Institute of Oceanology, Chinese Academy of Sciences
Article Information
- HUANG Hongjia, GAN Chuqiao, HUANG Jiahui, ZOU Chen, LI Hongye, LIU Jiesheng, YANG Weidong
- Variability of Prorocentrum donghaiense response to allelopathic action from Alexandrium pacificum in laboratory culture
- Journal of Oceanology and Limnology, 39(4): 1305-1315
- http://dx.doi.org/10.1007/s00343-020-0226-z
Article History
- Received Jun. 9, 2020
- accepted in principle Jul. 21, 2020
- accepted for publication Sep. 18, 2020
Harmful algal blooms (HABs), known as red tides, occurred in frequency and intensity, and spanned worldwide over the past two decades (Kremp et al., 2012; Cai et al., 2014). In China, eutrophication has become a serious environmental issue with the rapid development of economy, partly leading to the high frequent occurrence of HABs (Anderson et al., 2012; Kremp et al., 2012; Cai et al., 2014).
Prorocentrum donghaiense and Alexandrium pacificum are two frequent HAB-forming dinoflagellate species in China, especially in the East China Sea. As a dominant species, P. donghaiense has frequently formed large-scale blooms in the East China Sea since 1990s (Lu and Goebel, 2001; Lu et al., 2003). P. donghaiense blooms usually occur in area ranging from several thousand to more than ten thousand km2, lasting for several days to months, which results in great losses in economy and severe damages in marine environment. Different from P. donghaiense, A. pacificum is a notorious toxin-producing dinoflagellate species, which can produce paralytic shellfish poisoning (PSP) toxins and some extracellular substances (Tillmann and John, 2002) responsible for the PSP in humans.
It is reported that A. pacificum has an allelopathic effect on the growth of other algal species, including P. donghaiense (Wang et al., 2006; Yang et al., 2010), which supports its ability to form blooms. However, the two algal species usually exist in the same sea areas, and in some cases form bi-phase HABs in China, especially in Zhejiang sea area recently. At present, no evidence is shown that P. donghaiense produces some toxins or has allelopathic effect on other microalgal species (Glibert et al., 2012; Lin et al., 2014), but several studies have demonstrated P. donghaiense has high adaptability to a changing marine environment. P. donghaiense can make good use of the metabolized dissolved organic phosphorus in the water (Ou et al., 2008), in addition to that, intraspecific genotypic and phenotypic variability might contribute the high adaptability of P. donghaiense (Kremp et al., 2012). However, no mechanistic model yet is available that can fully explain the coexistence of P. donghaiense and A. pacificum.
Several studies have investigated the algal competition among marine algal species, which demonstrated that inter-specific competition might play an important role in the succession of marine algal species (Wang et al., 2006, 2013; Yang et al., 2010; Qiu et al., 2011). Interaction between algae has a potential to construct the composition and structure of phytoplankton community in marine environment (Xu et al., 2020). Allelopathy, an important inter-specific competition, can prevent competitive exclusion and promote phytoplankton diversity in aquatic ecosystems (Barreiro Felpeto et al., 2018). The allelopathic action may be complex and dynamic, and it seems to be determined by concrete group characteristics in the target and donor species (Fistarol et al., 2004). The co-existing competitors may differ in their responses to allelopathy, and both the involved target and donor species might exhibit allelopathic properties (Legrand et al., 2003; Prince et al., 2008; Granéli and Salomon, 2010; Poulin et al., 2018). Moreover, the intra-specific trait diversity including allelochemical potency can promote the success of a species (Van de Waal et al., 2015; Brandenburg et al., 2018). Therefore, it is of paramount significance to learn the variability in alga-alga interaction between P. donghaiense and A. pacificum for discovering underlying mechanism in their coexistence.
In this study, we focused on the responses of various strains of P. donghaiense isolated from Zhejiang sea area to the allelopathy of A. pacificum under laboratory conditions. Therefore, a series of experiments, including bi-algal culture and cell-free filtrate culture were performed to determine whether the response to the allelopathic action varied between different strains of P. donghaiense. In addition, the competition between P. donghaiense and A. pacificum was discussed.
2 MATERIAL AND METHOD 2.1 Culture conditionIn total, 16 isolates of P. donghaiense were collected in May in 2014, respectively (Table 1), and A. pacificum strain was collected from Daya Bay, Guangdong Province, the South China Sea. The collected algal samples were diluted up to the appropriate concentration, then algal cells were picked out by using a capillary tube. The isolated algal cells were cultivated using 6-well culture plates. All strains were grown as batch cultures in Erlenmeyer flasks containing f/2 medium, which was filter-sterilized through 0.22-μm filters (Millipore). The pH of cultures was adjusted to 8.0 with 1-mol/L HCl. The cultures were grown at 20±1 ℃ in an artificial climate incubator (RXZ-430E). Cool-white fluorescent tubes provided an irradiance of 68 mmol photons/(m2·s) with a 12-h/12-h light/dark regime. All of the cultures were shaken manually at set times daily.
