Institute of Oceanology, Chinese Academy of Sciences
Article Information
- ZHAO Miao, ZHANG Shenghui, HAN Haitao, PAN Dawei
- Heavy metals in sediments of Yellow Sea and East China Sea: Chemical speciation, distribution, influence factor, and contamination
- Journal of Oceanology and Limnology, 39(4): 1277-1292
- http://dx.doi.org/10.1007/s00343-020-0237-9
Article History
- Received Jun. 19, 2020
- accepted in principle Aug. 21, 2020
- accepted for publication Oct. 20, 2020
2 University of Chinese Academy of Sciences, Beijing 100049, China
Heavy metals are involved in almost every aspect of marine life and play a dual role in marine biogeochemical cycles. Most heavy metals (such as cobalt, cadmium, etc.) act as basic micronutrients at low concentrations but pose a threat to marine biota at higher concentrations because of the bioaccumulatable, nonbiodegradable, and toxic nature (Gu et al., 2012; Varol and Şen, 2012). Heavy metals are derived mainly from modern sewage and industrial emissions, and their flux into the aquatic environment is usually through waste dumping, and riverine and aeolian processes (Bak et al., 1997). Heavy metals that discharged into the ocean would be further accumulated in the sediments by the physical adsorption, coprecipitation, complexation, and ion exchange. For instance, dissolved arsenic and copper can be absorbed into sediment by precipitating amorphous iron oxyhydroxides; cobalt can easily form complexes and be incorporated into sediment; and molybdenum tends to coprecipitate with pyrite (Bryan and Langston, 1992; van de Velde et al., 2020). The absorption of heavy metals into sediment always goes with the redox reaction and particle-reaction, and these processes can significantly affect heavy metal biogeochemical processes (Liu et al., 2015). Additionally, metals can be released back to porewater from solid phases during the processes of organic matter mineralization, dissimilatory reduction, and pH decrease (van de Velde et al., 2017).
Heavy metals can be concentrated in sediments for 103–105 times of those in overlying water, and the metal effluxes from sediment to overlying seawater were usually in a very low level (Bryan and Langston, 1992). Therefore, the total concentration of heavy metals in solid phase could not reflect their potential toxicity precisely (Tessier et al., 1979). Different chemical speciation of metals have different environmental behaviors and ecotoxicology, metal fractionation has been proved critical in assessing their bioavailability and mobility in previous studies (Massas et al., 2009; Nagajyoti et al., 2010). In addition, the ratio of metals in solid phase and porewater is associated closely with their toxicity and fate of metals (He et al., 2017). Therefore, it is of great importance to study the metals fractionation and metals content in porewater.
In addition to sediment type, organic content, hydroxide of iron and manganese, bioturbation, and pH, sulfide is a critical factor on the concentration, toxicity, and destiny of metals (Chapman et al., 1998; Burton et al., 2006; He et al., 2017; Shyleshchandran et al., 2018). Substantial evidence has demonstrated that the deposition and release of heavy metal can be affected by sulfide significantly. For instance, acid volatile sulfide (AVS) played a major role in controlling the concentrations of heavy metal and reducing the biotoxicity of heavy metals in sediments (Machado et al., 2004). Mineralization of sulfur and iron would impose on the circulation of various metals such as arsenic, cobalt, and iron (Malkin et al., 2014). As an important reducing product of early mineralization process, hydrogen sulfide (H2S) can react with metals in the formation of metallic sulfide (e.g., FeS, PbS, and ZnS), and many divalent transition metals (e.g., Ni2+, Cd2+, and Cu2+) can be incorporated during transformation of FeS to pyrite (Morse and Luther Ⅲ, 1999). The biogeochemical cycles of heavy metals and sulfur in solid has attracted a great interest in the past few decades (Giblin and Howarth, 1984; He et al., 2011; Li et al., 2017). However, investigations on the impact of H2S in porewater on the speciation, distribution toxicity of heavy metals are very limited. Therefore, it is essential to delineate the relationship between sulfide and heavy metals in porewater.
Heavy metal circulation during mass exchange between continental shelf and open sea is an important part of global material cycle process. The Yellow Sea (YS) and East China Sea (ECS) is one of the largest continental shelves in the world with a total surface area of 1.2×106 km2, of which ~75% are in 50–100-m water depths. The YS and ECS not only have a wide shelf, typical muddy areas, complex current systems (coastal and warm), and diverse organisms, but also influenced by the Changjiang (Yangtze) River and Huanghe (Yellow) River having large sediment discharge (Yang and Liu, 2007; Liu et al., 2014). Meanwhile, due to the adjacency to the most economically developed region of China, large amounts of metals from various anthropogenic sources are released into the ECS and YS via atmospheric deposition and land runoff (Duce et al., 1991; McKee et al., 2004; Viers et al., 2009). Therefore, the ECS and YS is an ideal place to study the influence of natural and anthropogenic factors on heavy metals biogeochemistry. To study the chemical speciation, distribution, influence factor of heavy metals and evaluate the relationship between H2S and potential heavy metal hazards in ECS and YS sediments, vertical distribution of heavy metals (Cd, Cr, Cu, Pb, Zn, As, Co, Ni, and Mn) in porewater and the heavy metal fractionation (acid soluble, reducible, oxidizable, and residual fraction) were studied.
