Institute of Oceanology, Chinese Academy of Sciences
Article Information
- LIN Pengcheng, HU Huaming, LIU Fei, LI Mingzheng, LIU Huanzhang
- Reproductive characteristics of Psilorhynchus homaloptera Hora and Mukerji, 1935 (Cyprinidae: Psilorhynchidae) in the lower Yarlung Zangbo River, Tibet
- Journal of Oceanology and Limnology, 39(4): 1513-1522
- http://dx.doi.org/10.1007/s00343-020-0224-1
Article History
- Received Jun. 8, 2020
- accepted in principle Jul. 22, 2020
- accepted for publication Oct. 5, 2020
2 Key Laboratory of Aquatic Biodiversity and Conservation of Chinese Academy of Sciences, Chinese Academy of Sciences, Wuhan 430072, China
Psilorhynchus, the only genus of the family Psilorhynchidae, is distributed in the lower reach of the Yarlung Zangbo River in Tibet, China, the north and northeast of the Brahmaputra drainage in India, east Nepal, and the Ayeyarwady drainage of north Myanmar, adjacent to China (Yue, 2000; Conway and Mayden, 2007; Arunachalam and Muralidharan, 2008). Species of Psilorhynchus, commonly known as torrent minnows or torrent stone carp, are generally small, have an arched back and flattened ventral surface, and live in mountainous rivers and streams with turbulent flows. So far, studies on this genus mainly covered its distribution, taxonomy, and phylogenetic position through morphology, osteology, and molecular analyses (Yue, 2000; Conway and Mayden, 2007; He et al., 2008; Conway, 2011). Biological studies of Psilorhynchus are extremely limited. Conway et al. (2014) found a remarkable phenomenon of swimbladder sexual dimorphism in Psilorhynchus, wherein the male swimbladder is much larger and more complex than that of the female. Kaushik and Bordoloi (2017) reported the lengthweight relationships of Psilorhynchus balitora, and Li et al. (2018) and Dey et al. (2018) reported the length-weight relationships of Psilorhynchus homaloptera.
As the only species of Psilorhynchus distributed in China, Psilorhynchus homaloptera occurs in the Yarlung Zangbo-Brahmaputra River basin, living preferably in high-gradient streams with rocky bottom in the Himalayas (Talwar and Jhingran, 1991; Yue, 2000; Shangningam and Vishwanath, 2014). This species mainly ingests algae and aquatic invertebrates (Yue and Chen, 1998). Due to overexploitation and hydro-engineering construction, P. homaloptera has been listed as an endangered species in the China Red Data Book (Yue and Chen, 1998). Understanding of the biological background—especially the reproductive characteristics, including sexual maturity, spawning period, and fecundity—is urgently needed for the population conservation and management of the fish species (Angermeier, 1995). However, little biological information has been published on this species, which further hinders the conservation efforts.
Under this backdrop, we studied the reproductive characteristics of P. homaloptera collected in the Motuo reach of the lower Yarlung Zangbo River. The aims of this study were to identify the following characteristics of the species: (1) size at sexual maturity (TL50); (2) the spawning period according to gonad development and the size distribution of oocytes; and (3) the absolute fecundity, relative fecundity, and regression analysis of relationships among total length, total weight, ovary weight, age, and fecundity. At last, proposals and suggestions were given to the conservation and management for P. homaloptera in the region.
2 MATERIAL AND METHOD 2.1 Study area and fish collectionSampling was conducted in Motuo County, where the Yarlung Zangbo River flows across south Tibet and breaks through Yarlung Zangbo Grand Canyon, the deepest canyon in the world. Rivers and streams are in high gradient, and the substrates are mostly composed of cobbles and boulders with low proportions of gravel and sand. It is a corridor for warm, humid air currents from southeast Bengal Bay in the Indian Ocean to penetrate the Himalayas and Tibetan Plateau, so the climate in the canyon region is humid, with a mean annual air temperature of 16 ℃ and the annual precipitation of over 2 200 mm/a (Zhu et al., 2013).