The initial cell densities for P. donghaiense and A. pacificum in the co-culture were set in terms of their respective cellular volume (Wang et al., 2006). In the co-culture, the initial density of A. pacificum was 0.28×104 cells/mL, and it was 1.0×104 cells/mL or 1.0×105 cells/mL for P. donghaiense. Correspondingly, the size/density ratio was 1꞉1 and 10꞉1, respectively. Experiments were carried out in a series of 150-mL Erlenmeyer flasks, where 100 mL of culture was present. Monocultures of A. pacificum at 0.28×104 cells/mL and P. donghaiense at 1.0×104 cells/mL or 1.0×105 cells/mL were used as related controls. The co-culture experiment lasted for 7 days. Cell densities of the microalgae were counted daily under a microscope (Olympus, CKX41) using triplicate subsamples of 0.1 mL. The intrinsic growth rate of P. donghaiense strains in monoculture was calculated. Inhibition of A. pacificum against P. donghaiense in co-culture was expressed as the inhibitory rate (IR) as described by previous studies (Chen et al., 2015; Gao et al., 2015). Reciprocal interaction parameters between two species in the co-cultures were evaluated by using the Lotka-Volterra model (Uchida et al., 1999; Wu and Wang, 2011), in which parameter a indicates the inhibition degree of P. donghaiense by A. pacificum when compared to its self-interference (Wang et al., 2006).
2.3 Exudate bioassayIn this experiment, we cultivated each strain of P. donghaiense (1.0×105 cells/mL) in filtrate from A. pacificum culture. Filtrate was obtained from the A. pacificum culture at density of 1.12×104 cells/mL when it was in the exponential growth phase by using 0.22-μm filters (Uchida et al., 1999). The filtrate pH was adjusted to 7.8‒8.0 by 1-mol/L HCl, and the nutrient content of the filtrate was detected by using AutoAnalyzer3 (Brand Luebbe, Germany) and re-enriched to f/2 level (Wang et al., 2013). In control, no filtrate was added in P. donghaiense cultures, but sufficient nutrients were offered as f/2 level. This experiment lasted for 7 days. The cell density of P. donghaiense was measured daily under a microscope (Olympus, CKX41).
2.4 Statistical analysesStatistical analyses were performed using the software SPSS v.20.0, and all data were given as mean values±SD. To determine the significance in the growth between the experimental group and control, Student's t-test was carried out after testing for homogeneity of variance. The differences were considered significant at value of P < 0.05. To determine the changes in cell density over time, multiple comparison tests with a two-way analysis of variance (ANOVA) were performed with significance at P < 0.05. The allelopathic potency of A. pacificum towards P. donghaiense was evaluated by Hierarchilcal cluster analysis (Average Linkage, between groups) using SPSS v.20.0 in terms of the parameter a in bi-algal culture, and the inhibition rate on the first day both in bi-algal and in filtrate culture. Potential correlation between inhibition rate, intrinsic growth rate, and parameter a were evaluated by partial correlation analysis using SPSS v.20.0.
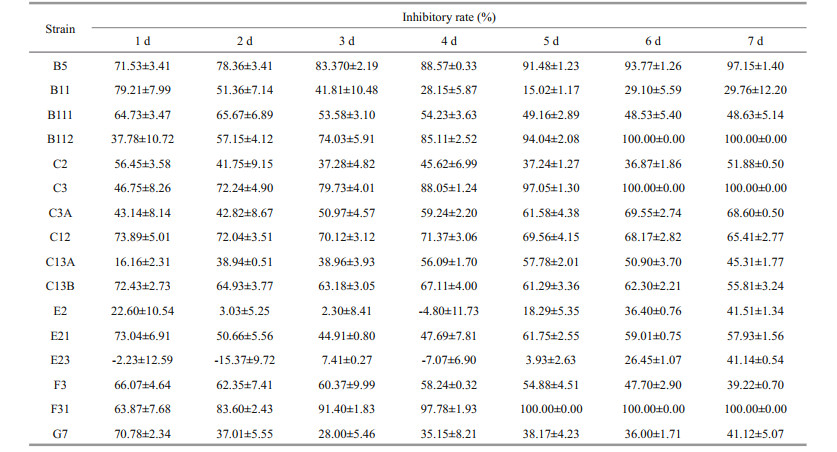
3 RESULT 3.1 Interaction between A. pacificum and P. donghaiense in bi-algal cultureAs shown in Fig. 1a, the growth of P. donghaiense were dramatically suppressed (P < 0.05) on day 3 in bi-algal cultures with 1.0×104 cells/mL of P. donghaiense and 0.28×104 cells/mL of A. pacificum, while the growth of A. pacificum remained almost unchanged compared to the control (P > 0.05). However, when the initial cell density of P. donghaiense was increased to 1.0×105 cells/mL, the growth of different P. donghaiense strains in the bi-algal cultures was significantly different. Some strains including B5, B112, E21, F31, and G7 died after about 6 days, while B11, C2, C12, and F3 were significantly suppressed on day 8 compared to the control (P < 0.05). Interestingly, the strains B111, C3, C3A, C13A, C13B, E2, and E23 showed obvious growth in spite of being inhibited in short-term period (Fig. 1b). The cell densities of the strains C3, C3A, and C13A were increased over time after co-cultured for about 2 days (ANOVA, P < 0.05).