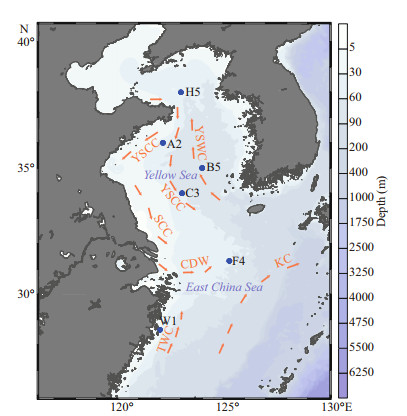
2 MATERIAL AND METHOD 2.1 Sediment samplingIn total, 12 sediment cores from six stations were collected in the ECS (F4, middle area of the ECS; W1, Zhejiang coastal region) and YS (H5, northern Yellow Sea; A2, southeastern coast of Shandong Peninsula; B5, west coast of South Korea; C3, middle of the YS) on board the R/V Dongfanghong 2 from 25 June to 12 July, 2018 (Fig. 1). At each site, two parallel sediment cores were sampled via PVC core liners (10-cm inner diameter; 20-cm long) and covered with overlying water immediately. Upon retrieval, cores were carefully inspected and only those with a visually undisturbed surface were used for further measurements. Well-preserved sediment samples for microsensor H2S profiling were measured using commercial micro-electrodes (20-μm tip diameter, Unisense A.S., Denmark) at in situ temperatures to avoid artefacts. The other sediment cores were sliced every 2 cm and porewater was collected using Rhizon Soil Moisture Samplers under N2 in a glove bag to prevent any oxidation of the reduced species present in sediments. Sediment slices were sealed in plastic bags and extracted porewater samples were transferred into 10-mL polyethylene plastic bottles soaked in nitric acid for more than 24 h (Ji et al., 2018). All of the above pretreated samples were sealed and stored at 4 ℃ prior to analyses.
|
| Fig.1 Locations of sampling stations and circulation system YSCC: Yellow Sea Coastal Current; YSWC: Yellow Sea Warm Current; SCC: Subei Coastal Current; CDW: Changjiang (Yangtze) Diluted Water; TWC: Taiwan Warm Current; KC: Kuroshio Current. The color bar from white to blue indicates that the water depth is gradually increasing. |
Samples were analyzed immediately after they arrived at laboratory. Identification of metals (Cd, Pb, Cr, Zn, As, Co, Cu, Mn, and Ni) in porewater was carried out using inductively coupled plasma mass spectrometer (ICP-MS) (ELAN DRC Ⅱ, Hong Kong) in accordance with the China's National Standard Method (State Environmental Protection Administration of China (SEPA), 2004). Samples for SO42- analysis were diluted 10 times and measured by ion selective electrode method with an ion chromatograph (Dionex ICS3000, USA). A total organic carbon analyzer (TOC-VCPH, Shimadzu, Japan) was used to measure total carbon (TC) and dissolved organic carbon (DOC), and DOC content was obtained by first acidifying the sample to remove the dissolved inorganic carbon (DIC), then subtracting the DOC from the TC to obtain the DIC content (Kao et al., 2003).
2.3 Analysis of solid phase chemistrySediment slices were first lyophilized in a freeze dryer, and then ground in an agate mortar. The grain size of sediments at all depths was measured with a laser particle size meter (Marlvern Mastersizer 2000F, Britain) after removing shell and other larger particles. Dried samples for total organic carbon (TOC) and metal contents determination were passed through a 100-mesh sieve and mixed. The metals (Cd, Cr, Cu, Pb, Zn, As, Co, Ni, and Mn) in the YS and ECS were continuously extracted using a modified Community Bureau of Reference (BCR) three-step leaching method (Rauret et al., 1999; Gao and Chen, 2012). In this study, the studied metals were divided into the following four groups: (1) acid soluble fraction (weak acid sensitive and exchangeable): easy to dissolve, migrate, and transform, reflecting the impacts of human sewage discharge and biotoxicity; combine with carbonate, highly sensitive to pH; (2) reducible fraction (combine with iron-manganese oxides): can reflect environmental pollution from human activities; (3) oxidizable fraction (bind to sulfides and organic matter): reflect the results of aquatic activities and human discharge of organic-rich wastewater; and (4) residual fraction: generally present in lithogenic minerals such as silicates, primary, and secondary minerals, the result of natural geological aeolian deposits. They are not easily released under natural conditions, can be stabilized in sediments for a long time, and are not easily absorbed by plants; the residual fraction is affected by mineral composition, rock weathering, and soil erosion (Han et al., 2005). The treated solute samples were evaluated by ICPMS (Perkin-Elmer Optima 7000 DV, USA). In addition, atomic emission spectrometry (Thermo IRIS Intrepid Ⅱ) was used to analyze the Al content and calculate the enrichment factor (EF) for each element in sediments. TOC and total nitrogen (TN) were measured with elemental analyzer (Vario Micro cube, Germany) after removing the inorganic carbon by adding HCl.
