Institute of Oceanology, Chinese Academy of Sciences
Article Information
- YU Yu, LI Yingxia, LI Bing, REN Yichao, DONG Xiaoyu
- Identification and quantification of lead source in sediment in the northern East China Sea using stable lead isotopes
- Journal of Oceanology and Limnology, 39(5): 1887-1900
- http://dx.doi.org/10.1007/s00343-020-0286-0
Article History
- Received Jul. 24, 2020
- accepted in principle Sep. 9, 2020
- accepted for publication Nov. 2, 2020
Lead has attracted a great attention because of its worldwide pollution in the atmosphere caused by the uses of leaded gasoline since the mid-20th century (Nriagu, 1990; Gobeil et al., 1995; Bollhöfer and Rosman, 2000), which has resulted in severely detrimental effect on organisms and human health (Handlogten et al., 2000; Rahman and Singh, 2019). Pb enters environment mainly through mining, ore processing, Pb-acid battery recycling, pipe construction, ammunition, pesticides, pigment in paints, electronic wastes, dyes and ceramic glazes, etc. (Rahman and Singh, 2019). Identification of the sources and pathways of Pb contaminants is important for deep understanding of Pb cycling in modern era.
Sediment in lake or ocean is an ideal recording medium tracking past environmental changes, especially human-induced contamination (Bäckström et al., 2004; Tribovillard et al., 2006). Lead contents and fractions in sediment have revealed an increasing anthropogenic Pb input in the coastal East China Sea (ECS) with rapid economy development in China in the past three decades (Huh and Chen, 1999; Yu et al., 2014). However, the sources of Pb contaminants in the ECS yet remains to be precisely identified, and cannot be specified merely by the total concentrations or chemical fractions of Pb. Lead is present in the environment in four main isotopes: 204Pb (1.04%– 1.65%), 206Pb (20.84%–27.48%), 207Pb (17.62%– 23.65%), and 208Pb (51.28%–56.21%) (Rosman and Taylor, 1998). 206Pb, 207Pb, and 208Pb arise from radioactive decay of 238U, 235U, and 232Th, respectively, while 204Pb is non-radiogenic. The abundance of Pb isotopes in a sample is dependent strictly on the concentrations of Pb, U, and Th as well as the lengths of the decay processes. Different types of ore deposits and anthropogenic sources of Pb have distinct isotope ratios (Cheng and Hu, 2010). In addition, Pb isotopic composition could not be significantly affected by natural physical, chemical, and biological processes (Komárek et al., 2008). Therefore, lead isotope ratios could be used as the "fingerprints" of Pb sources and pathways. It has been widely applied to discriminate Pb sources in lake sediments (Li et al., 2012a), marine sediments (Hinrichs et al., 2002), soils (Ettler et al., 2005), and atmospheric particles (Bollhöfer and Rosman, 2000).
Potential source of Pb could be identified according to the distance between the mixture and the source in iso-space. The proportion of each source could also be calculated by a linear mixing model. For example, in previous studies, the contributions of two Pb sources were calculated through a binary mixing model (Zheng et al., 2004; Choi et al., 2007). However, only n+1 sources can be discriminated through n isotope ratios by the linear mixing model, which is a maximum of four sources if using three Pb isotope ratios (207Pb/206Pb, 208Pb/206Pb, and 206Pb/204Pb). Obviously, it is not enough for tracking multiple Pb sources in an environment. Several stable isotope mixing models, including the IsoSource (Phillips and Gregg, 2003), MixSIR (Moore and Semmens, 2008), SIAR (Parnell et al., 2010), and IsotopeR (Hopkins III and Ferguson, 2012) have been developed to quantify the proportional contributions of various sources in a mixture. These models use iteration method and Bayesian estimation to resolve the problems of larger number of sources and uncertainties (Phillips et al., 2014). These models were usually used to quantify the diet of animals based on δ13C and δ15N in the organisms (Inger et al., 2006; Fortin et al., 2007; Reid et al., 2008). However, there were no reports using these models for Pb isotopes. Herein, we determined the Pb isotope ratios in surface and core sediments in the ECS, and further used a stable isotope mixing model "Simmr" (Parnell et al., 2010, 2013) to quantifiy the proportional contributions of multiple Pb sources. The results would facilitate to identify the sources and pathways of Pb contaminants and reconstruct the history of Pb contamination in the ECS.
2 MATERIAL AND METHOD 2.1 Sample collectionEast China Sea is the largest marginal sea in the western Pacific Ocean, with a vast continental area of 0.5×1012 m2. The Changjiang (Yangtze) River is the major land material source of the ECS, shedding annually 8.9×1011 m3 of fresh water and 3.97×1011 kg of clastics into the ECS (Liu et al., 2007; Yu et al., 2013). In this study, 18 surface sediment samples were collected in the northern ECS (121.0°E–125.5°E, 27.8°N–33.0°N) with a box sampler during a cruise of "Runjiang" in March 2016 (Fig. 1) (Supplementary Table S1). One gravity core (DH3-1, length of 92 cm) was collected in the muddy area along the Zhejiang coast (122.5°E, 29.0°N) using a gravity corer during a cruise of R/V Kexue 3 in October 2015. After sampling, the cored and surface sediment samples were frozen for laboratory analyses. In laboratory, the core was segmented at 2-cm intervals. Aliquots of sediment samples were freeze-dried and then ground to 200 mesh with agate mortar.
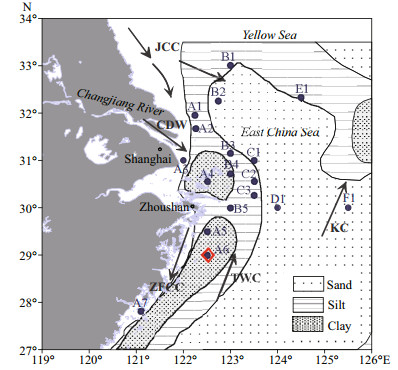
|
| Fig.1 Sampling site of core (DH3-1) and eighteen surface sediments in the northern ECS Dots represent surface sediments; red diamond shows the core sediment sample. Arrows show the regional circulation model (CDW: Changjiang River Dilute Water; JCC: Jiangsu Coastal Current; ZFCC: Zhejiang-Fujian Coastal Current; TWC: Taiwan Warm Current; KC: Kuroshio Current) (Liu et al., 2007). Sediment type is also shown in the figure (Hu and Yang, 2001). |
210Pb activity of sediment samples at 10-cm intervals in core DH3-1 was analyzed to date the core. 210Pb activity was determined by measuring its granddaughter 210Po, which was assumed to be in equilibrium with 210Pb. Spiking with 208Po and selfplating was performed according to Zaborska et al. (2007). After deposition, 210Po and 208Po on the disk were measured using an α spectroscope (YQ-14). The activity of 210Pb in the sample was determined based on chemical recovery by comparing the measured and spiked activities of 208Po. The relative standard deviations (RSDs) of 210Pb activity were less than 10%. The supported 210Pb was calculated as the average of several 210Pb activities in deep layers (70– 92 cm, below the zone of 210Pb exponential decline). A constant initial concentration (CIC) model was used to calculate the sedimentation rate (Robbins and Edgington, 1975). Wet sediment samples were treated with 10% H2O2 and 1 mol/L HCl to remove organic matter and carbonate, then disaggregated by ultrasonic and measured for grain size with a Lase Particle Size Analyzer (Cilas 940L).
