluated in an order to obtain metabolites with antifungal activity. According to the plate assays, strain 2H11 was proved to significantly inhibit the growth of F. solani (Fig. 1). In addition, the expansion of the edge of F. solani colony was inhibited even when B. subtilis 2H11 was not contacted, which indicated that the metabolites produced by B. subtilis 2H11 could inhibit the growth of F. solani hyphae. By comparing the 16S rRNA sequence with relevant sequences in NCBI website, it was found that the strain had the highest homology with B. subtilis strain JCM1465 (Supplementary Fig. S1), and the sequence similarity reached 99%. Therefore, the marine bacterial strain 2H11 was designated as B. subtilis 2H11.
|
| Fig.1 Antifungal assay of B. subtilis 2H11 (b) against F. solani (a) |
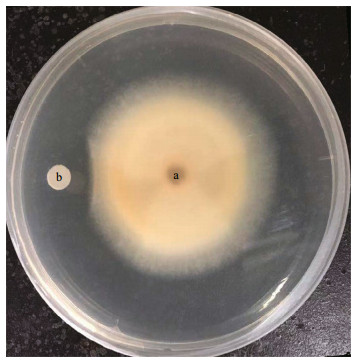
To obtain compound(s) with antifungal activity from B. subtilis 2H11, purification was performed as described in the Materials and Methods. Using F. solani as the indicator fungus, the paper disc method was used to trace the bioactive compound(s). In the RP-HPLC chromatogram (Fig. 2a), we found a single peak related to antifungal activity, indicating that the compound here has inhibitory effect on the growth of F. solani. Then we tested the activity of the compound obtained by RP-HPLC by the filter paper disk test, and found that they had strong antifungal activity against F. solani (Fig. 2b). The chemical structure of the compound was further analyzed and elucidated by mass spectrometry.
|
| Fig.2 Purification and characterization of the active compounds from B. subtilis 2H11 inhibiting the growth of F. solani a. HPLC analysis of the active component from B. subtilis 2H11; b. the inhibitory effect of the active compound from B. subtilis 2H11 on the growth of F. solani mycelia; c. chemical structures and fragmentation patterns of C17-fengycin B, which was isolated from B. subtilis 2H11 as the active compound. |
In the results of HCD-MS and HCD-MS/MS, we found the singly- and doubly-protonated molecular ions at m/z 1 505.85 [M+H]+ and m/z 753.43 [M+2H]2+, respectively (Fig. 3a). Therefore, the molecular weight of the purified active compound was 1 505 Da. It should be noted that the result is consistent with the MS data of fengycin cyclic lipopeptide (CLP) previously reported (Zhang and Sun, 2018). The typical fragmentation ions m/z 994 and 1 108 in the secondary mass spectrometry were caused by the loss of fatty acid-Glu (-398 Da) and fatty acid-Glu-Orn (-512 Da) from the N-terminal segment with a Val residue at position 6 in C17-fengycin B cyclic decapeptide, respectively (Fig. 3b) (Chen et al., 2010; Pecci et al., 2010). The loss of fatty acid-Glu and fatty acid-Glu-Orn usually results in the simultaneous emergence of nine peptide (Orn-Tyr-Thr-Glu-Val-Pro-Glu-Tyr-Ile, m/z 1 108) and octapeptide (Tyr-Thr-Glu-Val-Pro-Glu-Tyr-Ile, m/z 994) as integral fragmentation ions. In conclusion, HCD-MS and HCD-MS/MS spectral data are consistent with the corresponding information of C17-fengycin B (Fig. 2c) (Pecci et al., 2010). Therefore, the antifungal metabolite produced by B. subtilis 2H11 was demonstrated as C17-fengycin B.