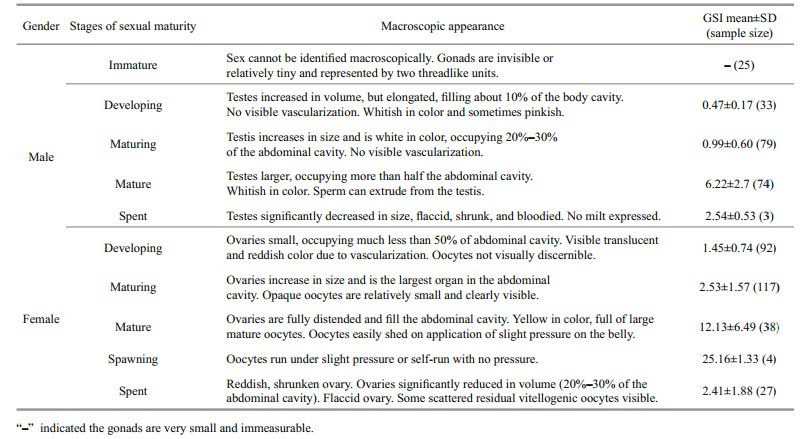
In total, 801 P. homaloptera individuals were collected from December 2015 to November 2016 in the lower Yarlung Zangbo River and its tributaries (Figs. 1 & 2). Sampling was not conducted in January or February 2016 because snowstorms made it inaccessible to Motuo. The specimens were captured using backpack electro-fishing gear and killed with MS-222. The total length (TL) of each captured individual was measured to the nearest 1 mm, and the total weight (W) and gonadal weight (WG) were measured to the nearest 0.01 g. Sex was determined macroscopically and the gonadal development stages were classified based on macroscopic features (Table 1). Water temperature at sampling locations was measured with thermometer in situ and monthly accumulated rainfall data were obtained from the Motuo Meteorological Bureau.

|
| Fig.1 Psilorhynchus homaloptera (107 mm in TL and 7.8 g in W) collected from the lower Yarlung Zangbo River in the study |

|
| Fig.2 Points of sampling for Psilorhynchus homaloptera in the lower Yarlung Zangbo River from December 2015 to November 2016 |
|
The size at sexual maturity was defined as the length at which 50% of the population reached sexual maturity (Lt50), and this definition was applied to estimate maturity macroscopically for each sex (Somerton, 1980). Gonad development in females and males were classified into six stages according to macroscopic gonad development characteristics of the common Cyprinidae fish in Brulé et al. (2003) and Huo et al. (2013). A logistic formula for determining the proportion (P) of mature fish was applied to assess the size at 10-mm (TL) size intervals: P=(1+e-k(TLmid–TL50))-1 (Chen and Paloheimo, 1994), where TLmid is the midpoint of the TL class, TL50 is the mean TL at sexual maturity, P is the proportion of mature fish, and k is the slope.
2.3 Spawning periodThe spawning period was evaluated through comprehensive consideration of monthly variations in the gonado-somatic index (GSI), monthly proportions of macroscopic maturity stages, and the size distribution of oocytes.
The GSI of mature individuals was calculated using the equation GSI=WG/W×100 (Brewer et al., 2008). ANOVA followed by Tukey's post hoc test was applied to test whether there were significant differences in GSI among months (Zar, 1999; Ma et al., 2012).
2.4 FecundityTo study individual fecundity, 34 females were processed using the gravimetric method (Bagenal and Braum, 1978). Subsamples from anterior, middle, and posterior parts of the ovaries were sampled. Fecundity was estimated as F=n×W/w, where n is the number of oocytes in the subsamples, W is the weight of ovaries, and w is the weight of sample parts. To assess absolute fecundity, all oocytes that had begun vitellogenesis were counted as potentially ripe eggs. The relative fecundity was calculated as the number of ripe oocytes per gram of total weight (Adebisi, 1987). Regression analysis was applied to study the relationships between the TL, W, ovary weight (Wo), age (A), and fecundity of P. homaloptera. Age was estimated from scales.
2.5 Statistical analysisThe data were described as means±standard deviation (SD). Data and images were processed using Microsoft Excel 2010 and OriginPro 2016. SPSS software (version 16.0) was used for statistical analyses, and P < 0.05 was considered significantly different.