2.3 Pb content analysisAll the surface sediment and sliced core samples at 2-cm intervals were analyzed for Pb and Sc content. About 0.05-g ground sediment were digested in airtight Teflon vessel with HNO3-HF-HClO4 mixture at 180 ℃ for 48 h. After digestion, the uncapped vessel was evaporated to dryness at 150 ℃, and then the residue was dissolved with 1∶2 HNO3 and digested again with air-tight vessel at 180 ℃ for 24 h. Then the solution was diluted with 2% HNO3 and analyzed for the concentration of Pb and other trace elements (e.g. Sc) with inductively coupled plasma mass spectrometry (ICP-MS) (Elan DRC II). Two blank samples were performed throughout the experiment, and their trace-element concentrations were below the detection limit of ICP-MS. Analytical precision was assured by triplicate analysis every 8 samples, and the RSDs for Pb and Sc analysis were < 5%. Standard reference materials (GBW07315, GBW07316, BCR-2, and BHVO-2) were used to guarantee the accuracy of the total concentration analysis. The recoveries ranged from 90%–110%.
2.4 Pb isotope analysisStable Pb isotopic compositions in sediments were analyzed in Guangzhou Institute of Geochemistry, Chinese Academy of Sciences. About 20 mg of ground sediment were digested in a polytetrafluoroethylene (PTFE) digestion bomb using a HNO3-HF mixture at 180 ℃ for 24 h. After cooling, the solution was evaporate to dryness on a hot plate, 0.5-mL HBr was added and evaporated again to dryness, and 4-mL 0.8-mol/L HBr was added to dissolve the residue at 135 ℃ for 5 h. The solution was centrifuged at 3 000 r/min for about 3 min and then the supernatant was loaded onto a pre-conditioned AG1-X8 anion exchange column to separate Pb. Pb isotopes were measured using a multi-collector ICP-MS (MC-ICP-MS) (Nu plasma). A Tl spike was added to all samples and standards to correct mass fractionation (White et al., 2000). A lead isotope standard (NIST SRM, 981) was used during the determination for calibration and quality control. The average measured ratios of 206Pb/204Pb, 207Pb/204Pb, and 208Pb/204Pb of NIST 981 were 16.930 6±0.001 0, 15.483 3±0.001 1, and 36.673 5±0.003 3 (2SD (standard deviation), n=11), respectively, close to the certified standard values (16.932 2, 15.485 5, and 36.685 6). A standard material BHVO-2 was analyzed for Pb isotopes to ensure the accuracy of Pb isotope analysis. The determined 206Pb/204Pb, 207Pb/204Pb, and 208Pb/204Pb ratios of BHVO-2 were 18.647 2±0.001 4 (2SD), 15.522 2±0.003 3, and 38.205 7±0.001 9, respectively, consistent with the reported values of 18.647 4±0.024 2, 15.533 4±0.009 4, and 38.236 7±0.018 2 (2SD) (Weis et al., 2006).
2.5 Statistical analysis 2.5.1 Enrichment factor calculationEnrichment factor (EF) of Pb in sediment is calculated as the following:
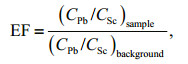 (1)
(1)where (CPb/CSc)sample and (CPb/CSc)background are ratios of Pb content to Sc content in sample and background sediment, respectively. Average contents of Pb and Sc in deep core DH3-1 (60–82 cm) were used as background values.
2.5.2 Two end-member mixing model (keeling plot)According to Keeling (1961), when the samples are mixtures (m) of two component end members with the assumption that one source remain stable (s) and its flux is much higher than the other varied source (v), the following equation could be written according to the mass and isotope balance:
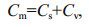 (2)
(2)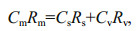 (3)
(3)where C and R denote to concentration and isotope ratios of Pb, and s and v represent riverine and anthropogenic end-member in this study, respectively. The following equation could be deduced to:
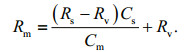 (4)
(4)A linear trend should exist between Rm and 1/Cm, and the isotope ratio of anthropogenic Pb (Rv) was obtained from the intercept of the line. The contributions of riverine and anthropogenic endmember (fs and fv, respectively) were calculated by the following equation:
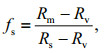 (5)
(5)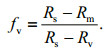 (6)
(6)The contributions of multiple sources of Pb in sediments were quantified through a stable isotope mixing model in Simmr package (version 0.3). This model runs in R 3.5.3 (R Core Team, 2019) based on Pb isotope data in sediments and potential sources. Simmr is designed as an upgrade of the SIAR model that contains a slightly more sophisticated mixing model and uses Just Another Gibbs Sampler (JAGS) to run the model (Parnell et al., 2010, 2013). 207Pb/206Pb, 208Pb/206Pb, and 206Pb/204Pb ratios in sediments were put into the model as the mixtures. Means and SDs of Pb isotope ratios of potential Pb sources were put into the model as the source data. Other parameters in the model such as concentration and correction coefficient were set as default (NULL).
The Simmr model was run separately for core DH3-1 and surface sediment. For core DH3-1, two different data sets were used to run the model: (1) Pb isotope ratios in sediment was used as the mixture of one group and six sources including geogenic and five anthropogenic sources were used as the sources; (2) anthropogenic Pb isotope ratios calculated by the keeling plot was used as the mixture (one group) and five anthropogenic sources were used as the sources. For surface sediments, two different runs were conducted: (1) Pb isotope ratios in thirteen surface sediments were used as the mixture as one group and seven sources including two geogenic (Changjiang River and Huanghe (Yellow) River sediment) and five anthropogenic sources were used as the sources; (2) the same data set but the mixture was set as 13 groups. Every time the model was run with 1×105 iterations and 4 chains, removing 1×104 for burn-in and thinning by 20. Gelman-Rubin convergence diagnostics were conducted to test if the model has run properly. The Gelman diagnostic values were all between 1 and 1.03, indicating satisfactory runs. The posterior distribution for each sources was reported as 95% credible intervals.