|
| Fig.3 Structure elucidation of C17-fengycin B by HCD-MS (a) and HCD-MS/MS (b) |
The secondary metabolites of marine-derived Bacillus have been one of the hotspots in natural products research. Many kinds of compounds have been isolated from their metabolites. Among them, fengycin CLPs have attracted much attention due to their antifungal (Chen et al., 2010; Ma et al., 2014; Nam et al., 2015). In this essay, F. solani, the pathogenic fungus of root rot, was selected as the target. C17-fengycin B was dissolved in dimethyl sulfoxide (DMSO). The original concentration was 0.80 mg/mL. The antifungal results of C17-fengycin B are shown in Fig. 4. The inhibitory effects of C17-fengycin B were compared with those of polymyxin B that is a polypeptide antibiotic and has a strong inhibitory effect on a variety of microorganisms. It has been widely used in many fields, and is a typical lipopeptide antibiotic. Therefore, we set it as the positive control to facilitate comparative analysis.
|
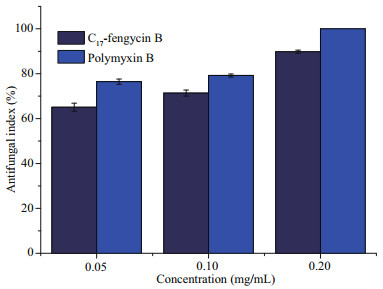
| Fig.4 Antifungal activity of C17-fengycin B and polymyxin B against F. solani |
It was observed that C17-fengycin B exhibited relatively high inhibitory effect against F. solani, and the inhibitory rates were 65.10%, 71.37%, and 89.80% at the concentrations of 0.05, 0.10, and 0.20 mg/mL, respectively. At the same doses, the inhibition rate of positive control polymyxin B was slightly higher than those of C17-fengycin B, and polymyxin B could inhibit the growth of F. solani 100% at 0.20 mg/mL. Some studies have shown that there is a certain relationship between the antifungal activity of lipopeptide antibiotics and their hydrophobicity (Thiericke and Rohr, 1993). Generally, in the RP-HPLC system, the elution time has a positive relationship with the hydrophobicity of molecules, that is, the longer the elution time, the stronger the hydrophobicity performance. Therefore, we conclude that hydrophobic groups in C17-fengycin B may play an important role for the antifungal activity.
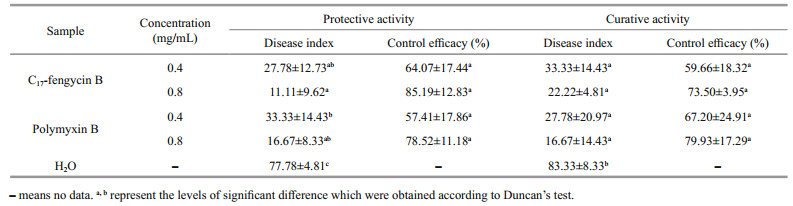
3.4 In-vivo protective and curative activities of C17-fengycin BThe protective and curative activities of C17-fengycin B are shown in Table 1 and Supplementary Fig.S2. The disease indexes of tomato irrigated with C17-fengycin B were significantly lower than that of the negative control group irrigated with water. In addition, the control efficacy of C17-fengycin B increased with its concentrations. At the concentration of 0.8 mg/mL, the protective activity and curative activity of C17-fengycin B were 85.19% and 73.50%, respectively, which were close to those of positive control polymyxin B. There were no obvious pathological changes in the roots of seedlings treated with C17-fengycin B, and the roots were not blackened. Moreover, in contrast to the positive control polymyxin B, the protective activity of C17-fengycin B was higher than that of its curative activity at the same concentration, which indicated that the antifungal activity of C17-fengycin B would not be damaged by the soil and would play a sustainable role in the application.