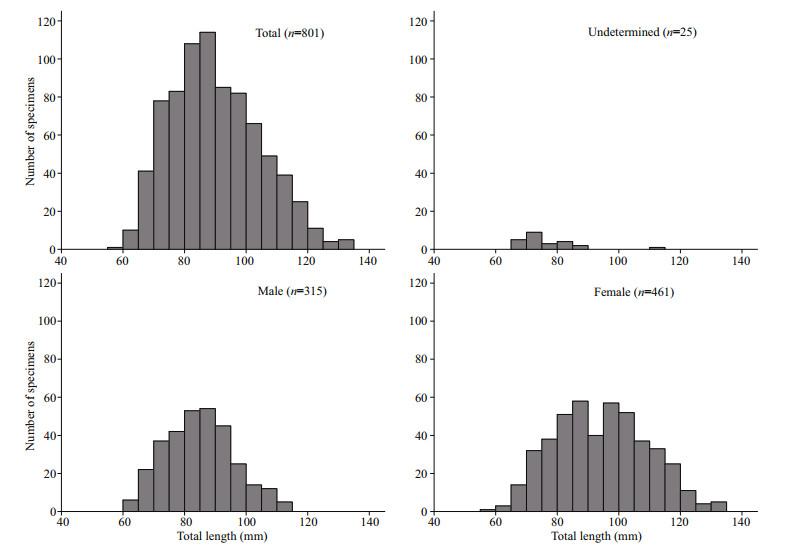
3 RESULT 3.1 Length-frequency distribution and sex ratioThe total length of P. homaloptera specimens ranged from 57 to 134 mm, and the total weight ranged from 1.7 to 18.6 g. Of the 801 individuals caught, 315 were males, with 60–113 mm in TL; 461 were females, with 57–134 mm in TL; and 25 were undetermined, with 61–111 mm in TL (Fig. 3). Lengthfrequency distributions were significantly different between males and females (Kolmogorov-Smirnov test, Z=4.215, P < 0.001). The number of males (289) in the < 100-mm TL class was nearly equal to that of females (301), while there were more females (160) in the > 100-mm TL class than there were males (26) (Fig. 3).
|
| Fig.3 Length-frequency distribution for female (n =461), male (n =315), and unsexed (n =25) Psilorhynchus homaloptera captured in the lower Yarlung Zangbo River |
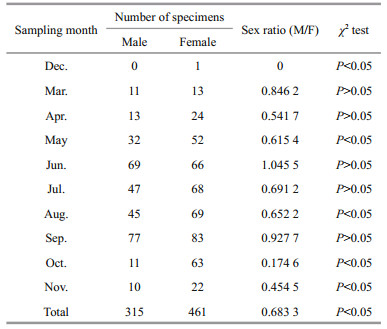
The male: female sex ratio (M/F) was 0.68∶1 for the population, which was significantly different from 1∶1 (χ2=27.469, P < 0.05, Table 2). There were no significant differences in the M/F from the expected 1∶1 ratio in March, April, June, July or September (P > 0.05), while the sex ratio was strongly biased towards females during the other months (P < 0.05).
|
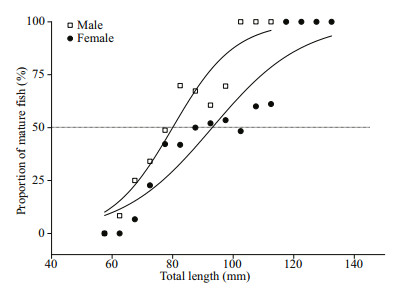
The size at sexual maturity revealed distinct differences between males and females (Fig. 4). Estimated TL50 values were 80.0 mm for males and 93.1 mm for females. The logistic functions are expressed as follows: P=(1+e-0.097(TLmid–80.0))-1 (n=315, R2=0.914) for males and P=(1+e-0.067(TLmid–93.1))-1 (n=461, R2=0.879) for females. The observed minimum total length of mature individuals was 65 mm for males and 75 mm for females.
|
| Fig.4 Logistic functions for Psilorhynchus homaloptera fitted to the cumulative percentage of sexually mature individuals in relation to size The solid line showed the mean total length at 50% maturity. |
In the September, GSI showed the highest values (females: 11.45; males: 7.49), a relatively high percentage of mature stages (females: 50.60%; males: 88.31%), and the highest egg size (mean oocyte diameter: 1.928±0.219 mm) (Figs. 5–7). The GSI of September was significantly higher than that of any other month (ANOVA, Tukey's post hoc, P < 0.05). Spawning ovaries occurred in September. Spent ovaries and testicles both appeared from October to November (Fig. 6). The mean oocyte diameter (1.928±0.219 mm) in September was significantly higher than those between July and August (Fig. 7, ANOVA, Tukey's post hoc, P < 0.05). GSI revealed an increasing tendency in May and August for both sexes (Fig. 5a). Mean oocyte diameter also showed an increasing trend in July and October (Fig. 7). Therefore, we concluded that P. homaloptera is a single spawner that spawns between September and October, as indicated by mean monthly GSI values, maturity stages, and mean oocyte diameter. During August and September, monthly water temperature and rainfall reached the maximum (Fig. 5b).