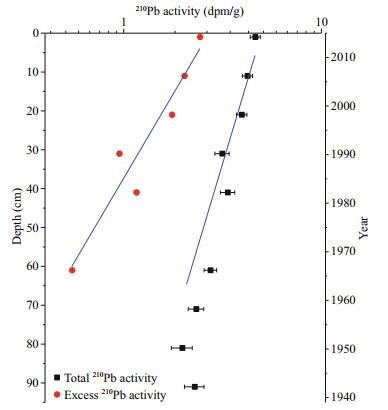
3 RESULT AND DISCUSSION 3.1 Sediment chronologyIn core DH3-1, 210Pb activity decreases exponentially between 0–60 cm (R2=0.92) and reaches its background value of 2.2 dpm/g at 70–92 cm (Fig. 2). A CIC model was used to calculate the sedimentation rate, yielding 1.16 cm/a. It is consistent with previous reports in the same area, e.g. 1.05– 1.11 cm/a based on 137Cs reported by Huh and Su (1999) and 0.98 cm/a reported by Hao et al. (2008). Based on the sedimentation rate of 1.16 cm/a, the core DH3-1 (0–92 cm) is dated from 1939 to 2015 AD.
|
| Fig.2 Depth profiles of total and excess 210Pb activity (dpm/g) in core DH3-1 The x-axis is on a log scale. |
Total Pb content in core DH 3-1 ranges from 27.7 to 35.1 μg/g. Pb content generally remains stable below 30 cm on average of 29.3 μg/g (Fig. 3), but increases dramatically upward above 30 cm (after the 1990s), reaching a maximum of 35.1 μg/g in 2–4 cm. Pb content rises by 19.8% from 1990s to 2010s. It is worth noting that there is a sharp decrease in Pb content in 4–10 cm. It might result from the obvious reduction in clay content and larger d50 at this depth (Supplementary Fig.S1).
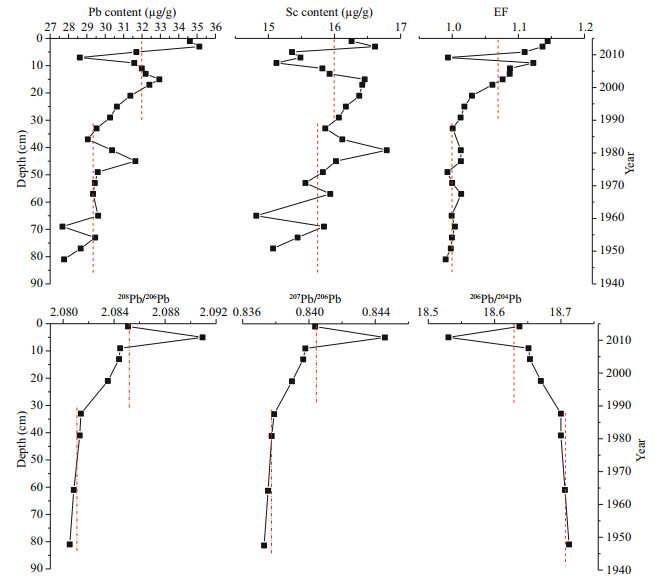
|
| Fig.3 Vertical profiles of Pb and Sc content, EF of Pb, and Pb isotopic ratios in the core DH3-1 Red dashed lines represent the average level in the upper (0-34 cm) and lower (34-82 cm) sections of the core, respectively. EF: enrichment factor. |
Sc is a conservative element and oceanic Sc mainly originates from terrestrial lithogenic input (Otosaka et al., 2004). It is related to the natural occurring part of Pb as it is strongly correlated with fine particles (RSc-clay= 0.391, RSc-d50=-0.458, P < 0.05, n=29) and Pb content in core sediment (RSc-Pb=0.577, P < 0.01, n=29). Sc meets the standards of a reference element (N'guessan et al., 2009) and has been successfully used as reference element in previous studies (Zhang et al., 2007; Yu et al., 2013). Therefore, Sc was used as reference in EF calculation. EF of Pb generally remains stable at 1 in deep sediment (Fig. 3), but markedly increases upward above 34 cm (after the late 1980s), reaching 1.14 in surface.
207Pb/206Pb, 208Pb/206Pb, and 206Pb/204Pb in core DH3-1 range between 0.837–0.845, 2.081–2.091, and 18.530–18.712, respectively (Supplementary Table S2). Generally, 206Pb/204Pb exhibits opposite distribution trend to 207Pb/206Pb and 208Pb/206Pb (Fig. 3). They all remain stable below 32 cm with average values of 0.838, 2.081, and 18.705, respectively. These values were used as the background Pb isotope values of core DH3-1. The major material source of the coastal muddy patch is the Changjiang River sediment. Therefore, these values were also deemed as the background Pb isotope compositions of the Changjiang River sediment. They are in agreement with the reported background values of 0.839 and 2.081 for 207Pb/206Pb, and 208Pb/206Pb, respectively, deduced from the Changjiang River Pb line by Choi et al. (2007).
In the upper core (0–32 cm), 207Pb/206Pb and 208Pb/206Pb dramatically increase while 206Pb/204Pb decreases (Fig. 3). The increasing trend of Pb content, EF, and isotope ratios in the upper core suggest a rise of anthropogenic Pb input after the late 1980s. From the 1980s to 2000s, the number of cars grew with economic development, and leaded gasoline was widely used in China during the period (Cheng and Hu, 2010). The combustion of leaded gasoline with high 207Pb/206Pb ratios (0.902±0.006) discharged high concentration of Pb into environment and increased 207Pb/206Pb and 208Pb/206Pb in sediment (Chen et al., 2005; Li et al., 2012a). Except for the abnormally high values in 4–6 cm, the increasing of 207Pb/206Pb and 208Pb/206Pb slowed down in 0–14 cm (early 2000s to 2015). It is probably due to the phase out of leaded gasoline in the early 2000s in China, and after when coal combustion with relatively low 207Pb/206Pb ratios (0.862±0.010) (Mukai et al., 2001) became the major source of Pb contaminants (Li et al., 2012b). Leaded gasoline has remarkable high Pb isotope ratios (Chen et al., 2005), but 207Pb/206Pb and 208Pb/206Pb in the core during 1990–2000 is not so high. It is due to low proportion of anthropogenic Pb before 2000 suggested by low EFs of Pb (1.00–1.03 during 1900–2000).