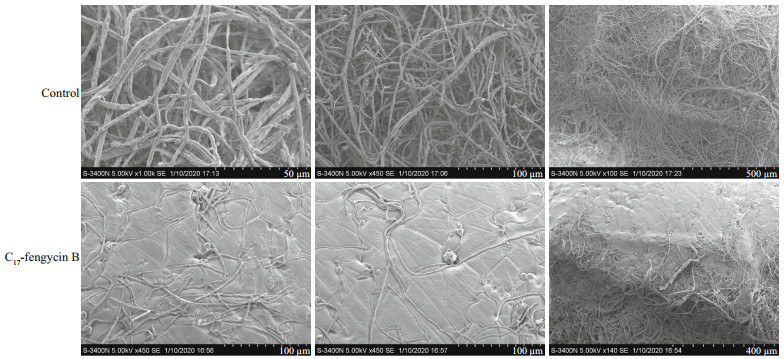
To study the effects of C17-fengycin B on the morphology of F. solani hyphae, SEM observation was conducted. As shown in Fig. 5, the hyphae of F. solani in the control group grew normally, with plump and intact trunks. In contrast, the morphology of hyphae treated with C17-fengycin B was significantly different with the control. Compared with the control group, the amount of hyphae treated with C17-fengycin B was significantly reduced, and the hyphae appeared rough and shriveled.
|
| Fig.5 Effects of C17-fengycin B on the morphology of F. solani hyphae observed by SEM In the control groups, F. solani hyphal cells were treated with methanol (0.04% [v/v]). In the test groups, F. solani hyphal cells were treated with 0.05 mg/mL C17-fengycin B. |
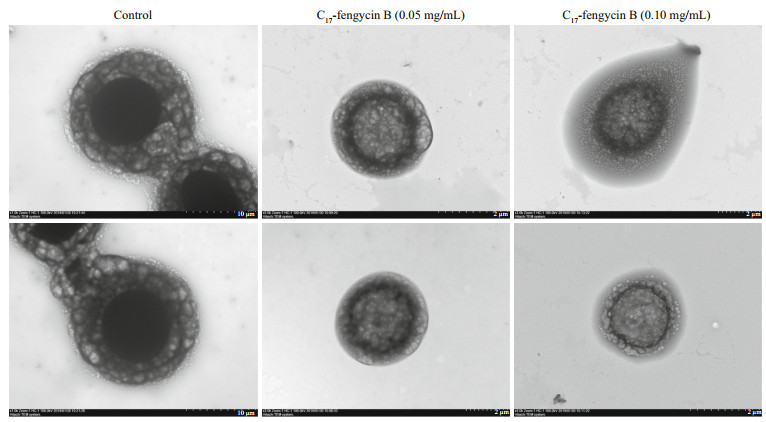
TEM was used to observe the effects of C17-fengycin B on the morphology changes of F. solani spores (Fig. 6). The normal fungal spores have intact and regular cell walls and membranes, with dense cytoplasm and uniform spatial distribution in the intracellular space. However, after C17-fengycin B treatment, the cytoplasm became sparse and light, and the cell membrane became incomplete or even ruptured. At the same time, we also found that higher concentration of C17-fengycin B destroyed spores more seriously, which indicated that the antifungal activity of C17-fengycin B was positively correlated with its concentration. In conclusion, the electron microscopy results showed that C17-fengycin B can damage the cytoplasm and membrane of F. solani and destroy the integrity of cells.
|
| Fig.6 Effects of C17-fengycin B on the morphology of F. solani spores observed by TEM In the control groups, F. solani spores were treated with methanol (0.04% [v/v]). In the test groups, F. solani spores were treated with 0.05 mg/mL and 0.10 mg/mL C17-fengycin B, respectively. |
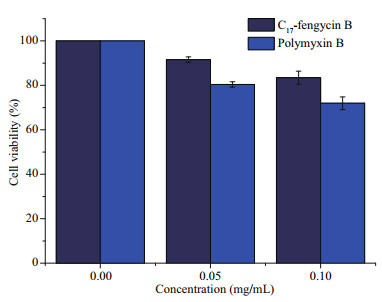
As we all know, most chemical fungicides have strong toxicity. These fungicides are harmful to insects, animals, and human beings. Therefore, the search for new fungicides with low toxicity and high efficiency has always been an important field of pesticide research. At present, most of the studies on the cytotoxicity of fengycin CLPs focus on their antitumor activity, but few on the cytotoxicity of normal animal cells (Yin et al., 2013; Ramachandran et al., 2017; Rofeal and El-Malek, 2020). The results showed that some fengycin CLPs had significant cytotoxicity to human embryonic kidney 293 (HEK293), human internalized keratinocyte (HaCaT), human cervical cancer (Hela), and human type Ⅱ alveolar epithelial (A549) cell lines. In this study, we used LO2 cells as the target to test the cytotoxicity of C17-fengycin B. The effects are listed in Fig. 7, showing that at the concentrations of 0.05 and 0.10 mg/mL, the cell viability of LO2 cells treated with C17-fengycin B was 91.54% and 83.46%, respectively. In addition, at the same concentration, the cytotoxicity of C17-fengycin B was significantly lower than that of positive control polymyxin B. These results indicate that C17-fengycin B has good biocompatibility to LO2 cells and has the potential to develop into an environmental friendly biological fungicide.