|
| Fig.1 Changes in cell density of A. pacificum and P. donghaiense in co-cultures with initial cell density of P. donghaiense at 1.0×104 cells/mL (a) or 1.0×105 cells/mL (b) Data are expressed as mean±SD (n=3). P.d: P. donghaiense; P.d-co: P. donghaiense in co-cultures with A. pacificum; A.p: A. pacificum; A.p-co: A. pacificum in co-cultures with P. donghaiense. |
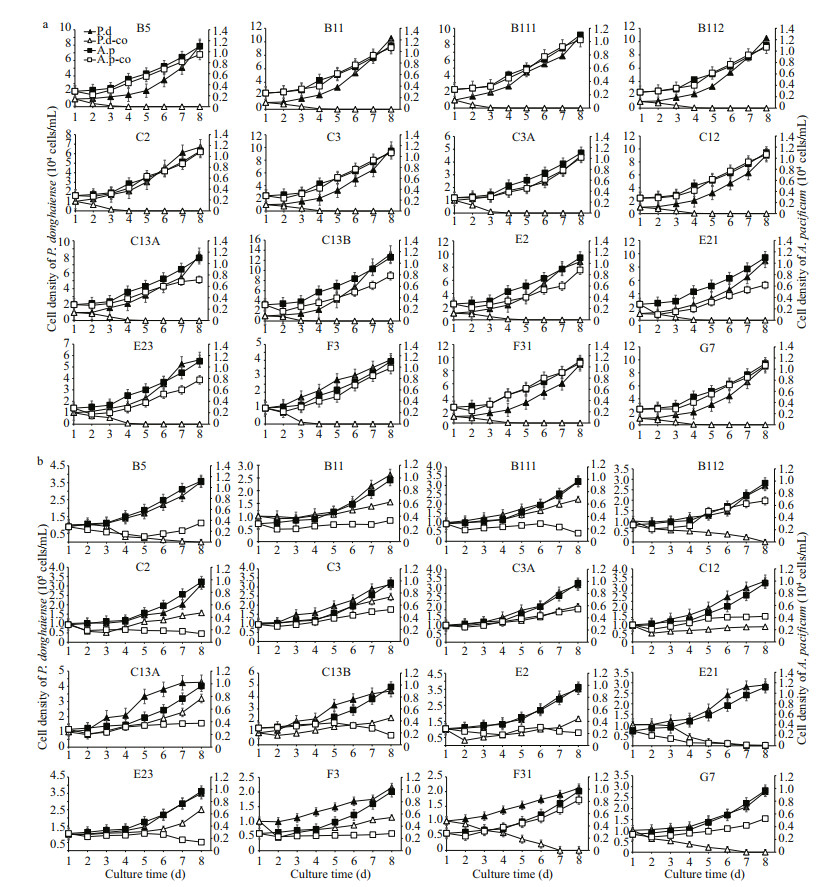
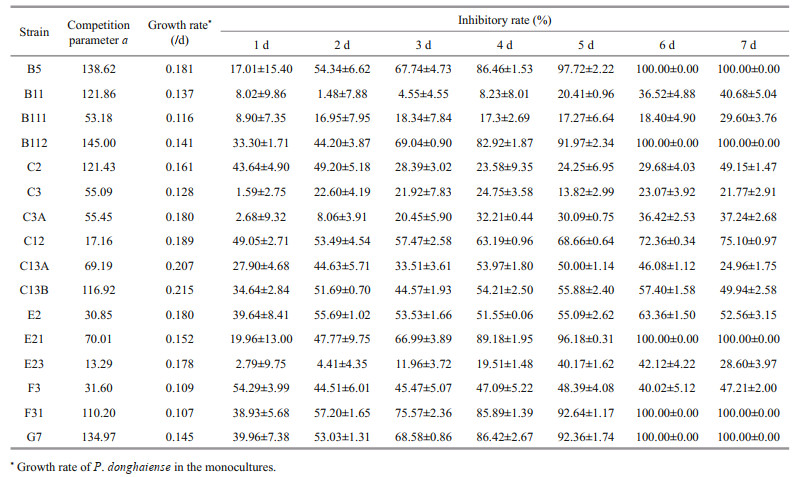
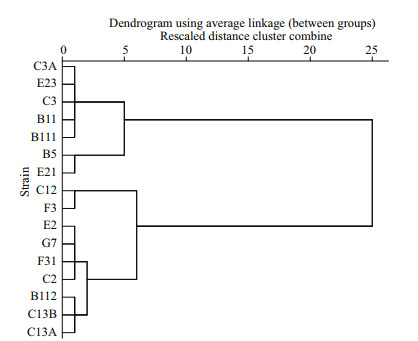
Intrinsic growth rate in monoculture, inhibitory rates, and parameter a of different strains of P. donghaiense in bi-algal culture with initial cell density of 1.0×105 cells/mL are presented in Table 2. Different strains of P. donghaiense exhibited different responses to A. pacificum allelopathy. For example, the IR of P. donghaiense at 6 d after co-culture with A. pacificum ranged from 18.40%±4.90% to 100.00%±0.00%. Based on the IR of A. pacificum on the growth of P. donghaiense on the first day in bi-algal culture, 16 strains of P. donghaiense can be divided into 3 hierarchical groups from weak to strong as follows: (1) weak (W): E23, C3A, C3, B111, B11, E21, B5; (2) moderate (M): F31, G7, E2, C2, C13A, C13B, B112; (3) strong (S): F3, C12 (Fig. 2).
|
|
| Fig.2 A hierarchical cluster of 16 strains of P. donghaiense in terms of the inhibitory effect of A. pacificum against P. donghaiense on the first day after co-culture, where squared Euclidean distance was selected as the measurement The abscissa means rescaled distance cluster combine. |
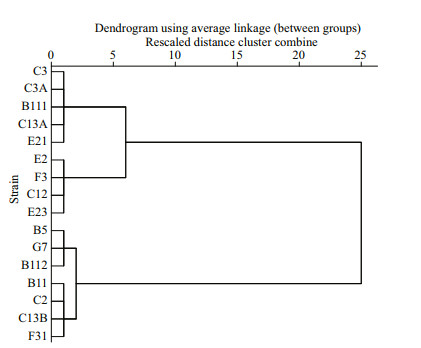
Similarly, different strains of P. donghaiense in bi-algal culture showed different parameter a, which ranged from 13.29 to 145.00. In terms of the parameter a, 16 strains of P. donghaiense can also be divided into 3 hierarchical groups follows (Fig. 3): (1) weak (W): E2, F3, C12, E23; (2) moderate (M): C3, C3A, B111, C13A, E21; (3) strong (S): B5, G7, B112, B11, C2, C13B, F31.
|
| Fig.3 A hierarchical cluster of 16 strains of P. donghaiense based on the parameter a of P. donghaiense, where squared Euclidean distance was selected as the measurement The abscissa means rescaled distance cluster combine. |
Interestingly, different strains of P. donghaiense also displayed different intrinsic growth rates in monocultures. However, partial correlation analysis showed that the inhibitory rate of different strains of P. donghaiense was not correlated with parameter a and intrinsic growth rate.