At the beginning of the 2000s, Pb content dropped slightly, but EFs of Pb remained stable and continued to increase afterwards. Li et al. (2012b) has reported a sharp decline in total Pb emission in China at the beginning of 2000s due to the phase out of leaded gasoline. However, this sharp decline has not been recorded in sediment, probably because that Pb in sediment was not only controlled by its external input but also by its depositional processes which is impacted by many factors such as hydrodynamic conditions and organic matter (OM). The influence of hydrodynamic conditions was eliminated by normalization to Sc, and thus EF is used to reflect the anthropogenic and biogenic input of Pb. Primary productivity in the coastal sea increased due to eutrophication in recent decades. OM deposition is increasing with eutrophication in the coastal sea in recent decades (Wang et al., 2003; Yu et al., 2012), leading to increasing Pb deposition combined with OM and Pb EFs in sediment (Yu et al., 2014). Therefore, probably due to the impact of OM, Pb concentration and EF remained at a higher level around 2000 in sediment.
3.3 Pb contents and isotopic compositions in surface sedimentsPb content in surface sediments ranges between 19.3 and 47.3 μg/g with an average of 27.5 μg/g. The highest Pb content is observed in the areas beyond the estuary. Pb content gradually declines towards the outer shelf (Fig. 4). This variation is consistent with the distribution of mud content and d50 which was reported by Yu et al. (2013). The seaward decreasing trend of fine particle and Pb content results from the dispersal of the Changjiang River sediment (Liu et al., 2007), as well as dynamic conditions such as the upwellings.

|
| Fig.4 Spatial distributions of Pb content (μg/g), EF, and Pb isotope ratios (207Pb/206Pb, 208Pb/206Pb, and 206Pb/204Pb) in surface sediments of the northern ECS |
207Pb/206Pb, 208Pb/206Pb, and 206Pb/204Pb ratios in surface sediments range from 0.838 to 0.844, from 2.081 to 2.097 and from 18.549 to 18.705, respectively (Supplementary Table S2). Opposite to Pb content, 207Pb/206Pb and 208Pb/206Pb ratios increase from the estuary toward the outer shelf (Fig. 4). This distribution of Pb isotope ratios is likely related to atmospheric input of anthropogenic Pb. Atmosphere is the major initial recipient of anthropogenic Pb (Komárek et al., 2008). For PM10 in the atmosphere of Shanghai, Pb EF is as high as 674 and 207Pb/206Pb and 208Pb/206Pb ratios reach up to 0.861±0.002 and 2.105±0.005, respectively (Zheng et al., 2004). They are remarkably higher than Pb EF (2.02) (Fig. 4) and isotope ratios (0.844 and 2.089 for 207Pb/206Pb and 208Pb/206Pb, respectively) in estuary sediments (site A3 in Fig. 1). From the estuary to the outer shelf, riverine sediment input decreases and thus the proportion of atmospheric Pb input grows larger, leading to increase in Pb isotope ratios in sediment. Likewise, a southeastward increase in 207Pb/206Pb and 208Pb/206Pb in area northeast to the estuary is due to the decreasing input of terrestrial sediment, which is from the abandoned Huanghe River delta by the southeastward Jiangsu Coastal Current (JCC) and has much lower 207Pb/206Pb and 208Pb/206Pb ratios (0.840 and 2.090, respectively) than atmospheric particles (Choi et al., 2007; Hu et al., 2012).
3.4 Two end-member mixing modelIsotope diagrams of 208Pb/206Pb vs. 207Pb/206Pb and 206Pb/204Pb vs. 207Pb/206Pb in the surface and core sediments of the ECS and other potential Pb sources are shown in Fig. 5. It is shown that ECS sediments are very close to Huanghe River sediment and loess, but are much lower in 208Pb/206Pb and 207Pb/206Pb and higher in 206Pb/204Pb than anthropogenic sources. Most of the sediment samples are located on the Chinese Pb line except five surface sediments (sites F1, D1, B5, E1, and A1 in Fig. 1) in 208Pb/206Pb vs. 207Pb/206Pb diagram. The deviation of these samples is likely related to the influence of anthropogenic input. It is shown that except cement and Pb mining and smelting, other potential sources of Pb are located on Chinese Pb line. In 206Pb/204Pb vs. 207Pb/206Pb diagram, cement and Pb mining and smelting are located on different sides of the line with approximately equal distances to the line. Hence, if cement and Pb mining and smelting contribute equally to marine sediment, sediment samples would still be on the line. However, in 208Pb/206Pb vs. 207Pb/206Pb diagram, cement, and Pb mining and smelting are located on the same side of the line. Their mixture will lead to deviation of sediment from the line. More importantly, these surface sediments are located in relict sedimentation area with few modern material input (Shen and Pan, 2011; Zhu et al., 2011) (Fig. 1). The relative contribution of anthropogenic sources to these sediments is much bigger than that to sediment with more geogenic material input, thus lead to larger deviations of these samples from the line.

|
| Fig.5 Lead isotope ratio diagrams for core and surface sediments in the ECS and potential Pb sources Chinese coal and Pb line are from Mukai et al. (2001); Pbanth-calculated is obtained from keeling plot of Pb isotopes in core sediments. References of other Pb sources are listed in Table 1. |
Linear regression between Pb isotope ratios in sediments and Sc/Pb was conducted. Isotope data at 4–6 cm in core DH3-1 were excluded from the regression because of its obvious deviation. Regression results show that the linear correlation between 207Pb/206Pb, 208Pb/206Pb, and 206Pb/204Pb and Sc/Pb in core DH3-1 are significant (R2=0.930, 0.891, and 0.926, respectively), but the correlation is weak for surface sediment (R2=0.025–0.087) (Supplementary Fig.S2). Thus, sedimentary Pb in core DH3-1 could be considered as the mixture of two sources, which are the geogenic and anthropogenic sources. Anthropogenic Pb isotopic compositions (Pbanth-calculated) were obtained from the intercept of the line according to Eq.4, which are 0.859, 2.114, and 18.180 for 207Pb/206Pb, 208Pb/206Pb, and 206Pb/204Pb, respectively. It is located close to PM10 in Shanghai atmosphere (Fig. 5), verifying that atmospheric input is the major input pathway of anthropogenic Pb in the ECS. The proportions of the two end-members in each layer were calculated according to Eqs.5 and 6 (Supplementary Table S3). The results show that anthropogenic Pb increased from 2.8% to 12.6% of total Pb since the late 1990s, while geogenic Pb decreased from 97.2% to 87.4%.
It is found that surface sediment does not obey the two-end member mixing model in view of weak correlation between Pb isotopes and Sc/Pb. The model assumes that there are two end members of Pb, one of which is stable and large and the other is variant and small (Keeling, 1961). The former end member is geogenic source in the ECS, which mainly consists of the Changjiang River sediment and the old Huanghe River delta sediment. The contribution of these two geogenic sources remains stable for core sediments but vary with site for surface sediments. Therefore, surface sediments do not meet the assumption of the model, but core sediments do.