|
| Fig.7 The toxicity of C17-fengycin B on LO2 cells |
In this study, the presence of C17-fengycin B (0.05 mg/mL) appeared to have no effect on LO2 cells. Compared with chemical method, microbial fermentation has the advantages of low cost, environment-friendly, and easily scaled up for large scale synthesis. This study demonstrated that C17-fengycin B can be used as a biofungicide to control root rot caused by F. solani. C17-fengycin B showed strong antifungal activity in vitro and in vivo, and had a low cytotoxicity to LO2 cells.
The results can provide a new idea for the control of root rot in the future. The biosynthesis of C17-fengycin B by B. subtilis is an efficient, green, and sustainable method. This will greatly reduce the environmental pollution and pesticide residues caused by chemical pesticide production. However, before developing the agricultural application of C17-fengycin B, it is necessary to do more researches on the antifungal mechanism of C17-fengycin B.
4 CONCLUSIONThis study was conducted to determine the activity of cyclic lipopeptide C17-fengycin B, produced by deep-sea bacterium B. subtilis 2H11, in the suppression of F. solani (pathogen of the tomato root rot disease). The structure of C17-fengycin B was confirmed by HCD-MS and HCD-MS/MS. The antifungal effects of C17-fengycin B against F. solani was evaluated in-vitro and in-vivo. The results show that C17-fengycin B could effectively inhibit the growth of F. solani. At the same dose, the inhibition rate of C17-fengycin B was slightly lower than that of positive control polymyxin B. In addition, a cytotoxicity assay of C17-fengycin B was also performed, and the results show that the cytotoxicity of C17-fengycin B was significantly lower than that of polymyxin B. Moreover, the hyphae and spores of F. solani treated with C17-fengycin B were observed by electron microscope, and the antifungal mechanism of C17-fengycin B was discussed. In conclusion, C17-fengycin B has a good antifungal activity and biocompatibility, it could be a potential candidate for the development of biocontrol pesticide.
5 DATA AVAILABILITY STATEMENTThe data that support the findings are all presented herein.
Electronic supplementary materialSupplementary material (Supplementary Figs.S1–S2) is available in the online version of this article at https://doi.org/10.1007/s00343-020-0215-2.
Al-Mughrabi K I, Vikram A, Peters R D, Howard R J, Grant L, Barasubiye T, Lynch K, Poirier R, Drake K A, Macdonald I K, Lisowski S L I, Jayasuriya K E. 2013. Efficacy of Pseudomonas syringae in the management of potato tuber diseases in storage. Biological Control, 64(3): 315-322.
DOI:10.1016/j.biocontrol.2012.11.011 |
Azizbekyan R R. 2019. Biological preparations for the protection of agricultural plants (Review). Applied Biochemistry and Microbiology, 55(8): 816-823.
DOI:10.1134/S0003683819080027 |
Benaouali H, Hamini-Kadar N, Bouras A, Benichou S L, Kihal M, Henni J E. 2014. Isolation, pathogenicity test and physicochemical studies of Fusarium oxysporum f.sp radicis lycopersici. Advances in Environmental Biology, 8(10): 36-49.
|
Blunt J W, Carroll A R, Copp B R, Davis R A, Keyzers R A, Prinsep M R. 2018. Marine natural products. Natural Product Reports, 35(1): 8-53.