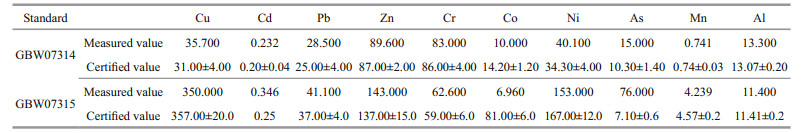
2.4 Statistical analysisOne-way analysis of variance (ANOVA), Tukey's test, and the two-sample t-test were performed to demonstrate the differences between treatments. SPSS 22.0 (SPSS Inc. Chicago, IL, USA) was used for regression analysis. A P < 0.05 was considered statistically significant. R packages: Corrplot and Ocean Data View 5.3.4 were used for graphic works. Standard substances (GBW07314, GBW07315) were used to test the accuracy of metal measurement, the method error was controlled within 5% (Table 1). In addition, the pollution load index (Ipl) (Tomlinson et al., 1980), geoaccumulation index (Igeo) (Mǜller, 1969), ecological harm coefficient (Eri) (Hakanson 1980), enrichment factor (EF) (Kaushik et al., 2009; Sakan et al., 2009), and mean effects range-median (ERM) (Carr et al., 1996; Zhang et al., 2019) were also applied to assess the environment effects of heavy metals in ECS and YS sediments. Furthermore, the upper continental crust values (Taylor and McLennan, 1995) were used as references for unpolluted sediments, and the Earth's average shales metal content (Turekian and Wedepoh, 1961) was chosen as the metal background concentration for indicators calculation (Al was chosen for normalization to calculate EF).
|
The research stations spanned in a large area and are affected by various natural and human factors, the sediments characteristics differ from area to area, thus the grain size compositions, TOC, and TON at each station were measured and the results are shown in Fig. 2. The mean percentage of sand content in H5 and B5 was high, while in A2, C3, F4, and W1, clay content was high. Sand content in sediments was the lowest at W1 station. TOC content showed a down-ward trend at the depth of 0–2 cm. TOC content unevenly distributed in C3, F4, and W1 in depth, while decrease from top to bottom in other stations. The distribution trend of TN was similar to TOC and the mean content is shown in Table 2. The TOC% was positively correlated with clay composition (R=0.588, P < 0.001), but negatively related with sand content (R=-0.625, P < 0.001), revealing that the enrichment capacity of fine-grain was higher than coarse-grain (Xing et al., 2011). The value of the carbon/nitrogen ratio (TOC/TN) was used to determine the source of organic: TOC/TN > 12, terrestrial organics; TOC/ TN < 8, marine-derived organic matter (Sampei and Matsumoto, 2001). Values of TOC/TN are shown in Table 2. Organic matter in W1 was influenced by land derived input, which is closer to land while marine plankton provided more organic matter for the sediments of B5. Organic matter in other stations was affected by the combination of two factors.

|
| Fig.2 Vertical profile of sediments grain size composition, total organic carbon concentration and total nitrogen concentration |
The vertical distributions of SO42-, DIC, and DOC in sediment porewater are shown in Fig. 3. There were no obvious vertical variations of SO42- and DIC in the sediment porewater within 10 cm. DIC concentrations were lower than that of DOC except W1 and B5. Furthermore, a markedly hypoxic in the Fujian and Zhejiang coastal region during the summer was inferred based on bottom O2 concentrations at W1 (Table 2).

|
| Fig.3 Vertical profiles for SO42-, DIC, and DOC at sampling stations |
H2S porewater distributions in this study are shown in Fig. 4. Generally, H2S concentration was below the detection limit (0.1 μmol/L) in the surface sediments because of the high oxygen concentration (Holmer and Storkholm, 2001). H2S was increasing gradually with the increase of depth and usually reached a maximum at 3–5 cm. The maximum H2S values (H2SMAX) are shown in Table 2, and the values and microsensor depth profiles of H2S were consistent with those in the southern North Sea (van de Velde et al., 2016). The formation of H2S was mainly resulted from sulfate and sulfate reducing bacteria, which relied on the degradable organic matter under anaerobic conditions. Therefore, the distribution of H2S was mainly associated with the content of organic matter and sulfate (Nedwell and Abram, 1978; Pester et al., 2012). We found that the maximum value of H2S in H5 (5.99 μmol/L) was higher than those in other tested stations, while maximum value of H2S was lower in A2 (0.50 μmol/L). Concentrations of DOC in porewater of H5 were high (> 11.0 mg/L), which promoted the activity of sulfate-reducing bacteria and enhanced the sulfate reduction rate. The content of SO42- in A2 was low (< 26.5 mmol/L), which was the limitation of sulfate reduction in A2. We found that the spatial distributions of H2S were similar at C3 and F4, probably because of the similar SO42- contents and DOC concentrations in these areas.