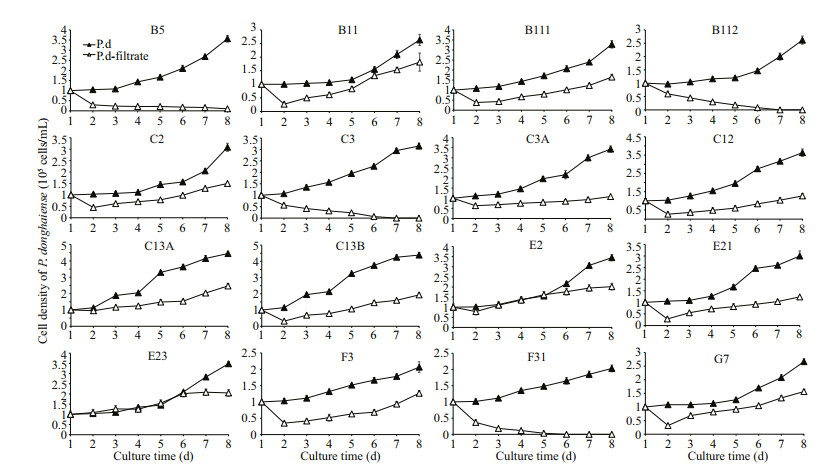
3.2 Effect of filtrate from A. pacificum on the growth of P. donghaienseTo further study the inhibitory mechanism of A. pacificum on the growth of P. donghaiense, we conducted the exudate bioassay. As shown in Fig. 4, similar to the results in bi-algal system, the growth of B5, B112, C3, and F31 were inhibited significantly when cultured in A. pacificum filtrate through all the experiment periods (P < 0.05). However, the growth of strains B11, B111, C2, C3A, C12, C13A, C13B, E2, E21, F3, and G7 were inhibited on the first two days after culture (P < 0.05), and cell densities of strains B11, C2, C12, C13B, E2, E21, and G7 began to increase since the third day (ANOVA, P < 0.05). It is of note that the exudate bioassay results were not always consistent with that of bi-algal experiment. In bi-algal culture, strains E21 and G7 were inhibited severely, whereas they displayed an obvious growth trend despite some inhibition in filtrate of A. pacificum. In contrast, the cell density of C3 was increased in bi-algal culture, though it was lower than that in monoculture after co-cultured for 2 days (P < 0.05). However, cell density of C3 in A. pacificum filtrate was decreased to zero.
|
| Fig.4 Effect of filtrate from A. pacificum on the growth of P. donghaiense Data are expressed as mean±SD (n=3). Vertical lines show the standard deviation. P.d: P. donghaiense; P.d-filtrate: P. donghaiense in culture with A. pacificum filtrate. |
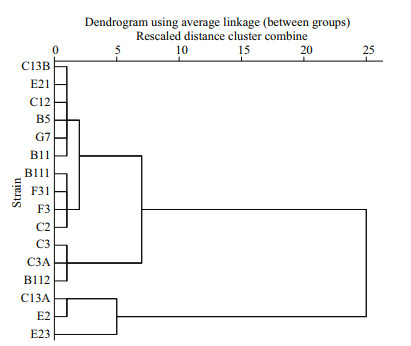
Inhibitory rates of A. pacificum filtrate on P. donghaiense are presented in Table 3. The IR of P. donghaiense in A. pacificum filtrate varied from -15.37%±9.72% to 100.00%±0.00%. According to the IR of P. donghaiense on the first day in filtrate culture, the 16 strains of P. donghaiense can be divided into 3 hierarchical groups as follows (Fig. 5): (1) weak (W): C13A, E2, E23; (2) moderate (M): C3, C3A, B112; (3) strong (S): C13B, E21, C12, B5, G7, B11, B111, F31, F3, C2.
|
| Fig.5 A hierarchical cluster of the 16 P. donghaiense strains in terms of the inhibitory effect of A. pacificum filtrate against P. donghaiense on the first day after culture, where squared Euclidean distance was selected as the measurement The abscissa means rescaled distance cluster combine. |
Allelopathic effects are a natural phenomenon among higher plants and also between higher plants and microorganisms in ecosystems (Roy et al., 2006; Yang et al., 2010). This ecological phenomenon also exists among marine microalgae, and it is considered an important factor that promotes the HABs-forming species over other algal species. Many studies have dealt with the allelopathic action of Alexandrium species (Alpermann et al., 2010). In the present study, we found that the growth of P. donghaiense was depressed deeply in the co-culture with initial cell density of 1.0×104 cells/mL for P. donghaiense and 0.28×104 cells/mL for A. pacificum, suggesting that allelopathy was one of the important phenotypes of A. pacificum, which is in line with the results of previous studies (Wang et al., 2006; Alpermann et al., 2010; Yang et al., 2010; Yin et al., 2010).