DOI:10.1039/C7NP00052A |
Bonilla-Landa I, de la Cruz O L, Sanchéz-Rangel D, Ortíz-Castro R, Rodriguez-Haas B, Barrera-Méndez F, de León Gómez R E D, Javier Enríquez-Medrano F, Luis Olivares-Romero J. 2018. Design, synthesis and biological evaluation of novel fungicides for the management of Fusarium DieBack disease. Journal of the Mexican Chemical Society, 62(3): 86-98.
|
Carroll A R, Copp B R, Davis R A, Keyzers R A, Prinsep M R. 2020. Marine natural products. Natural Product Reports, 37(2): 175-223.
DOI:10.1039/C9NP00069K |
Chang X L, Dai H, Wang D P, Zhou H H, He W Q, Fu Y, Ibrahim F, Zhou Y, Gong G S, Shang J, Yang J Z, Wu X L, Yong T W, Song C, Yang W Y. 2018. Identification of Fusarium species associated with soybean root rot in Sichuan Province, China. European Journal of Plant Pathology, 151(3): 563-577.
DOI:10.1007/s10658-017-1410-7 |
Chen L L, Wang N, Wang X M, Hu J C, Wang S J. 2010. Characterization of two anti-fungal lipopeptides produced by Bacillus amyloliquefaciens SH-B10. Bioresource Technology, 101(22): 8 822-8 827.
DOI:10.1016/j.biortech.2010.06.054 |
Chittem K, Mathew F M, Gregoire M, Lamppa R S, Chang Y W, Markell S G, Bradley C A, Barasubiye T, Goswami R S. 2015. Identification and characterization of Fusarium spp. associated with root rots of field pea in North Dakota. European Journal of Plant Pathology, 143(4): 641-649.
DOI:10.1007/s10658-015-0714-8 |
Coetzee M P A, Wingfield B D, Wingfield M J. 2018. Armillaria root-rot pathogens: species boundaries and global distribution. Pathogens, 7(4): 83.
DOI:10.3390/pathogens7040083 |
Cui J Q, Wang Y, Han J, Cai B Y. 2016. Analyses of the community compositions of root rot pathogenic fungi in the soybean rhizosphere soil. Chilean Journal of Agricultural Research, 76(2): 179-187.
DOI:10.4067/S0718-58392016000200007 |
da Rosa P D, Ramirez-Castrillon M, Borges R, Aquino V, Fuentefria A M, Goldani L Z. 2019. Epidemiological aspects and characterization of the resistance profile of Fusarium spp. in patients with invasive fusariosis. Journal of Medical Microbiology, 68(10): 1 489-1 496.
DOI:10.1099/jmm.0.001059 |
D'Agostino M, Lemmet T, Dufay C, Luc A, Frippiat J P, Machouart M, Debourgogne A. 2018. Overinduction of CYP51A gene after exposure to azole antifungals provides a first clue to resistance mechanism in Fusarium solani species complex. Microbial Drug Resistance, 24(6): 768-773.
DOI:10.1089/mdr.2017.0311 |
de Rodríguez D J, Hernández-Castillo D, Rodríguez-García R, Angulo-Sánchez J L. 2005. Antifungal activity in vitro of Aloe vera pulp and liquid fraction against plant pathogenic fungi. Industrial Crops and Products, 21(1): 81-87.
DOI:10.1016/j.indcrop.2004.01.002 |
Egamberdieva D, Wirth S J, Shurigin V V, Hashem A, Abd Allah E F. 2017. Endophytic bacteria improve plant growth, symbiotic performance of chickpea (Cicer arietinum L.) and induce suppression of root rot caused by Fusarium solani under salt stress. Frontiers in Microbiology, 8: 1 887.