|
| Fig.4 Microsensor depth profiles for H2S at sampling stations |
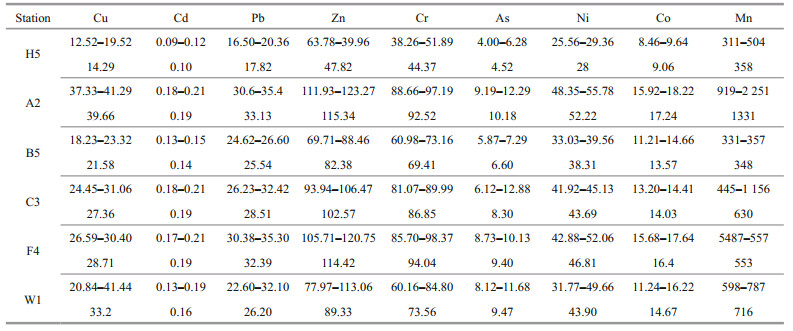
Vertical distributions of Cd, Cr, Cu, Pb, Zn, As, Co, Ni, and Mn in the porewater at different stations are depicted in Fig. 5. Cd concentrations were about 1 nmol/L in the uppermost porewater samples, and close to the detection limit with the increase of depth. The maximum Cr value was 0.078 μmol/L found in a sample at H5. There were no significant differences of mean content of Cu in the study area (0.039– 0.048 μmol/L). High values of Co and Ni were mainly concentrated at B5 and H5, while low values mainly appeared at A2. Co and Ni were significantly correlated (R=0.87, P < 0.01), indicating that they had consistent removal routes and sources. High values of Zn and Pb distributed at A2 while low values of them at H5 and B5. Zn concentrations were relatively higher compared with those of other heavy metals. Though Zn and Pb concentrations varied greatly, a significant positive correlation (R=0.50, P < 0.01) was found between them and their lowest values appeared at station B5. Meanwhile, Co and Ni had significant negative correlations with Zn and Pb in porewater samples (R=0.62, P < 0.01 for Co and Zn; R=0.42, P < 0.01 for Co and Pb; R=0.61, P < 0.01 for Ni and Zn; R=0.46, P < 0.01 for Ni and Zn), which is similar to a study in North Sea where Cd concentrations were very low in porewater (van de Velde et al., 2017). The contents of As were higher at C3 and W1. The Mn depth profiles of porewater varied greatly at different stations.

|
| Fig.5 Variability of dissolved metals (dCd, dCr, dCu, dPb, dZn, dCo, dNi, dAs) in porewater and total metals (TCd, TCr, TCu, TPb, TZn, TCo, TNi, TAs; sum of the four fractionations) in sediment in ECS and YS |
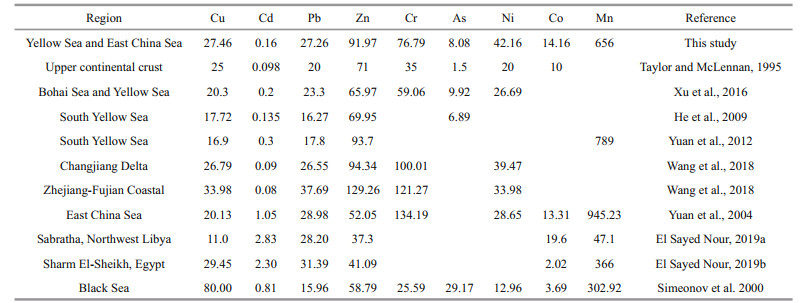
Total concentrations of metals within 10 cm are shown in Fig. 5. Low values of total Mn were mainly at B5 and H5, while the maximum Mn concentration occurred in the uppermost layer at B5. This Mn distribution trend in sediment was consistent with that in porewater. Average heavy metal concentrations in sediments were in a decreasing order of Zn > Cr > Ni > Cu > Pb > Co > As > Cd (Table 3). This trend is also consistent with results from studies in the Bohai Sea in which the mean Cd concentrations were the lowest and Zn concentrations were the highest (Gao and Chen, 2012). Overall, the ranges and mean metal concentrations we obtained in the study region were close to the values obtained by other studies (Table 4). The average concentrations of all heavy metals in the study area were higher than those of the upper continental crust. Compared with the investigation about heavy metals from the Black Sea, the mean concentrations of Cu, Cd, and As in our study were significantly low, while other metals were high. Moreover, Zn and Mn contents in sediment of YS and ECS were relatively high compared with some coast of the Africa in Libya and Egypt.