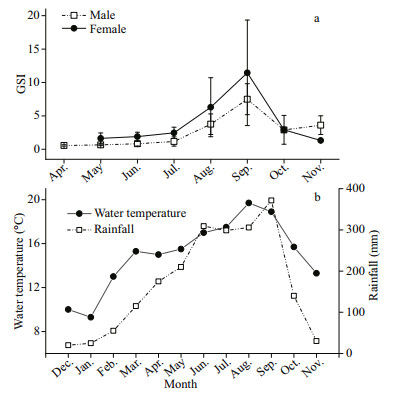
|
| Fig.5 Monthly gonado-somatic index (GSI) of Psilorhynchus homaloptera males and females and the monthly water temperature and rainfall in the lower Yarlung Zangbo River a. from April to November 2016; b. from January to November 2016. GSI values are expressed as means±standard deviation. |
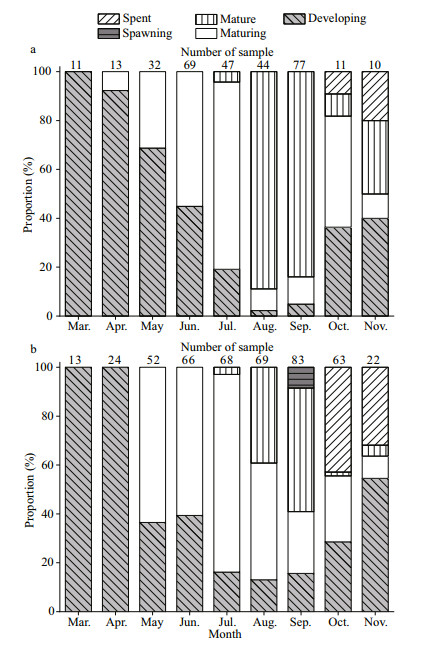
|
| Fig.6 Monthly distribution of macroscopic gonad maturity stages in Psilorhynchus homaloptera fish from December 2015 to November 2016 a. male; b. female. |
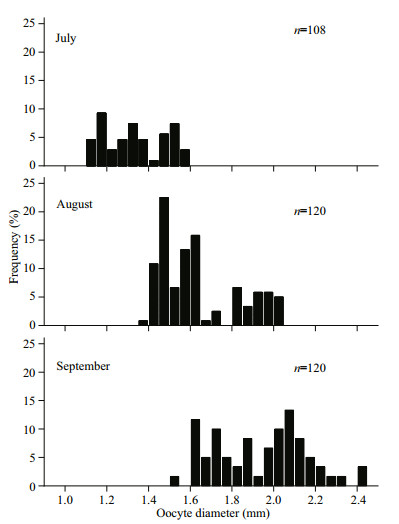
|
| Fig.7 Monthly size-frequency of Psilorhynchus homaloptera oocytes from July to September 2016 |
The mean absolute fecundity was 557±204 eggs per fish, with 191–1 007 eggs per fish and 95–134 mm in TL. The relative fecundity was calculated as 50.9±10.9 eggs per g of female weight, with 24.2– 73.8 eggs per g of female weight.
The fecundity of P. homaloptera increased linearly with the increasing total length, total weight, ovary weight, and age (Fig. 8), and the fitted regression equations were as follows:
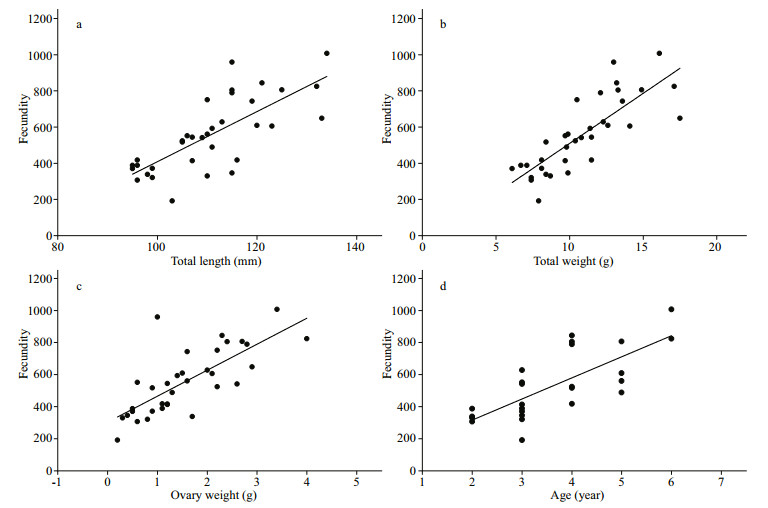
|
| Fig.8 Relationships among total length (a), total weight (b), ovary weight (c), age (d), and fecundity of Psilorhynchus homaloptera |
Total length: F=-976.648+13.855TL, n=34, R2=0.554;
Total weight: F=-44.359+55.393W, n=34, R2=0.653;
Ovary weight: F=314.878+110.603W0, n=34, R2=0.539;
Age: F=53.937+131.435A, n=25, R2=0.550.
Total weight was more highly correlated with fecundity than total length, ovary weight, or age were.