DOI:10.3389/fmicb.2017.01887 |
Hu W C, Wang G C, Li P X, Wang Y N, Si C L, He J, Long W, Bai Y J, Feng Z S, Wang X F. 2014. Neuroprotective effects of macranthoin G from Eucommia ulmoides against hydrogen peroxide-induced apoptosis in PC12 cells via inhibiting NF-κB activation. Chemico-Biological Interactions, 224: 108-116.
DOI:10.1016/j.cbi.2014.10.011 |
Kobayashi J I. 2016. Search for new bioactive marine natural products and application to drug development. Chemical & Pharmaceutical Bulletin, 64(8): 1 079-1 083.
|
Kordali S, Cakir A, Ozer H, Cakmakci R, Kesdek M, Mete E. 2008. Antifungal, phytotoxic and insecticidal properties of essential oil isolated from Turkish Origanum acutidens and its three components, carvacrol, thymol and p-cymene. Bioresource Technology, 99(18): 8 788-8 795.
DOI:10.1016/j.biortech.2008.04.048 |
Lewis K A, Tzilivakis J, Warner D J, Green A. 2016. An international database for pesticide risk assessments and management. Human and Ecological Risk Assessment: an International Journal, 22(4): 1 050-1 064.
DOI:10.1080/10807039.2015.1133242 |
Li D H, Carr G, Zhang Y H, Williams D E, Amlani A, Bottriell H, Mui A L F, Andersen R J. 2011. Turnagainolides A and B, cyclic depsipeptides produced in culture by a Bacillus sp.: Isolation, structure elucidation, and synthesis. Journal of Natural Products, 74(5): 1 093-1 099.
DOI:10.1021/np200033y |
Ma Z W, Hu J C, Wang X M, Wang S J. 2014. NMR spectroscopic and MS/MS spectrometric characterization of a new lipopeptide antibiotic bacillopeptin B1 produced by a marine sediment-derived Bacillus amyloliquefaciens SH-B74. The Journal of Antibiotics, 67(2): 175-178.
DOI:10.1038/ja.2013.89 |
Matheron M E, Porchas M. 2000. Impact of azoxystrobin, dimethomorph, fluazinam, fosetyl-Al, and metalaxyl on growth, sporulation, and zoospore cyst germination of three Phytophthora spp. Plant Disease, 84(4): 454-458.
DOI:10.1094/PDIS.2000.84.4.454 |
Nam J, Jung M Y, Kim P I, Lee H B, Kim S W, Lee C W. 2015. Structural characterization and temperature-dependent production of C17-fengycin B derived from Bacillus amyloliquefacienssubsp. plantarum BC32-1. Biotechnology and Bioprocess Engineering, 20(4): 708-713.
DOI:10.1007/s12257-015-0350-3 |
Nile A S, Kwon Y D, Nile S H. 2019. Horticultural oils: possible alternatives to chemical pesticides and insecticides. Environmental Science and Pollution Research, 26(21): 21 127-21 139.
DOI:10.1007/s11356-019-05509-z |
Patzke H, Zimdars S, Schulze-Kaysers N, Schieber A. 2017. Growth suppression of Fusarium culmorum, Fusarium poae and Fusarium graminearum by 5-n-alk(en) ylresorcinols from wheat and rye bran. Food Research International, 99: 821-827.
DOI:10.1016/j.foodres.2017.05.022 |
Pecci Y, Rivardo F, Martinotti M G, Allegrone G. 2010. LC/ESI-MS/MS characterisation of lipopeptide biosurfactants produced by the Bacillus licheniformis V9T14 strain. Journal of Mass Spectrometry, 45(7): 772-778.
DOI:10.1002/jms.1767 |
Ramachandran R, Shrivastava M, Narayanan N N, Thakur R L, Chakrabarti A, Roy U. 2017. Evaluation of antifungal efficacy of three new cyclic lipopeptides of the class bacillomycin from Bacillus subtilis RLID 12.1. Antimicrobial Agents and Chemotherapy, 62(1): e01457-17.