3.5 Simmr modelling for core sedimentsAccording to the Annual Statistic Report on Environment in China (2010–2015), 77.9 t of Pb contaminants was discharged annually through waste water in 2015, 81% of which was from nonferrous metal mining and smelting industry. Li et al. (2012b) has reported that total atmospheric lead emission in China ranged from 3 900 t to 14 700 t from 1990 to 2009, among which vehicle gasoline combustion, coal combustion, non-ferrous metal smelting, cement production, iron and steel industry were the major contributors. The amount of atmospheric Pb emission is far larger than Pb discharge in wastewater, confirming that atmosphere is the major transport pathway of Pb contaminant (Li et al., 2012b).
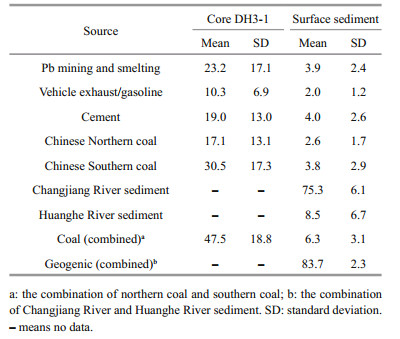
Simmr model was used to quantify the contributing proportions of potential Pb sources to sedimentary Pb in the ECS. Five anthropogenic Pb sources were considered, including Pb mining and smelting, Chinese northern coal, Chinese southern coal, cement production, and vehicle exhaust/gasoline combustion. Their Pb isotopic data were from previous reports (Table 1). Pb isotopes for Pb mining and smelting were the averages of three metallurgic dust sample from Shanghai and two Pb-Zn mines from Hunan Province (Zheng et al., 2004; Shan et al., 2019). Hunan Province is located in the middle reaches of the Changjiang River and is the largest non-ferrous metal production area in China. The lithogenic part of the Changjiang River and Huanghe River sediment were considered as two major geogenic sources of Pb in the ECS. Background Pb isotopic ratios in Huanghe River sediment were obtained from deep sediment of a core from the Huanghe River delta reported by Hu et al. (2012).
There are two ways to run the model for core sediment: one is using Pbanth-calculated isotope ratios as the mixture the other is using Pb isotopes in sediment as the mixture. Both methods were conducted and they yielded comparable results (Supplementary Table S4). In order to focus on the anthropogenic sources, the results of the former method are discussed (Fig. 6). Number in the lower left diagonal of the matrix plots are the correlation coefficients between the sources. Small correlations (the maximum of -0.46) indicate that all the sources were finely distinguished from each other (Supplementary Fig.S3). The order of the contribution is southern coal > Pb mining and smelting > cement > northern coal > vehicle exhaust/gasoline in their means (Table 2), accounting for 3.3%–66.4%, 2.3%–65.6%, 2.1%–54.1%, 1.9%–50.7%, and 1.4%– 27.5% of anthropogenic Pb, respectively (Supplementary Table S4). The ranges of the estimated proportions are relatively wide, due to large SDs of the source, especially cement. Southern coal is relatively poorly constrained, with wide shape of density plot (Supplementary Fig.S3). If we combine the southern coal and northern coal into one source named "coal", the result is much more meaningful. Coal plays a main role in anthropogenic sources, accounting for 47.5%±18.8% of anthropogenic Pb.
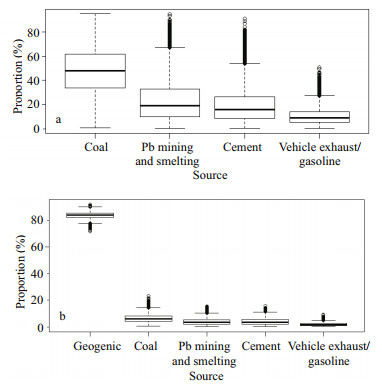
|
| Fig.6 Proportion (%) of each source to anthropogenic Pb in core DH3-1 (a) and to the total Pb in surface sediment (b) |
|
Pb isotope ratios in surface sediments were used as the mixture, and seven sources including five anthropogenic and two geogenic sources were used to run the model. The results show that the Changjiang River is the largest contributor, accounting for 61.1%– 84.0% of total Pb. The Huanghe River sediment only accounts for 0.7%–24.2% of total Pb (Supplementary Table S5). The correlation coefficient between the Changjiang River and Huanghe River sediment is as high as -0.94, indicating that these two sources are indistinguishable (Supplementary Fig.S3). Therefore, the subsequent analysis combined them into one named "geogenic". Northern coal and southern coal were also combined into "coal" to enhance the constraint of the result.
The distributions of five end-members are quite narrow (Supplementary Fig.S3), indicating finely constrained solutions. Geogenic source is the largest contributor to Pb in surface sediments (78.9%– 88.0%). High proportion of lithogenic input is reasonable because of the discharge of huge amount of Changjiang River sediment, which is mainly of lithogenic origin with low contamination extent (Müller et al., 2008). It is supported by low Pb EF in sediments (1.16–2.02 in surface and 0.99–1.14 in core sediment). This phenomenon is also found in the Huanghe River estuary and adjacent waters, which are affected by large quantity of Huanghe River sediment and are low in Pb EF in sediment (0.7–1.3) (Hu et al., 2015a, b ). Anthropogenic sources account for 12.0%–21.1% of total Pb, among which coal is the largest anthropogenic source, accounting for 1.8%– 13.7% of total sedimentary Pb and averagely 38.6% of anthropogenic Pb. Pb mining and smelting, cement, and vehicle exhaust contribute averagely 23.9%, 24.5%, and 12.3% of anthropogenic Pb, respectively. These results are comparable with those in core sediment. The relatively low proportions of vehicle exhaust/gasoline proves that the major contamination source of atmospheric Pb in China has been converted from vehicle exhaust to coal combustion after the phase-out of leaded gasoline since the beginning of 2000s (Zheng et al., 2004; Chen et al., 2005). The proportions of cement and vehicle exhaust obtained in this study are higher than those reported by Li et al. (2012b), which reported about 6% and 4.5% in 2009 based on emission factor of these sources all over China, respectively. Different proportions obtained by this study and Li et al. (2012b) are probably resulted from the following reasons. Firstly, the contribution of iron and steel industry to Pb emission was not considered in this study based on their Pb isotopic characteristics. Chen et al. (2005) reported 207Pb/206Pb in iron ore and iron ore sinter dust in Shanghai to be 0.744±0.002 and 0.825±0.002, respectively. They are far from Pb isotope ratios in sediments and Pbanthcalculated ratios, suggesting that the contribution of this source might be neglected. However, Li et al. (2012b) pointed that iron and steel industry contributed around 4% of total Pb in 2009 and it became the fifth-highest source of anthropogenic Pb in China. Our model did not consider iron and steel production and other small sources such as waste incineration and oil combustion, and thus resulted in higher proportions of cement and vehicle exhaust. Secondly, spatial variations of these sources would also contribute to this inconsistency, because the study of Li et al. (2012b) is based on Pb emission in whole China. However, as Li et al. (2012b) pointed that, though small, the contributions of iron and steel production, waste incineration and oil combustion were increasing annually. Therefore, further research is needed to investigate the contribution of these sources, their Pb isotopic characteristics and their impacts on Pb isotopes in environment as well.