Allelopathic activity usually depends on the initial cell density of the associated target and donor species (Wang et al., 2006; Yamasaki et al., 2011). To further explore the response of various P. donghaiense strains to A. pacificum, we conducted the bi-algal culture with the 10꞉1 size/density ratio (1.0×105 cells/mL for P. donghaiense, 0.28×104 cells/mL for A. pacificum). In contrast with the results in the 1꞉1 bi-algal culture, different P. donghaiense strains showed different responses to the co-culture with A. pacificum. The inhibition of strains B111, C3, C3A, C13A, and E23 was relatively weak, and these strains could maintain their growth, while the growth of strains B5, B112, E21, and G7 was depressed significantly, and the number of algal cells approached to zero. It is obvious that strains B111, B11, C2, C3, C3A, C13A, and E23 were able to maintain growth in the co-culture system though the growth of both algal species was inhibited to some extent. These results indicate that the two algal species P. donghaiense and A. pacificum can coexist, even if A. pacificum is allelopathic. Kaur et al. (2019) proposed that green alga Scenedesmus abundans could resist against allelochemicals from Microcystis aeruginosa by biochemical and proteomic reprogramming. However, whether and how these P. donghaiense strains resist allelopathic compounds from A. pacificum remain to be elucidated in the future.
Cell-free filtrate cultivation is usually employed to determine the allelopathic effect of algae on target species (Zhang et al., 2015), so we observed the effect of A. pacificum filtrate on the growth of different strains of P. donghaiense. Similar to the results in bi-algal culture, the growth of strains B5 and B112 was significantly inhibited when cultured in the A. pacificum filtrate for 6 days, while the growth of strains B11, B111, C2, C3A, and C13A was inhibited on the first two days, and began to increase from the third day. These results suggest that the A. pacificum filtrate really affect the growth of P. donghaiense, possibly through extracellular substances produced by A. pacificum (Tillmann and John, 2002; Chen et al., 2015).
It is worth noting that the exudate bioassay results were not always consistent with that of bi-algal experiment. In bi-algal culture, strains E21 and G7 were severely inhibited, whereas they were inclined to grow despite some inhibition in the filtrate of A. pacificum. Other forms of competition between A. pacificum and P. donghaiense, allelochemicals instability and physiological diversity might account for the discrepancy (Tillmann and Hansen, 2009). Unfortunately, the nature of allelochemicals of A. pacificum is quite complex, and remains unclear to date, though several studies have been conducted (Ma et al., 2009, 2011; Chen et al., 2015). Surprisingly, cell density of strain C3 was increased despite the inhibition in bi-algal culture, whereas A. pacificum filtrate could be lethal to it, which might be attributed to the potential physiological adaptation and high growth rate of strain C3 (Cullen and MacIntyre, 1999). However, the growth rate of strain C3 is only 0.128/d, lower than most of the strains of P. donghaiense analyzed. The intrinsic growth rate of microalgae plays an important role in the reciprocal interaction system. In general, high growth rate favors the donor species over other co-existing competitors. However, we did not observe a negative correlation between growth rate and inhibitory rate, suggesting that the diversity of P. donghaiense in response to allelopathic action of A.pacificum might be also related to other physiological characters. Heterotrophic bacteria may interfere with allelopathic action between phytoplankton species by degrading allelochemicals or producing some harmful compounds by themselves (Hulot and Huisman, 2004). In our study, we performed all experiments in axenic and nutrient-enriched batch cultures. However, we cannot easily exclude the possible role of symbiotic bacteria in the A. pacificum allopathy against P. donghaiense, especially when trying to explain any phenomena in field.
Based on the inhibition of P. donghaiense on the first day in bi-algal culture and in filtrate culture, and parameter a, the 16 strains of P. donghaiense can be divided into 3 hierarchical groups, but the three hierarchical groups do not completely match each other. For examples, strains E21 and B5 are clustered into W group according to the inhibition in co-culture, but they are attributed to group M and S, respectively, based on the parameter a. In terms of the inhibition of A. pacificum filtrate, the two strains are ascribed to S group. These inconsistences suggest the complexity of response to A. pacificum in different strains of P. donghaiense. Remarkably, P. donghaiense is able to adapt to low irradiance, low temperature ranging from 10 to 20 ℃ and a broad range of salinities (Xu et al., 2010). High genetic diversity of P. donghaiense population might form the basis for variability in phenotypes such as the intrinsic growth rate, sensitivity to allelochemicals, suitable growth conditions, and empower it to display different reaction to allelopathic action of A. pacificum (Huang et al., 2020). Intraspecific variability has been regarded as a key driver for biodiversity sustenance in ecosystems (De Laender et al., 2014). Intraspecific variable phenotypic traits including nitrogen and carbon uptake rates, movement behaviors, and responses to changing environments have been reported in dinoflagellates (Van de Waal et al., 2015), diatoms (Godhe and Rynearson, 2017), and raphidophytes (Harvey et al., 2015). The variability in phenotype of P. donghaiense remains an interesting topic for future study.