4 DISCUSSIONReproductive biology is necessary to comprehend life history and stock assessment. It can also be important for understanding why some species might be more susceptible to exploitation from fisheries or other environmental stresses and essential for population conservation and management planning (Cortés, 2000). However, information on the biological features of P. homaloptera from the lower Yarlung Zangbo River remains deficient. These data will help fill current gaps regarding the biology and reproductive strategy of this species.
In the present study, the predominance of females in the overall population and the population of larger adults reflects females' greater longevity. Similar results were observed in Schizothoracinae fishes (Schizothorax o'connori, Schizothorax waltoni, Schizothorax curvilabiatus) and Garra tibetana from the Yarlung Zangbo River (Ma et al., 2012; Zhou et al., 2015; Wang, 2018; Gong, 2019) and other subtropical fishes (Lemes et al., 2017; Zaniboni-Filho et al., 2017). This phenomenon may be related to factors such as growth and mortality rates, migration, physiological changes affected by temperature change, feeding regime, and reproductive cycle (Zaniboni-Filho et al., 2017).
Reproductive effort of a fish can be measured as the egg mass per female, which is the product of fecundity and egg size (Duarte and Alcaraz, 1989). Investigating fecundity and egg size allows for the examination of energetic trade-offs between reproduction and survival. The relative fecundity of P. homaloptera varied from 24.2 to 73.8 eggs per g of female weight, with a mean value of 50.9±10.9 eggs per g of female weight. This value is slightly higher than that of other species in the lower Yarlung Zangbo River. Wang (2018) and Gong (2019) show that the relative fecundity of Schizothorax curvilabiatus and Garra tibetana are 5–14 and 7–24 eggs per gram, respectively. Moreover, it can be concluded that the relative fecundity of P. homaloptera is much lower than those of other cyprinids in the adjacent waters, e.g., 163 in the common carp Cyprinus carpio (Sivakumaran et al., 2003), 198 in Labeo gonius from Brahmaputra (Chowdhary and Shahid, 2018), 275 in Barilus bendelisis from Garhwal Himalaya (Saxena et al., 2016), and 692 in a widespread cyprinid fish Hemiculter leucisculus from Alma-Gol wetlands (Patimar et al., 2008).
Egg diameter also is an important factor for determining fish reproductive strategy. Most developed oocytes of P. homaloptera are 1.9 mm in diameter. Coburn (1986) reported that the egg sizes of 71 species of eastern North American cyprinids ranged from approximately 0.7 mm—in several species of Notropis, Hybognathus hankinsoni, and Hybopsis aestivalis—to 2.0 mm in Campostoma anomalum. Therefore, the reproductive traits of P. homaloptera are characterized by relatively low fecundity and large eggs. Large eggs generally have greater fitness than do smaller ones in a given environment. Thus, the trade-off between relatively low fecundity and large eggs can increase the offspring fitness, since more energy is invested in each offspring (Wootton, 1998). Large eggs enable the production of relatively larger and well-developed larvae. These individuals have a greater ability to swim against currents in hillstreams, giving them access to a wider range of potential prey (McDowall, 2009). These characteristics are associated with the equilibrium strategy guild proposed by Winemiller (1995), which largely coincides with the K strategy for adapting to low-resource habitats. The species in this guild make great efforts to ensure the survival of their offspring by providing them with enough yolk and/or by way of direct parental care during the early stages of life (Winemiller, 1995; Wootton, 1998).
The estimation of GSI and egg diameter distribution of P. homaloptera revealed that the fish spawns once a year, mainly from September and October, indicating that P. homaloptera is a single spawner with a short spawning period. These results are confirmed by the back-calculation of otolith daily increments from P. homaloptera larva that we captured in November (unpublished data). Many studies indicate that freshwater fish spawning could be affected by riverine physical or chemical conditions, such as photoperiod (Jones and Petreman, 2015), water temperature (Chang et al., 2017), water discharge (King et al., 2016), dissolved oxygen (Sear et al., 2014), and climate (Strüssmann et al., 2010). In the present study, the final maturation and ovulation of P. homaloptera seem to be well synchronized with the highest water temperature and rainfall, which occurs in August and September (Fig. 5). Heavy rainfall causes flooding, and this with the increase in water temperature have been shown to have a positive influence on gonadal recrudescence in many riverine fishes. In the Murray River, Australia, golden and silver perch (Macquaria ambigua, Bidyanus bidyanus) exhibit more spawning activities during the flood period, when water temperatures are above 20 ℃; furthermore, fish native to this region may show the highest spawning outcomes a) when the magnitude of floods increases following lower antecedent flow conditions and b) at water temperatures of 18–20 ℃ (King et al., 2009, 2016).