It is worth noting that Pb contaminant in atmosphere is mainly from industrial dust and fume emission, to which coal combustion by power and heat plant, Pb mining and smelting and cement production are three largest contributors in China (Annual Statistic Report on Environment in China, 2010–2015). However, only the dust and fume discharge load is currently monitored and controlled, but there is no control on Pb concentration in the dust and fume (Annual Statistic Report on Environment in China, 2010–2015; GB 13223-2011). Therefore, considering high volatility of Pb and severe atmospheric contamination, it is urgently needed to monitor Pb discharge by dust and fume from these industries to control Pb emission into environment.
The contributing proportions of different sources might vary with sites, thus the proportions at each site were calculated as well and their spatial distributions were drawn based on the means (Fig. 7). It is shown that the proportion of geogenic source gradually decreases while the proportions of anthropogenic sources increase from the coast towards the outer shelf. This confirms that the Changjiang River sediment is the major geogenic source of Pb in the ECS, whose impact decreases towards the outer shelf, while atmospheric input is the primary anthropogenic source whose influence grows larger from coast to outer shelf.
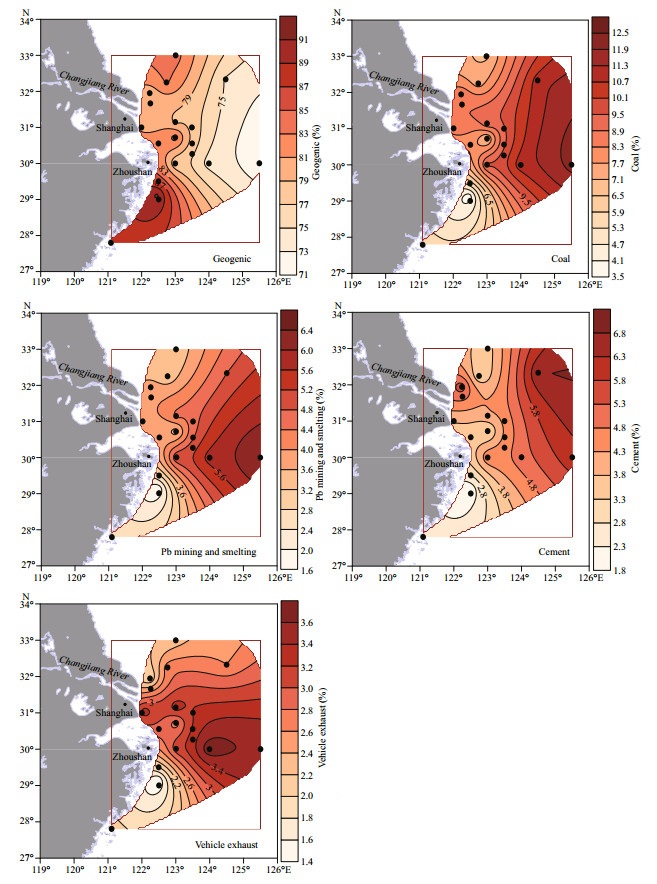
|
| Fig.7 Spatial distributions of the mean proportion (%) of each source The data are obtained from the Simmr modelling on the condition that each site was set as one group. |
Pb stable isotopes and contents in core and surface sediments in the northern ECS were determined to identify the sources of Pb, especially anthropogenic sources. Gradual increase in 207Pb/206Pb and 208Pb/206Pb in the upper core indicates the rising anthropogenic input of Pb after the late 1980s. Natural occurring part of riverine sediment (the Changjiang River and eroded old Huanghe River delta) is the major lithogenic source of Pb in the ECS, which mainly influence the inner shelf and area northeast to the estuary. Atmospheric input is the major anthropogenic source of Pb in the ECS, which causes high 207Pb/206Pb and 208Pb/206Pb ratios in sediment of the outer shelf. The Simmr modelling results show that 12.0%–21.1% of sedimentary Pb is from anthropogenic source, among which coal combustion is the largest contributor (47.5%±18.8%), and Pb mining and smelting, cement, and vehicle exhaust contribute 23.2%±17.1%, 19.0%±13.0%, and 10.3%±6.9%, respectively. Although the range of estimated proportion in this study is relatively large due to larger SDs of anthropogenic sources, the Simmr model is superior to two or three end-member linear mixing model and provides a deeper insight into the contribution and variation of multiple Pb sources in ocean. This study raises the alarm to reduce coal consumption in China, as well as to monitor and control Pb discharge into atmosphere by industrial dust and fume emission.
5 DATA AVALABILITY STATEMENTThe datasets analyzed during the current study are available from the corresponding author on reasonable request.
Bäckström M, Karlsson S, Allard B. 2004. Metal teachability and anthropogenic signal in roadside soils estimated from sequential extraction and stable lead isotopes. Environmental Monitoring and Assessment, 90(1-3): 135-160.
DOI:10.1023/B:EMAS.0000003572.40515.31 |
Bollhöfer A, Rosman K J R. 2000. Isotopic source signatures for atmospheric lead: the southern Hemisphere. Geochimica et Cosmochimica Acta, 64(19): 3 251-3 262.
DOI:10.1016/S0016-7037(00)00436-1 |
Chen J M, Tan M G, Li Y L, Zhang Y M, Lu W W, Tong Y P, Zhang G L, Li Y. 2005. A lead isotope record of shanghai atmospheric lead emissions in total suspended particles during the period of phasing out of leaded gasoline. Atmospheric Environment, 39(7): 1245-1253.
DOI:10.1016/j.atmosenv.2004.10.041 |
Cheng H F, Hu Y A. 2010. Lead (Pb) isotopic fingerprinting and its applications in lead pollution studies in China: a review. Environmental Pollution, 158(5): 1134-1146.