|
Different chemical speciation of heavy metals was closely associated with their mobility and biological toxicity (Jia et al., 2018). Metal fractionation in the investigated area was shown in Fig. 6. Residual fraction occupied the highest proportion, generally more than 75% except Mn. This result is consistent with previous research of the Bohai Sea that Cr, Ni, Pb, Zn, and Cu were preferentially bound to the residual phase (Li and Jing, 2015). Average percentages of acid soluble Cd, Co, Ni, Cr, Cu, Zn, As, and Pb were accounts for merely 1.57%, 6.4%, 3.3%, 0.9%, 4.1%, 3.2%, 3.2%, and 4.5% respectively. Moreover, the acid soluble fraction was generally considered the weakest bonded metals with most mobility and potential toxicity (Gao and Chen, 2012). Therefore, the low percentages of acid soluble metals indicated low mobility and environmental toxicity in the ECS and YS. Meanwhile, significant negative correlations were found between some acid soluble metals in solid and their concentrations in porewater (R=-0.95, P < 0.05 for Mn in H5; R=-0.98, P < 0.05 for Zn in A2). This further indicated that acid soluble fraction was important in metal reciprocal transformation between solid phase and water phase. For Cd, Cu, Zn, As, and Pb, the reducible group percentages (2.3% for Cd, 16.0% for Cu, 6.3% for Zn, 17.3% for As, 15.8% for Pb) were generally higher than the acid soluble and oxidizable groups. This may be due to reducible Fe-Mn minerals that have a strong ability to absorb these metals (Jain and Loeppert, 2000; Trivedi and Axe, 2000). When these heavy metals enter the sediment, they would be adsorbed onto iron and manganese (oxyhydr)oxides (Stockdale et al., 2010). In deeper layers, these heavy metals would be released into the porewater when (oxyhydr) oxides were reduced by organic matter (van de Velde et al., 2016). For Cr, Co, and Ni, the oxidable fraction was the main non-residual speciation, and the ratio accounted for 3.7%, 10.1%, 9.2%, respectively. This reflected high affinity of sulfide and organic matter to these metals (Huerta-Diaz et al., 1998). In addition, oxidable fraction mainly include two types, organic-bind metals and sulfide-bind metals, and there were spatial differences in the main components of oxidizable fraction of different metals. In the study area, significant positive correlations between oxidable fraction of Pb, Mn, and TOC were found in YS sediments. Meanwhile, significant positive correlation between oxidable fraction of Cd and TOC was also found in ECS sediments. The significant correlations between TOC and metals above revealed that organic-bind might be the primary form of the oxidable fraction of Pb and Mn in YS and Cd in ECS, while other metals might be reserved mainly via sulfide-bind (Chen et al., 2020).

|
| Fig.6 Different groups of heavy metals in ECS and YS sediments during summer |
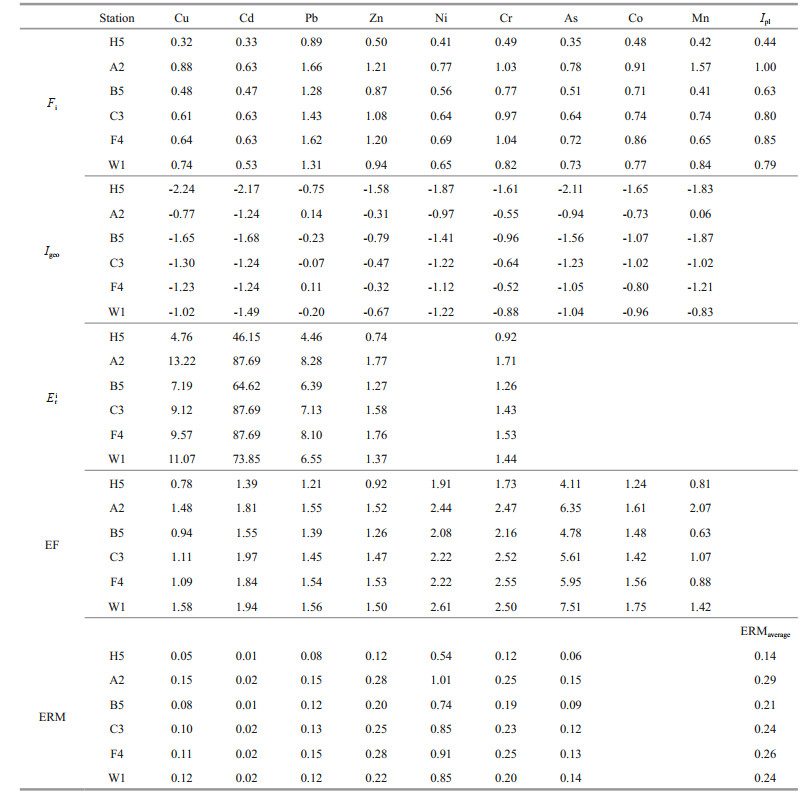
Five popular methods were used to assess sediment metals pollution and potential ecologic risk (Table 5). Ipl can directly reflect the contribution degree of each heavy metal to pollution which calculated by the contamination factor (Fi) (Tomlinson et al., 1980). In this study, the Ipl value was 1.00 in A2 and less than 1 in other stations. It meant that no metals pollution was found in most region, while moderate pollution in A2. Igeo is a quantitative index to study heavy metals pollution and is widely used to assess heavy metals pollution in sediments. After calculated, most Igeo values were less than 0 except Pb in F4 and Mn in A2, indicated that no pollution in most areas and slightly Pb pollution in F4 and Mn pollution in A2. Eri was also used to assess potential metals ecological risk, which was established by Swedish scholar Hakanson (1980) by applying the principle of sedimentology. According the criterion, Cu, Pb, Cr, and Zn were without ecological risk in study areas (Eri < 40), moderate potential ecologic risk (40 < Eri < 80) with Cd in H5, B5, and W1, and severe potential ecologic risk (80 < Eri < 120) in other stations. In addition, considering that heavy metals in sediments are always present in the form of complex mixtures, we used the mean ERM quotient method to determine the possible biological effects of combined toxicant groups. The calculated mean ERM quotients in this study were 0.14, 0.29, 0.21, 0.24, 0.26, and 0.24 in order from north to south, suggesting that the combination of heavy metals had a 21% possibility of being toxic (0.11 < ERM < 0.5) in the ECS and YS. The enrichment factor has been widely used to assess the influence of anthropogenic activities on sediment heavy metal contamination (Çevik et al., 2009; Kaushik et al., 2009). As shown in Table 5, Cu, Cd, Pb, Zn, Co, Ni, Mn, and Cr exhibited minor enrichment (1 < EF < 3) in most sampls, while As exhibited moderately severe enrichment in the study region during summer. Although the range and mean concentrations of As were normal, the As enrichment was still striking, especially at station W1 (EF=7.51), which was located in China's largest fishing ground of Zhoushan Archipelago. In general, the heavy metals pollution conditions in H5, B5, and W1 was mildly better than that in A2, C3, and F4. The concentration of Pb in F4, Mn in A2, Cd in A2, C3, F4, and As in all station have risen above environmental evaluation criteria. Therefore, we should control the inflow of these metals into the sea and decrease the ecological risk.