Blooms of P. donghaiense have been studied intensively in recent years. However, the reasons responsible for these blooms remain elusive. Allelopathy in phytoplankton is closely associated with competition for resources. Studies show that P. donghaiense can utilize a broad array of nutrients compound, especially nitrogen, which is an important regulator of the proliferation of P. donghaiense (Hu et al., 2012). Zhang et al. (2019) demonstrated that P. donghaiense evolved diverse dissolved organic phosphorus (DOP) utilization strategies to adapt to low phosphorus (P) environments, and that DOPs might play critical roles in the P. donghaiense bloom formation. Increasing studies have demonstrated the ability of dinoflagellates to respond to changing environments, which determine their success and proliferation, as well as bloom formations (Gobler et al., 2017). Many studies have dealt with the diversity of allelopathic activity among species and strains within the same species (Hakanen et al., 2014; Chen et al., 2015; Van de Waal et al., 2015). Meanwhile, a few studies have shown that allelopathic action is usually species dependent (Hakanen et al., 2014; Prasetiya et al., 2016). Here, maybe for the first time, we found the diversity of different strains of target species to allelopathic action, and variability in growth rate of P. donghaiense, suggesting that high adaptability of P. donghaiense to the marine environment might be related to the phenotypic variability (Kremp et al., 2012). Alternatively, the immediate response of P. donghaiense to environmental changes can involve both acclimation based on phenotypic plasticity, and adaptation based on selection (Barrett and Schluter, 2008). As a result, P. donghaiense could display a high level of phenotypic variability, which may stabilize a species response to environment changes, finally could co-exist with A. pacificum, and in some cases achieve dominance and form bloom with A. pacificum in Zhejiang sea area.
5 CONCLUSIONDifferent strains of P. donghaiense showed different intrinsic growth rate in monoculture. The toxic dinoflagellate A. pacificum had a density-dependent allelopathic effect on the growth of P. donghaiense, but different strains of P. donghaiense exhibited different responses to the allelopathic action from A. pacificum. Some strains of P. donghaiense might be resist to chemicals from A. pacificum, and could grow well despite being inhibited in short-time period. This variability of P. donghaiense in reaction to allelopathic action from A. pacificum made it possible that P. donghaiense and A. pacificum co-exist in the same sea area, and even form blooms in certain conditions. The variability in phenotype of P. donghaiense deserves further attention as an interesting topic for future studies. Our findings might advance the understanding to the phenotype of P. donghaiense and suggest a potential mechanism involved in the coexistence of P. donghaiense and A. pacificum in the same area.
6 DATA AVAILABILITY STATEMENTThe data generated in the current study were available from the corresponding author or the first author on reasonable request.
Alpermann T J, Tillmann U, Beszteri B, Cembella A D, John U. 2010. Phenotypic variation and genotypic diversity in a planktonic population of the toxigenic marine dinoflagellate Alexandrium tamarense (Dinophyceae). Journal of Phycology, 46(1): 18-32.
DOI:10.1111/j.1529-8817.2009.00767.x |
Anderson D M, Alpermann T J, Cembella A D, Collos Y, Masseret E, Montresor M. 2012. The globally distributed genus Alexandrium: multifaceted roles in marine ecosystems and impacts on human health. Harmful Algae, 14: 10-35.
DOI:10.1016/j.hal.2011.10.012 |
Barreiro Felpeto A, Roy S, Vasconcelos V M. 2018. Allelopathy prevents competitive exclusion and promotes phytoplankton biodiversity. Oikos, 127(1): 85-98.
DOI:10.1111/oik.04046 |
Barrett R D H, Schluter D. 2008. Adaptation from standing genetic variation. Trends in Ecology & Evolution, 23(1): 38-44.
DOI:10.1016/j.tree.2007.09.008 |
Brandenburg K M, Wohlrab S, John U, Kremp A, Jerney J, Krock B, Van de Waal D B. 2018. Intraspecific trait variation and trade-offs within and across populations of a toxic dinoflagellate. Ecology Letters, 21(10): 1 561-1 571.
DOI:10.1111/ele.13138 |
Cai Z P, Zhu H H, Duan S S. 2014. Allelopathic interactions between the red-tide causative dinoflagellate Prorocentrum donghaiense and the diatom Phaeodactylum tricornutum. Oceanologia, 56(3): 639-650.
DOI:10.5697/oc.56-3.639 |
Chen J, Ye Q, Gu H F, Li H Y, Lv S H, Liu J S, Yang W D. 2015. Variability in the allelopathic action of the Alexandrium tamarense species complex along the coast of China. Harmful Algae, 47: 17-26.
DOI:10.1016/j.hal.2015.05.008 |
Cullen J J, MacIntyre J G. 1999. Behavior, physiology and the niche of depth-regulating phytoplankton. In: Heidelberg G M, Anderson D M, Cembella A D, Hallegraeff G M eds. Physiological Ecology of Harmful Algal Blooms. Springer-Verlag, Heidelberg. p. 1-21.
|
De Laender F, Melian C J, Bindler R, Van den Brink P J, Daam M, Roussel H, Juselius J, Verschuren D, Janssen C R. 2014. The contribution of intra- and interspecific tolerance variability to biodiversity changes along toxicity gradients. Ecology Letters, 17(1): 72-81.
DOI:10.1111/ele.12210 |
Fistarol G O, Legrand C, Selander E, Hummert C, Stolte W, Granéli E. 2004. Allelopathy in Alexandrium spp. : effect on a natural plankton community and on algal monocultures. Aquatic Microbial Ecology, 35(1): 45-56.
DOI:10.3354/ame035045 |
Gao H J, Song Y H, Lv C J, Chen X M, Yu H B, Peng J F, Wang M. 2015. The possible allelopathic effect of Hydrilla verticillata on phytoplankton in nutrient-rich water. Environmental Earth Sciences, 73(9): 5 141-5 151.
DOI:10.1007/s12665-015-4316-8 |
Glibert P M, Burkholder J M, Kana T M. 2012. Recent insights about relationships between nutrient availability, forms, and stoichiometry, and the distribution, ecophysiology, and food web effects of pelagic and benthic Prorocentrum species. Harmful Algae, 14: 231-259.