DOI:10.1016/j.envpol.2009.12.028 |
Choi M S, Yi H I, Yang S Y, Lee C B, Cha H J. 2007. Identification of Pb sources in Yellow Sea sediments using stable Pb isotope ratios. Marine Chemistry, 107(2): 255-274.
DOI:10.1016/j.marchem.2007.07.008 |
Ettler V, Vaněk A, Mihaljevič M, Bezdička P. 2005. Contrasting lead speciation in forest and tilled soils heavily polluted by lead metallurgy. Chemosphere, 58(10): 1449-1459.
DOI:10.1016/j.chemosphere.2004.09.084 |
Fortin J K, Farley S D, Rode K D, Robbins C T. 2007. Dietary and spatial overlap between sympatric ursids relative to salmon use. Ursus, 18(1): 19-29.
DOI:10.2192/1537-6176(2007)18[19:DASOBS]2.0.CO;2 |
Gobeil C, Johnson W K, Macdonald R W, Wong C S. 1995. Sources and burden of lead in St. Lawrence estuary sediments: isotopic evidence. Environmental Science & Technology, 29(1): 193-201.
|
Handlogten M E, Shiraishi N, Awata H, Huang C F, Miller R T. 2000. Extracellular Ca2+-sensing receptor is a promiscuous divalent cation sensor that responds to lead. American Journal of Physiology-Renal Physiology, 279(6): F1 083-F1 091.
DOI:10.1152/ajprenal.2000.279.6.F1083 |
Hao Y C, Guo Z G, Yang Z S, Fan D J, Fang M, Li X D. 2008. Tracking historical lead pollution in the coastal area adjacent to the Yangtze River Estuary using lead isotopic compositions. Environmental Pollution, 156(3): 1 325-1 331.
DOI:10.1016/j.envpol.2008.02.023 |
Hinrichs J, Dellwig O, Brumsack H J. 2002. Lead in sediments and suspended particulate matter of the German Bight: natural versus anthropogenic origin. Applied Geochemistry, 17(5): 621-632.
DOI:10.1016/S0883-2927(01)00124-X |
Hopkins III J B, Ferguson J M. 2012. Estimating the diets of animals using stable isotopes and a comprehensive Bayesian mixing model. PLoS One, 7(1): e28478.
DOI:10.1371/journal.pone.0028478 |
Hu B Q, Li G G, Li J, Bi J Q, Zhao J T, Bu R Y. 2012. Provenance and climate change inferred from Sr-Nd-Pb isotopes of late Quaternary sediments in the Huanghe (Yellow River) Delta, China. Quaternary Research, 78(3): 561-571.
DOI:10.1016/j.yqres.2012.07.005 |
Hu D X, Yang Z S. 2001. Key processes of ocean flux in the East China Sea. China Ocean Press, Beijing, China. (in Chinese)
|
Hu N J, Huang P, Zhang H, Zhu A M, Liu J H, Zhang J, He L H. 2015a. Anthropogenic Pb input into Bohai Bay, China: evidence from stable Pb isotopic compositions in sediments. Continental Shelf Research, 109: 188-197.
DOI:10.1016/j.csr.2015.09.004 |
Hu N J, Peng H, Liu J H, Shi X F, Ma D Y, Zhu A M, Zhang J, Hui Z, He L H. 2015b. Tracking lead origin in the Yellow River Estuary and nearby Bohai Sea based on its isotopic composition. Estuarine, Coastal and Shelf Science, 163: 99-107.
DOI:10.1016/j.ecss.2015.06.010 |
Huh C A, Chen H Y. 1999. History of lead pollution recorded in East China Sea sediments. Marine Pollution Bulletin, 38(7): 545-549.
DOI:10.1016/S0025-326X(98)00111-8 |
Huh C A, Su C C. 1999. Sedimentation dynamics in the East China Sea elucidated from 210Pb, 137Cs and 239, 240Pu. Marine Geology, 160(1-2): 183-196.
DOI:10.1016/S0025-3227(99)00020-1 |
Inger R, Ruxton G D, Newton J, Colhoun K, Robinson J A, Jackson A L, Bearhop S. 2006. Temporal and intrapopulation variation in prey choice of wintering geese determined by stable isotope analysis. Journal of Animal Ecology, 75(5): 1 190-1 200.
DOI:10.1111/j.1365-2656.2006.01142.x |
Keeling C D. 1961. The concentration and isotopic abundances of carbon dioxide in rural and marine air. Geochimica et Cosmochimica Acta, 24(3-4): 277-298.
DOI:10.1016/0016-7037(61)90023-0 |
Komárek M, Ettler V, Chrastný V, Mihaljevič M. 2008. Lead isotopes in environmental sciences: a review. Environment International, 34(4): 562-577.
DOI:10.1016/j.envint.2007.10.005 |
Li H B, Yu S, Li G L, Deng H. 2012a. Lead contamination and source in Shanghai in the past century using dated sediment cores from urban park lakes. Chemosphere, 88(10): 1 161-1 169.
DOI:10.1016/j.chemosphere.2012.03.061 |
Li Q, Cheng H G, Zhou T, Lin C Y, Guo S. 2012b. The estimated atmospheric lead emissions in China, 1990-2009. Atmospheric Environment, 60: 1-8.
DOI:10.1016/j.atmosenv.2012.06.025 |
Liu J P, Xu K H, Li A C, Milliman J D, Velozzi D M, Xiao S B, Yang Z S. 2007. Flux and fate of Yangtze River sediment delivered to the East China Sea. Geomorphology, 85(3-4): 208-224.
DOI:10.1016/j.geomorph.2006.03.023 |
Moore J W, Semmens B X. 2008. Incorporating uncertainty and prior information into stable isotope mixing models. Ecology Letters, 11(5): 470-480.
DOI:10.1111/j.1461-0248.2008.01163.x |
Mukai H, Tanaka A, Fujii T, Zeng Y Q, Hong Y T, Tang J, Guo S, Xue H S, Sun Z L, Zhou J T, Xue D M, Zhao J, Zhai G H, Gu J L, Zhai P Y. 2001. Regional characteristics of sulfur and lead isotope ratios in the atmosphere at several Chinese urban sites. Environmental Science & Technology, 35(6): 1 064-1 071.
|
Müller B, Berg M, Yao Z P, Zhang X F, Wang D, Pfluger A. 2008. How polluted is the Yangtze River? Water quality downstream from the Three Gorges Dam. Science of the Total Environment, 402(2-3): 232-247.
DOI:10.1016/j.scitotenv.2008.04.049 |
N'guessan Y M, Probst J L, Bur T, Probst A. 2009. Trace elements in stream bed sediments from agricultural catchments (Gascogne region, S-W France): where do they come from?. Science of the Total Environment, 407(8): 2 939-2 952.