|
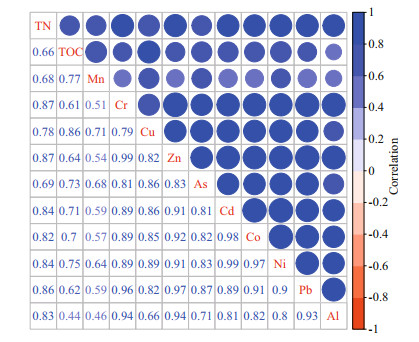
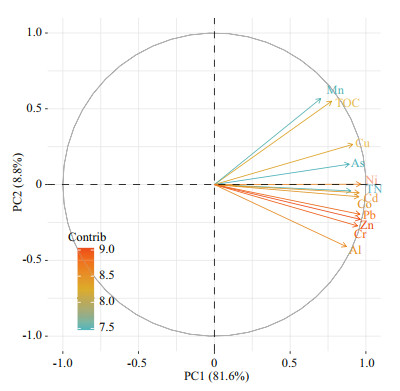
The potential relationship between heavy metals and their source issues can be revealed by correlation analyses and Principal Components Analysis (PCA) of TN, TOC, and metals (Figs. 7 & 8). All results indicate that there was a significant positive correlation between different heavy metals, indicating they had similar biogeochemical processes. The plot drawn with the scores of the first two principal components can be used for classifying metals. The first principal components (PC1) had very high positive loadings of all elements, which accounts for 81.6% of variance and showed an intense intrinsic connection between all studied parameters. Our result show TOC was correlated with composition of clay. Thus, the distribution of these metals was related with the natural weathering (Song et al., 2017). Principle component 2 (PC2=8.8%) classified metals into two categories, including a positive loading of Mn, Cu, and TOC, and a high negative loading of Cr, Zn, Pb, and Al. Cr, Zn, Pb, as well as Al, are more likely originated from sediment release. Given the EF results of As, the study area is generally enriched by As. Therefore, PC2 can be considered as an anthropogenic (Zhao et al., 2020).
|
| Fig.7 Correlation analyses between total nitrogen (TN), total organic carbon (TOC), and heavy metals in sediments of YS and ECS The color bar from blue to orange-red means positive to negative correlation; the darker the color, the stronger the correlation. |
|
| Fig.8 Component plot of total nitrogen (TN), total organic carbon (TOC), and heavy metals in sediments of YS and ECS The color bar from green to orange indicates that the contribution is gradually increasing. |
Numerous investigations have considered sulfide as an indicator for the assessment of heavy metals pollution in the aquatic environment, and sulfate reduction was of critical importance to control the levels of metals (Chen et al., 2020). Thus, it was needed to explore the influenced of H2S to metals behavior in porewater. In Fig. 6, there were obvious reduction in all stations at 3–5 cm depth of Pb and Zn, suggesting that H2S can react with them rapidly and this result is consistent with Morse and Luther Ⅲ (1999)'s finding about the kinetic and thermodynamic mechanisms of metal-sulfide interactions in porewater. Cu is liable to proceed oxidation-reduction reaction and bond with sulfide, resulting in the decrease of Cu content in 3–5-cm depth, where the H2SMAX occur. In addition, because of the low concentration of Cd, there was no obvious distribution trend and no obvious relationship was found between Cd and H2S in this study. As for Co, Ni, Cr, As, and Mn, these metals cannot react with H2S easily and quickly, hence these three metals had no corresponding distribution trends with H2S (Morse and Luther Ⅲ, 1999).