DOI:10.1016/j.hal.2011.10.023 |
Gobler C J, Doherty O M, Hattenrath-Lehmann T K, Griffith A W, Kang Y, Litaker R W. 2017. Ocean warming since 1982 has expanded the niche of toxic algal blooms in the North Atlantic and North Pacific oceans. Proceedings of the National Academy of Sciences of the United States of America, 114(19): 4 975-4 980.
DOI:10.1073/pnas.1619575114 |
Godhe A, Rynearson T. 2017. The role of intraspecific variation in the ecological and evolutionary success of diatoms in changing environments. Philosophical Transactions of the Royal Society B—Biological Sciences, 372(1728): 20160399.
DOI:10.1098/rstb.2016.0399 |
Granéli E, Salomon P S. 2010. Factors influencing allelopathy and toxicity in Prymnesium parvum. Journal of the American Water Resources Association, 46(1): 108-120.
DOI:10.1111/j.1752-1688.2009.00395.x |
Hakanen P, Suikkanen S, Kremp A. 2014. Allelopathic activity of the toxic dinoflagellate Alexandrium ostenfeldii: intra-population variability and response of co-occurring dinoflagellates. Harmful Algae, 39: 287-294.
DOI:10.1016/j.hal.2014.08.005 |
Harvey E L, Menden-Deuer S, Rynearson T A. 2015. Persistent intra-specific variation in genetic and behavioral traits in the raphidophyte, Heterosigma akashiwo. Frontiers in Microbiology, 6: 1 277.
DOI:10.3389/fmicb.2015.01277 |
Hu Z X, Mulholland M R, Duan S S, Xu N. 2012. Effects of nitrogen supply and its composition on the growth of Prorocentrum donghaiense. Harmful Algae, 13: 72-82.
DOI:10.1016/j.hal.2011.10.004 |
Huang H J, Gan C Q, Xiao S W, Zou C, Balamurugan S, Li H Y, Liu J S, Yang W D. 2020. Genetic diversity of Prorocentrum donghaiense population during bloom in the East China Sea revealed by microsatellite. Journal of Applied Phycology, 32(3): 1 851-1 862.
DOI:10.1007/s10811-020-02113-4 |
Hulot F D, Huisman J. 2004. Allelopathic interactions between phytoplankton species: the roles of heterotrophic bacteria and mixing intensity. Limnology and Oceanography, 49(4): 1 424-1 434.
DOI:10.4319/lo.2004.49.4_part_2.1424 |
Kaur S, Srivastava A, Kumar S, Srivastava V, Ahluwalia A S, Mishra Y. 2019. Biochemical and proteomic analysis reveals oxidative stress tolerance strategies of Scenedesmus abundans against allelochemicals released by Microcystis aeruginosa. Algal Research, 41: 101525.
DOI:10.1016/j.algal.2019.101525 |
Kremp A, Godhe A, Egardt J, Dupont S, Suikkanen S, Casabianca S, Penna A. 2012. Intraspecific variability in the response of bloom-forming marine microalgae to changed climate conditions. Ecology and Evolution, 2(6): 1 195-1 207.
DOI:10.1002/ece3.245 |
Legrand C, Rengefors K, Fistarol G O, Granéli E. 2003. Allelopathy in phytoplankton-biochemical, ecological and evolutionary aspects. Phycologia, 42(4): 406-419.
DOI:10.2216/i0031-8884-42-4-406.1 |
Lin J N, Yan T, Zhang Q C, Wang Y F, Liu Q, Zhou M J. 2014. In situ detrimental impacts of Prorocentrum donghaiense blooms on zooplankton in the East China Sea. Marine Pollution Bulletin, 88(1-2): 302-310.
DOI:10.1016/j.marpolbul.2014.08.026 |
Lu D D, Goebel J. 2001. Five red tide species in genus Prorocentrum including the description of Prorocentrum donghaiense Lu sp. nov. from the East China Sea. Chinese Journal of Oceanology and Limnology, 19(4): 337-344.
DOI:10.1007/BF02850738 |
Lu D D, Qi Y Z, Goebel J, Zou J Z, Gao Y H. 2003. Redescription of Prorocentrum donghaiense Lu and comparison with relevant Prorocentrum species. Chinese Journal of Applied Ecology, 14(7): 1 060-1 064.
(in Chinese with English abstract) |
Ma H Y, Krock B, Tillmann U, Cembella A. 2009. Preliminary characterization of extracellular allelochemicals of the toxic marine dinoflagellate Alexandrium tamarense using a Rhodomonas salina bioassay. Marine Drugs, 7(4): 497-522.
DOI:10.3390/md7040497 |
Ma H Y, Krock B, Tillmann U, Muck A, Wielsch N, Svatoš A, Cembella A. 2011. Isolation of activity and partial characterization of large non-proteinaceous lytic allelochemicals produced by the marine dinoflagellate Alexandrium tamarense. Harmful Algae, 11: 65-72.
DOI:10.1016/j.hal.2011.07.004 |
Ou L J, Wang D, Huang B Q, Hong H S, Qi Y Z, Lu S H. 2008. Comparative study of phosphorus strategies of three typical harmful algae in Chinese coastal waters. Journal of Plankton Research, 30(9): 1 007-1 017.
DOI:10.1093/plankt/fbn058 |
Poulin R X, Poulson-Ellestad K L, Roy J S, Kubanek J. 2018. Variable allelopathy among phytoplankton reflected in red tide metabolome. Harmful Algae, 71: 50-56.