DOI:10.1128/AAC.01457-17 |
Ramos L S, Prohaska P G. 1981. Sephadex LH-20 chromatography of extracts of marine sediment and biological samples for the isolation of polynuclear aromatic hydrocarbons. Journal of Chromatography A, 211(2): 284-289.
DOI:10.1016/S0021-9673(00)88048-6 |
Rofeal M, El-Malek A F. 2020. Valorization of lipopeptides biosurfactants as anticancer agents. International Journal of Peptide Research and Therapeutics.
DOI:10.1007/s10989-020-10105-8 |
Sav H, Rafati H, Oz Y, Dalyan-Cilo B, Ener B, Mohammadi F, Ilkit M, van Diepeningen A D, Seyedmousavi S. 2018. Biofilm formation and resistance to fungicides in clinically relevant members of the fungal genus Fusarium. Journal of Fungi (Basel, Switzerland), 4(1): 16.
DOI:10.3390/jof4010016 |
Schroers H J, Samuels G J, Zhang N, Short D P G, Juba J, Geiser D M. 2016. Epitypification of Fusisporium (Fusarium) solani and its assignment to a common phylogenetic species in the Fusarium solani species complex. Mycologia, 108(4): 806-819.
DOI:10.3852/15-255 |
Schut F, De vries E J, Gottschal J C, Robertson B R, Harder W, Prins R A, Button D K. 1993. Isolation of typical marine bacteria by dilution culture: growth, maintenance, and characteristics of isolates under laboratory conditions. Applied and Environmental Microbiology, 59(7): 2 150-2 160.
DOI:10.1128/aem.59.7.2150-2160.1993 |
Shafi J, Tian H, Ji M S. 2017. Bacillus species as versatile weapons for plant pathogens: a review. Biotechnology & Biotechnological Equipment, 31(3): 446-459.
|
Shugart L. 2017. Special Issue: emerging advances and challenges in pesticide ecotoxicology. Ecotoxicology, 26(3): 293-294.
DOI:10.1007/s10646-017-1786-3 |
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. 2013. MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular Biology and Evolution, 30(12): 2 725-2 729.
DOI:10.1093/molbev/mst197 |
Thiericke R, Rohr J. 1993. Biological variation of microbial metabolites by precursor-directed biosynthesis. Natural Product Reports, 10(3): 265-289.
DOI:10.1039/np9931000265 |
Von Schrenk H, Spaulding P. 1902. The bitter rot disease of apples. Science, 16(408): 669-670.
|
Wiggell P, Simpson C J. 1969. Observations on the control of phomopsis root rot of cucumber. Plant Pathology, 18(2): 71-77.
DOI:10.1111/j.1365-3059.1969.tb00469.x |
Xing X Y, Zhao X Y, Ding J, Liu D M, Qi G F. 2018. Enteric-coated insulin microparticles delivered by lipopeptides of iturin and surfactin. Drug Delivery, 25(1): 23-34.
DOI:10.1080/10717544.2017.1413443 |
Yin H P, Guo C L, Wang Y, Liu D, Lv Y B, Lv F X, Lu Z X. 2013. Fengycin inhibits the growth of the human lung cancer cell line 95D through reactive oxygen species production and mitochondria-dependent apoptosis. AntiCancer Drugs, 24(6): 587-598.
DOI:10.1097/CAD.0b013e3283611395 |
Zeigler D R, Nicholson W L. 2017. Experimental evolution of Bacillus subtilis. Environmental Microbiology, 19(9): 3 415-3 422.
DOI:10.1111/1462-2920.13831 |
Zhang L, Sun C. 2018. Fengycins, cyclic lipopeptides from marine Bacillus subtilis strains, kill the plant-pathogenic fungus Magnaporthe grisea by inducing reactive oxygen species production and chromatin condensation. Applied and Environmental Microbiology, 84(18): e00445-18.
DOI:10.1128/aem.00445-18 |
 2021, Vol. 39
2021, Vol. 39