In addition, the spawning period of P. homaloptera is different from those of Schizothorax curvilabiatus and Garra tibetana (February to April) in the lower Yarlung Zangbo River (Wang, 2018; Gong, 2019). This may be explained by the food limitation hypothesis (Kramer, 1978; Le Pape and Bonhommeau, 2015). As a transitional zone from the Tibetan Plateau to Indian peninsula, the Motuo reach has a humid subtropical climate with a high altitude gradient. During the rainy season, flooding and the consequent fast current may reduce algal biomass, regardless of nutrient enrichment, and further reduce food availability and quality. After that (autumn), the moist forest habitat attracts insects and fruits fall into the river, so the standing crops of benthic algae may increase when they are not scoured by rainy season floods (Kramer, 1978). Therefore, P. homaloptera spawning in autumn may be controlled by the juvenile food availability.
Above all, P. homaloptera's unique reproduction tactics may be attributed to the tradeoff between investing in higher quality, larger larvae and reducing their quantity to adapt the low-food-resource and volatile environmental conditions. Accordingly, reasonable conservation measures are needed to maintain the fish stock for future generations, as P. homaloptera is being heavily threatened in the wild. Minimum landing size limits can be set to allow juvenile fish to survive long enough to reproduce. Another measure is to close this region to fishing during the spawning period and establish a nature reserve to protect the spawning ground. Moreover, long-term monitoring is essential to getting more detailed information on the life history traits of P. homaloptera and maintaining the species' stock in the natural habitat.
5 CONCLUSIONOverall, the present study indicates that P. homaloptera, an important protected species in the Yarlung Zangbo River, is autumn-spawning fish whose spawning occurs between September and October. Males and females are 65 and 75 mm, respectively, at sexual maturity. The fecundity of the species is low and its eggs are large. These reproduction tactics can be regarded as an adaptation to the riverine conditions in the lower Yarlung Zangbo River. In view of our results, reasonable seasonal and space fishing management strategies for this species should be established. Long-term ecological studies are also necessary to reveal the life history traits for P. homaloptera.
6 DATA AVAILABILITY STATEMENTThe datasets generated and/or analyzed from the current study are available from the corresponding author upon reasonable request.
Adebisi A A. 1987. The relationships between fecundities, gonadosomatic indices and egg sizes of some fishes of Ogun River, Nigeria. Archiv für Hydrobiologie, 111: 151-156.
|
Angermeier P L. 1995. Ecological attributes of extinction-prone species: loss of freshwater fishes of Virginia. Conservation Biology, 9(1): 143-158.
DOI:10.1046/j.1523-1739.1995.09010143.x |
Arunachalam M, Muralidharan M. 2008. Description of a new species of the genus Psilorhynchus (Teleostei: Psilorhynchidae) from a Western Ghat stream in Southern India. Raffles Bulletin of Zoology, 56(2): 405-414.
|
Bagenal T B, Braum E. 1978. Eggs and early life history. In: Bagenal T B ed. Methods for Assessment of Fish Production in Fresh Waters. Blackwell Scientific Publishing, Oxford. p. 165–201.
|
Brewer S K, Rabeni C F, Papoulias D M. 2008. Comparing histology and gonadosomatic index for determining spawning condition of small-bodied riverine fishes. Ecology of Freshwater Fish, 17(1): 54-58.
DOI:10.1111/j.1600-0633.2007.00256.x |
Brulé T, Déniel C, Colás-Marrufo T, Renán X. 2003. Reproductive biology of gag in the southern Gulf of Mexico. Journal of Fish Biology, 63(6): 1505-1520.
DOI:10.1111/j.1095-8649.2003.00263.x |
Chang T, Gao X, Danley P D, Lin P, Li M, Liu H. 2017. Longitudinal and temporal water temperature patterns in the Yangtze River and its influence on spawning of the Chinese sturgeon (Acipenser sinensis Gray 1835). River Research and Applications, 33(9): 1145-1151.
DOI:10.1002/rra.3210 |
Chen Y, Paloheimo J E. 1994. Estimating fish length and age at 50% maturity using a logistic type model. Aquatic Sciences, 56(3): 206-219.
DOI:10.1007/BF00879965 |
Chowdhary S T, Shahid I. 2018. Maturity and breeding of Labeo gonius (Ham. ) from Brahmaputra. Journal of Biosphere, 7: 44-54.
|
Coburn M M. 1986. Egg diameter variation in Eastern North American minnows (Pisces: Cyprinidae): correlation with vertebral number, habitat and spawning behavior. The Ohio Journal of Science, 86(3): 110-120.
|
Conway K W, Britz R, Siegel D S. 2014. Different on the inside: extreme swimbladder sexual dimorphism in the South Asian torrent minnows. Biology Letters, 10(7): 20140348.