DOI:10.1016/j.hal.2017.12.002 |
Prasetiya F S, Safitri I, Widowati I, Cognie B, Decottignies P, Gastineau R, Morançais M, Windarto E, Tremblay R, Mouget J. 2016. Does allelopathy affect co-culturing Haslea ostrearia with other microalgae relevant to aquaculture?. Journal of Applied Phycology, 28(4): 2 241-2 254.
DOI:10.1007/s10811-015-0779-y |
Prince E K, Myers T L, Kubanek J. 2008. Effects of harmful algal blooms on competitors: allelopathic mechanisms of the red tide dinoflagellate Karenia brevis. Limnology and Oceanography, 53(2): 531-541.
DOI:10.4319/lo.2008.53.2.0531 |
Qiu X C, Yamasaki Y, Shimasaki Y, Gunjikake H, Shikata T, Matsubara T, Nagasoe S, Etoh T, Matsui S, Honjo T, Oshima Y. 2011. Growth interactions between raphidophytes Chattonella antiqua and Heterosigma akashiwo. Thalassas, 27(1): 33-45.
|
Roy S, Alam S, Chattopadhyay J. 2006. Competing effects of toxin-producing phytoplankton on overall plankton populations in the Bay of Bengal. Bulletin of Mathematical Biology, 68(8): 2 303-2 320.
DOI:10.1007/s11538-006-9109-5 |
Tillmann U, Hansen P J. 2009. Allelopathic effects of Alexandrium tamarense on other algae: evidence from mixed growth experiments. Aquatic Microbial Ecology, 57(1): 101-112.
DOI:10.3354/ame01329 |
Tillmann U, John U. 2002. Toxic effects of Alexandrium spp. on heterotrophic dinoflagellates: an allelochemical defence mechanism independent of PSP-toxin content. Marine Ecology Progress Series, 230: 47-58.
DOI:10.3354/meps230047 |
Uchida T, Toda S, Matsuyama Y, Yamaguchi M, Kotani Y, Honjo T. 1999. Interactions between the red tide dinoflagellates Heterocapsa circularisquama and Gymnodinium mikimotoi in laboratory culture. Journal of Experimental Marine Biology and Ecology, 241(2): 285-299.
DOI:10.1016/S0022-0981(99)00088-X |
Van de Waal D B, Tillmann U, Martens H, Krock B, van Scheppingen Y, John U. 2015. Characterization of multiple isolates from an Alexandrium ostenfeldii bloom in The Netherlands. Harmful Algae, 49: 94-104.
DOI:10.1016/j.hal.2015.08.002 |
Wang J T, Zhang Y W, Li H, Cao J. 2013. Competitive interaction between diatom Skeletonema costatum and dinoflagellate Prorocentrum donghaiense in laboratory culture. Journal of Plankton Research, 35(2): 367-378.
DOI:10.1093/plankt/fbs098 |
Wang Y, Yu Z M, Song X X, Zhang S D. 2006. Interactions between the bloom-forming dinoflagellates Prorocentrum donghaiense and Alexandrium tamarense in laboratory cultures. Journal of Sea Research, 56(1): 17-26.
DOI:10.1016/j.seares.2006.04.002 |
Wu L F, Wang Y N. 2011. Estimation the parameters of Lotka-Volterra model based on grey direct modelling method and its application. Expert Systems with Applications, 38(6): 6 412-6 416.
DOI:10.1016/j.eswa.2010.09.013 |
Xu N, Duan S S, Li A F, Zhang C W, Cai Z P, Hu Z X. 2010. Effects of temperature, salinity and irradiance on the growth of the harmful dinoflagellate Prorocentrum donghaiense Lu. Harmful Algae, 9(1): 13-17.
DOI:10.1016/j.hal.2009.06.002 |
Xu W J, Tan L J, Guo X, Wang J T. 2020. Isolation of anti-algal substances from Cylindrotheca closterium and their inhibition activity on bloom-forming Prorocentrum donghaiense. Ecotoxicology and Environmental Safety, 190: 110180.
DOI:10.1016/j.ecoenv.2020.110180 |
Yamasaki Y, Ohmichi Y, Shikata T, Hirose M, Shimasaki Y, Oshima Y, Honjo T. 2011. Species-specific alleopathic effects of the diatom Skeletonema costatum. Thalassas, 27(1): 21-32.
|
Yang W D, Xie J, van Rijssel M, Li H Y, Liu J S. 2010. Allelopathic effects of Alexandrium spp. on Prorocentrum donghaiense. Harmful Algae, 10(1): 116-120.
DOI:10.1016/j.hal.2010.08.001 |
Yin J, Xie J, Yang W D, Li H Y, Liu J S. 2010. Effect of Alexandrium tamarense on three bloom-forming algae. Chinese Journal of Oceanology and Limnology, 28(4): 940-944.
DOI:10.1007/s00343-010-9925-1 |
Zhang S F, Yuan C J, Chen Y, Lin L, Wang D Z. 2019. Transcriptomic response to changing ambient phosphorus in the marine dinoflagellate Prorocentrum donghaiense. Science of the Total Environment, 692: 1 037-1 047.
DOI:10.1016/j.scitotenv.2019.07.291 |
Zhang Y W, Wang J T, Tan L J, Cao J, Li H. 2015. Effect of allelopathy on the competition and succession of Skeletonema costatum and Prorocentrum donghaiense. Marine Biology Research, 11(10): 1 093-1 099.
DOI:10.1080/17451000.2015.1062519 |
 2021, Vol. 39
2021, Vol. 39