DOI:10.1016/j.scitotenv.2008.12.047 |
Nriagu J O. 1990. The rise and fall of leaded gasoline. Science of the Total Environment, 92: 13-28.
DOI:10.1016/0048-9697(90)90318-O |
Otosaka S, Honda M C, Noriki S. 2004. La/Yb and Th/Sc in settling particles: vertical and horizontal transport of lithogenic material in the western North Pacific. Geochemical Journal, 38(6): 515-525.
DOI:10.2343/geochemj.38.515 |
Parnell A C, Inger R, Bearhop S, Jackson A L. 2010. Source partitioning using stable isotopes: coping with too much variation. PLoS One, 5(3): e9672.
DOI:10.1371/journal.pone.0009672 |
Parnell A C, Phillips D L, Bearhop S, Semmens B X, Ward E J, Moore J W, Jackson A L, Grey J, Kelly D J, Inger R. 2013. Bayesian stable isotope mixing models. Environmetrics, 24(6): 387-399.
|
Phillips D L, Gregg J W. 2003. Source partitioning using stable isotopes: coping with too many sources. Oecologia, 136(2): 261-269.
DOI:10.1007/s00442-003-1218-3 |
Phillips D L, Inger R, Bearhop S, Jackson A L, Moore J W, Parnell A C, Semmens B X, Ward E J. 2014. Best practices for use of stable isotope mixing models in food-web studies. Canadian Journal of Zoology, 92(10): 823-835.
DOI:10.1139/cjz-2014-0127 |
R Core Team. 2019. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria.
|
Rahman Z, Singh V P. 2019. The relative impact of toxic heavy metals (THMs) (arsenic (As), cadmium (Cd), chromium (Cr) (VI), mercury (Hg), and lead (Pb)) on the total environment: an overview. Environmental Monitoring and Assessment, 191(7): 419.
DOI:10.1007/s10661-019-7528-7 |
Reid D J, Quinn G P, Lake P S, Reich P. 2008. Terrestrial detritus supports the food webs in lowland intermittent streams of south-eastern Australia: a stable isotope study. Freshwater Biology, 53(10): 2 036-2 050.
DOI:10.1111/j.1365-2427.2008.02025.x |
Robbins J A, Edgington D N. 1975. Determination of recent sedimentation rates in Lake Michigan using Pb-210 and Cs-137. Geochimica et Cosmochimica Acta, 39(3): 285-304.
DOI:10.1016/0016-7037(75)90198-2 |
Rosman K J R, Taylor P D P. 1998. Isotopic compositions of the elements 1997 (Technical Report). Pure and Applied Chemistry, 70(1): 217-235.
DOI:10.1351/pac199870010217 |
Shan L, Jiang J S, Kang B, Wang J, Long W G, Ke X Z, Liu J J, Xu D M, Niu Z J. 2019. Metallogenic material sources of important non-ferrous metal deposits in northeastern Hunan Province: evidence from S and Pb isotopes. Geological Bulletin of China, 38(5): 884-900.
(in Chinese with English abstract) |
Shen H T, Pan D A. 2011. Turbidity Maximum in the Changjiang Estuary. China Ocean Press, Beijing, China. (in Chinese)
|
Tribovillard N, Algeo T J, Lyons T, Riboulleau A. 2006. Trace metals as paleoredox and paleoproductivity proxies: an update. Chemical Geology, 232(1-2): 12-32.
DOI:10.1016/j.chemgeo.2006.02.012 |
Wang B D, Wang X L, Zhan R. 2003. Nutrient conditions in the Yellow Sea and the East China Sea. Estuarine, Coastal and Shelf Science, 58(1): 127-136.
DOI:10.1016/S0272-7714(03)00067-2 |
Weis D, Kieffer B, Maerschalk C, Barling J, De Jong J, Williams G A, Hanano D, Pretorius W, Mattielli N, Scoates J S, Goolaerts A, Friedman R M, Mahoney J B. 2006. High-precision isotopic characterization of USGS reference materials by TIMS and MC-ICP-MS. Geochemistry, Geophysics, Geosystems, 7(8): Q08006.
|
White W M, Albarède F, Télouk P. 2000. High-precision analysis of Pb isotope ratios by multi-collector ICP-MS. Chemical Geology, 167(3-4): 257-270.
DOI:10.1016/S0009-2541(99)00182-5 |
Yu Y, Song J M, Duan L Q, Li X G, Yuan H M, Li N. 2014. Sedimentary trace-element records of natural and human-induced environmental changes in the East China Sea. Journal of Paleolimnology, 52(4): 277-292.
DOI:10.1007/s10933-014-9793-3 |
Yu Y, Song J M, Li X G, Duan L Q. 2012. Geochemical records of decadal variations in terrestrial input and recent anthropogenic eutrophication in the Changjiang Estuary and its adjacent waters. Applied Geochemistry, 27(8): 1 556-1 566.
DOI:10.1016/j.apgeochem.2012.05.002 |
Yu Y, Song J M, Li X G, Yuan H M, Li N. 2013. Fractionation, sources and budgets of potential harmful elements in surface sediments of the East China Sea. Marine Pollution Bulletin, 68(1-2): 157-167.
DOI:10.1016/j.marpolbul.2012.11.043 |
Zaborska A, Carroll J, Papucci C, Pempkowiak J. 2007. Intercomparison of alpha and gamma spectrometry techniques used in 210Pb geochronology. Journal of Environmental Radioactivity, 93(1): 38-50.
DOI:10.1016/j.jenvrad.2006.11.007 |
Zhang H B, Luo Y M, Wong M H, Zhao Q G, Zhang G L. 2007. Defining the geochemical baseline: a case of Hong Kong soils. Environmental Geology, 52(5): 843-851.
DOI:10.1007/s00254-006-0526-4 |
Zheng J, Tan M G, Shibata T Y, Tanaka A, Li Y, Zhang G L, Zhang Y M, Shan Z C. 2004. Characteristics of lead isotope ratios and elemental concentrations in PM10 fraction of airborne particulate matter in Shanghai after the phase-out of leaded gasoline. Atmospheric Environment, 38(8): 1 191-1 200.
DOI:10.1016/j.atmosenv.2003.11.004 |
Zhu C, Wang Z H, Xue B, Yu P S, Pan J M, Wagner T, Pancost R D. 2011. Characterizing the depositional settings for sedimentary organic matter distributions in the Lower Yangtze River-East China Sea Shelf System. Estuarine, Coastal and Shelf Science, 93(3): 182-191.
DOI:10.1016/j.ecss.2010.08.001 |
 2021, Vol. 39
2021, Vol. 39