DOI:10.1098/rsbl.2014.0348 |
Conway K W, Mayden R L. 2007. The gill arches of Psilorhynchus (Ostariophysi: Psilorhynchidae). Copeia, 2007(2): 267-280.
DOI:10.1643/0045-8511(2007)7[267:TGAOPO]2.0.CO;2 |
Conway K W. 2011. Osteology of the South Asian Genus Psilorhynchus McClelland, 1839 (Teleostei: Ostariophysi: Psilorhynchidae), with investigation of its phylogenetic relationships within the order Cypriniformes. Zoological Journal of the Linnean Society, 163(1): 50-154.
DOI:10.1111/j.1096-3642.2011.00698.x |
Cortés E. 2000. Life history patterns and correlations in sharks. Reviews in Fisheries Science, 8(4): 299-344.
DOI:10.1080/10408340308951115 |
Dey A, Choudhury H, Basumatary S, Bharali R C, Sarma D. 2018. Length-weight relationships of three freshwater fish species from the Kameng River (Brahmaputra basin) in Arunachal Pradesh, northeast India. Journal of Applied Ichthyology, 34(4): 1002-1003.
DOI:10.1111/jai.13635 |
Duarte C M, Alcaraz M. 1989. To produce many small or few large eggs: a size-independent reproductive tactic of fish. Oecologia, 80(3): 401-404.
DOI:10.1007/BF00379043 |
Gong Z. 2019. Taxonomic revision and ecological adaptation study of species of genus Garra in the lower Yarlung Tsangpo River. Institute of Hydrobiology, Chinese Academy of Sciences, Wuhan.
|
He S P, Gu X, Mayden R L, Chen W J, Conway K W, Chen Y Y. 2008. Phylogenetic position of the enigmatic genus Psilorhynchus (Ostariophysi: Cypriniformes): evidence from the mitochondrial genome. Molecular Phylogenetics and Evolution, 47(1): 419-425.
DOI:10.1016/j.ympev.2007.10.012 |
Huo B, Xie C X, Ma B S, Yang X F, Huang H P. 2013. Reproductive biology of Oxygymnocypris stewartii in the Yarlung Zangbo River in Tibet, China. Environmental Biology of Fishes, 96(4): 481-493.
DOI:10.1007/s10641-012-0031-4 |
Jones N E, Petreman I C. 2015. Environmental influences on fish migration in a hydropeaking river. River Research and Applications, 31(9): 1109-1118.
DOI:10.1002/rra.2810 |
Kaushik G, Bordoloi S. 2017. Length-weight and length-length relationships of Balitora brucei Gray, 1830 and Psilorhynchus balitora (Hamilton, 1822) from the Ranganadi river of Assam, India. Journal of Applied Ichthyology, 33(6): 1301-1302.
DOI:10.1111/jai.13477 |
King A J, Gwinn D C, Tonkin Z, Mahoney J, Raymond S, Beesley L. 2016. Using abiotic drivers of fish spawning to inform environmental flow management. Journal of Applied Ecology, 53(1): 34-43.
DOI:10.1111/1365-2664.12542 |
King A J, Tonkin Z, Mahoney J. 2009. Environmental flow enhances native fish spawning and recruitment in the Murray River, Australia. River Research and Applications, 25(10): 1205-1218.
DOI:10.1002/rra.1209 |
Kramer D L. 1978. Reproductive seasonality in the fishes of a tropical stream. Ecology, 59(5): 976-985.
DOI:10.2307/1938549 |
Le Pape O, Bonhommeau S. 2015. The food limitation hypothesis for juvenile marine fish. Fish and Fisheries, 16(3): 373-398.
DOI:10.1111/faf.12063 |
Lemes D M R, Vizioli B, Marcon L, Bazzoli N. 2017. Reproduction of the internal inseminator Trachelyopterus galeatus (Linnaeus, 1766) (Siluriformes: Auchenipteridae). Journal of Applied Ichthyology, 33(1): 69-74.
DOI:10.1111/jai.13205 |
Li L, Ma B, Gong J, Chen Z, Cheng L, Wu S, Ji F. 2018. Length-weight relationships of 6 fish species from the lower reaches of the YarlungZangbo River, Southwest China. Journal of Applied Ichthyology, 34(4): 1062-1064.