In solid phase, the low values of total metal concentrations appeared in H5 and B5, where H2S were relatively higher compared with other stations (Figs. 4 & 5). Meanwhile, significantly negatively correlations between H2SMAX and total heavy metal concentrations were found in this study (R=-0.97, P < 0.01 for Cr; R=-0.93, P < 0.01 for Co; R=-0.96, P < 0.01 for Ni; R=-0.97, P < 0.05 for Zn; R=-0.95, P < 0.01 for As; R=-0.98, P < 0.01 for Cd; R=-0.93, P < 0.01 for Pb). This may be explained by the relatively strong reduction environments at H5 and B5, which could be resulted in the reduction and dissolution of oxidized heavy metals. The dissolved heavy metals either migrated upward to the surface of sediments and precipitated after re-oxidation, or migrated downward to form insoluble substances under reduction conditions and accumulated back into the deeper layers (Barbanti et al., 1995). In addition, the H2SMAX was also negatively correlated with acid soluble Cu (R=-0.96, P < 0.05), Zn (R=-0.90, P < 0.05), reducible As (R=-0.84, P < 0.05), Zn (R= -0.93, P < 0.01), oxidizable Pb (R=-0.82, P < 0.05), As (R=-0.96, P < 0.05), and residual Cu (R=-0.88, P < 0.05), As (R=-0.90, P < 0.05). This indicated that acid soluble Cu and Zn, reducible As, Zn, as well as oxidizable Pb and As, were greatly influenced by H 2S.
In addition, the difference between metals extracted simultaneously (SEM) and AVS was generally used to indicate the toxic effects of heavy metals on aquatic organisms and sediment quality in previous studies (Hinkey and Zaidi, 2007; Zhuang and Gao, 2014). However, it needs to take a long time and variety of reagents for the measurement of AVS and SEM currently. In this study, a significant negative correlation was found between H2S and mean ERM quotients as shown in Fig. 9, indicating that H2S can also accurately reflect the potential biological effects of combined toxicant groups in sediments. Therefore, we could use H2S to assess potential biological toxicity risk of metals. According to the formula obtained via the relationship between H2S and ERM, when H2S > 7.1 μmol/L, ERM < 0.1, sediments will only have a 9% likelihood of being toxic. Compared with AVS, the determination of H2S is simple and fast. Therefore, we suggest using the H2S content for the prejudgment of bio-toxic of heavy metals in sediments.
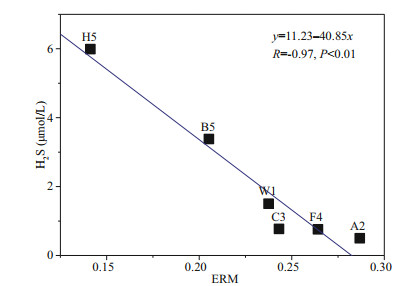
|
| Fig.9 Relationship between H2SMAX and mean ERM quotients in ECS and YS sediments during summer |
In this study, profiles of H2S, SO42-, DOC, TOC, TN along with fractions of Mn, Cd, Pb, Cr, Zn, As, Co, Cu, and Ni in ECS and YS sediments were collected and measured. Sulfate reduction mainly occurred in the North Yellow Sea and the west coast of South Korea sediments within 10 cm. H2S emerged gradually with the increase in depth and usually reached a maximum at around 3–5 cm. H2S was affected by DOC concentrations and sediment type. Sediment heavy metal concentrations were ranked in decreasing order of Mn > Zn > Cr > Ni > Cu > Pb > Co > As > Cd in the ECS and YS. Oxidizable Cr, Co, and Ni were the main non-residual component while reducible Cd, Cu, Zn, As, and Pb were the main non-residual component. For Mn, a considerable proportion was found among all four groups, while the residual group was dominated by other heavy metals at the sampling stations. Meanwhile, metals not only were affected by TOC (Mn, Pb, and Cd) and controlled by anthropogenic sources (Al, Cr, Zn, and Pb), but also respond to sulfide (Cd, Co, Pb, As, and Cu), and significantly negative correlations between H2S and total heavy metal concentrations were found in sediments within 10 cm. Our results suggest that heavy metals content in Southeastern coast of Shandong Peninsula were high (especially Mn and Cd), the control of metals emission from related industries needs to be strengthened in coastal areas. W1 station in ECS is an important fisheries area, so the hypoxia and As enrichment should call greater attention to the Fujian-Zhejiang coastal region during summer. In addition, the combination of heavy metals in this study could have a 21% possibility of being toxic and a significant negative correlation was found between H2SMAX and the mean ERM quotients, thus we suggest that H2S can be considered as an indicator for pre-estimating the potential ecological risk of heavy metals in sediments.
6 DATA AVAILABILITY STATEMENTAll data generated and/or analyzed during this study are included in this published article.
7 ACKNOWLEDGMENTWe are thankful to the captain and crew of the R/V Dong f anghong 2 for their help and cooperation during the expedition and in situ investigation.
Bak J, Jensen J, Larsen M M, Pritzl G, Scott-Fordsmand J. 1997. A heavy metal monitoring-programme in Denmark. Science of the Total Environment, 207(2-3): 179-186.
DOI:10.1016/S0048-9697(97)00262-3 |
Barbanti A, Bergamini M C, Frascari F, Miserocchi S, Ratta M, Rosso G. 1995. Diagenetic processes and nutrient fluxes at the sediment-water interface, northern Adriatic Sea, Italy. Marine and Freshwater Research, 46(1): 55-67.