DOI:10.1111/jai.13706 |
Ma B S, Xie C X, Huo B, Yang X F, Chen S S. 2012. Reproductive biology of Schizothorax o'connori (Cyprinidae: Schizothoracinae) in the Yarlung Zangbo River, Tibet. Zoological Studies, 51(7): 1066-1076.
|
McDowall R M. 2009. Early hatch: a strategy for safe downstream larval transport in amphidromous gobies. Reviews in Fish Biology and Fisheries, 19(1): 1-8.
DOI:10.1007/s11160-008-9085-y |
Patimar R, Abdoli A, Kiabi B H. 2008. Biological characteristics of the introduced sawbelly, Hemiculter leucisculus (Basilewski, 1855), in three wetlands of northern Iran: Alma-Gol, Adji-Gol and Ala-Gol. Journal of Applied Ichthyology, 24(5): 617-620.
DOI:10.1111/j.1439-0426.2008.01080.x |
Saxena N, Patiyal R S, Rawat K D, Tiwari V K. 2016. Population dynamics and reproductive biology of Barilius bendelisis (Cyprinidae: Cypriniformes) from river Gaula of Central Indian Himalaya. Revista de Biologia Tropical, 64(3): 1287-1295.
DOI:10.15517/rbt.v64i3.20844 |
Sear D A, Pattison I, Collins A L, Newson M D, Jones J I, Naden P S, Carling P A. 2014. Factors controlling the temporal variability in dissolved oxygen regime of salmon spawning gravels. Hydrological Processes, 28(1): 86-103.
DOI:10.1002/hyp.9565 |
Shangningam B, Vishwanath W. 2014. Redescription of Psilorhynchus homaloptera, a torrent minnow from Northeast India (Teleostei: Psilorhynchidae). Ichthyological Exploration of Freshwaters, 24(3): 237-248.
|
Sivakumaran K P, Brown P, Stoessel D, Giles A. 2003. Maturation and reproductive biology of female wild carp, Cyprinus carpio, in Victoria, Australia. Environmental Biology of Fishes, 68(3): 321-332.
DOI:10.1023/A:1027381304091 |
Somerton D A. 1980. A computer technique for estimating the size of sexual maturity in crabs. Canadian Journal of Fisheries and Aquatic Sciences, 37(10): 1488-1494.
DOI:10.1139/f80-192 |
Strüssmann C A, Conover D O, Somoza G M, Miranda L A. 2010. Implications of climate change for the reproductive capacity and survival of New World silversides (family Atherinopsidae). Journal of Fish Biology, 77(8): 1818-1834.
DOI:10.1111/j.1095-8649.2010.02780.x |
Talwar P K, Jhingran A G. 1991. Inland fishes of India and Adjacent Countries. A. A. Balkema, Rotterdam. 541p.
|
Wang J. 2018. Fish community structure in the lower reach Yarlung Tsangpo River and the biological traits of Schizothorax Curvilabiatusa. Institute of Hydrobiology, Chinese Academy of Sciences, Wuhan.
|
Winemiller K O. 1995. Fish ecology. In: Nierenberg W A ed. Encyclopedia of Environmental Biology, vol. 2. Academic Press, San Diego. p. 49–65.
|
Wootton R J. 1998. Ecology of Teleost Fishes. 2nd edn. Kluwer, Dordrecht. 386p.
|
Yue P Q, Chen Y Y. 1998. China Red Data Book of Endangered Animals: Pisces. Science Press, Beijing. 247p. (in Chinese)
|
Yue P Q. 2000. Psilorhynchidae. In: Yue P Q ed. Fauna Sinica Osteicthtyes Cypriniformes III. Science Press, Beijing, p. 434–437. (in Chinese)
|
Zaniboni-Filho E, Ribolli J, Hermes-Silva S, Nuñer A P O. 2017. Wide reproductive period of a long-distance migratory fish in a subtropical river, Brazil. Neotropical Ichthyology, 15(1): e160135.
DOI:10.1590/1982-0224-20160135 |
Zar J H. 1999. Biostatistical Analysis. 4th edn. Prentice Hall, Upper Saddle River. 663p.
|
Zhou X J, Xie C X, Huo B, Duan Y J, Yang X, Ma B S. 2015. Reproductive biology of Schizothorax waltoni (Cyprinidae: Schizothoracinae) in the Yarlung Zangbo River in Tibet, China. Environmental Biology of Fishes, 98(2): 597-609.
DOI:10.1007/s10641-014-0293-0 |
Zhu S, Wu Z H, Zhao X T, Xiao K Y. 2013. Glacial dammed lakes in the Tsangpo River during late Pleistocene, southeastern Tibet. Quaternary International, 298: 114-122.
DOI:10.1016/j.quaint.2012.11.004 |
 2021, Vol. 39
2021, Vol. 39