DOI:10.1071/MF9950055 |
Bryan G W, Langston W J. 1992. Bioavailability, accumulation and effects of heavy metals in sediments with special reference to United Kingdom estuaries: a review. Environmental Pollution, 76(2): 89-131.
DOI:10.1016/0269-7491(92)90099-V |
Burton E D, Phillips I R, Hawker D W. 2006. Factors controlling the geochemical partitioning of trace metals in estuarine sediments. Soil and Sediment Contamination: An International Journal, 15(3): 253-276.
DOI:10.1080/15320380600646290 |
Carr S R, Chapman D C, Long E R, Windom H L, Thursby G, Sloane G M, Wolfe D A. 1996. Sediment quality assessment studies of Tampa Bay, Florida. Environmental Toxicology and Chemistry, 15(7): 1 218-1 231.
DOI:10.1002/etc.5620150730 |
Çevik F, Göksu M Z L, Derici O B, Fındık Ö. 2009. An assessment of metal pollution in surface sediments of Seyhan dam by using enrichment factor, geoaccumulation index and statistical analyses. Environmental Monitoring and Assessment, 152(1-4): 309.
DOI:10.1007/s10661-008-0317-3 |
Chapman P M, Wang F Y, Janssen C, Persoone G, Allen H E. 1998. Ecotoxicology of metals in aquatic sediments: binding and release, bioavailability, risk assessment, and remediation. Canadian Journal of Fisheries and Aquatic Sciences, 55(10): 2 221-2 243.
DOI:10.1139/f98-145 |
Chen Y Q, Ge J W, Huang T, Shen L L, Chu Z D, Xie Z Q. 2020. Restriction of sulfate reduction on the bioavailability and toxicity of trace metals in Antarctic lake sediments. Marine Pollution Bulletin, 151: 110807.
DOI:10.1016/j.marpolbul.2019.110807 |
Duce R A, Liss P S, Merrill J T, Atlas E L, Buat‐Menard P, Hicks B B, Miller J M, Prospero J M, Arimoto R, Church T M, Ellis W, Galloway J N, Hansen L, Jickells T D, Knap A H, Reinhardt K H, Schneider B, Soudine A, Tokos J J, Tsunogai S, Wollast R, Zhou M. 1991. The atmospheric input of trace species to the world ocean. Global Biogeochemical Cycles, 5(3): 193-259.
DOI:10.1029/91GB01778 |
El Sayed Nour H. 2019a. Assessment of heavy metals contamination in surface sediments of Sabratha, Northwest Libya. Arabian Journal of Geosciences, 12(6): 177.
DOI:10.1007/s12517-019-4343-y |
El Sayed Nour H. 2019b. Distribution, ecological risk, and source analysis of heavy metals in recent beach sediments of Sharm El-sheikh, Egypt. Environmental Monitoring and Assessment, 191(9): 546.
DOI:10.1007/s10661-019-7728-1 |
Gao X L, Chen C T A. 2012. Heavy metal pollution status in surface sediments of the coastal Bohai Bay. Water Research, 46(6): 1 901-1 911.
DOI:10.1016/j.watres.2012.01.007 |
Giblin A E, Howarth R W. 1984. Porewater evidence for a dynamic sedimentary iron cycle in salt marshes. Limnology and Oceanography, 29(1): 47-63.
DOI:10.4319/lo.1984.29.1.0047 |
Gu Y G, Wang Z H, Lu S H, Jiang S J, Mu D H, Shu Y H. 2012. Multivariate statistical and GIS-based approach to identify source of anthropogenic impacts on metallic elements in sediments from the mid Guangdong coasts, China. Environmental Pollution, 163: 248-255.
DOI:10.1016/j.envpol.2011.12.041 |
Hakanson L. 1980. An ecological risk index for aquatic pollution control.a sedimentological approach. Water Research, 14(8): 975-1 001.
DOI:10.1016/0043-1354(80)90143-8 |
Han C M, Wang L S, Gong Z Q, Xu H X. 2005. Chemical forms of soil heavy metals and their environmental significance. Chinese Journal of Ecology, 24(12): 1 499-1 502.
(in Chinese with English abstract) |
He J, Lü C W, Fan Q Y, Xue H X, Bao J H. 2011. Distribution of AVS-SEM, transformation mechanism and risk assessment of heavy metals in the Nanhai Lake in China. Environmental Earth Sciences, 64(8): 2 025-2 037.
DOI:10.1007/s12665-011-1022-z |
He Y, Men B, Yang X F, Li Y X, Xu H, Wang D S. 2017. Investigation of heavy metals release from sediment with bioturbation/bioirrigation. Chemosphere, 184: 235-243.
DOI:10.1016/j.chemosphere.2017.05.177 |
He Z P, Song J M, Zhang N X, Zhang P, Xu Y Y. 2009. Variation characteristics and ecological risk of heavy metals in the south Yellow Sea surface sediments. Environmental Monitoring and Assessment, 157(1-4): 515-528.
DOI:10.1007/s10661-008-0552-7 |
Hinkey L M, Zaidi B R. 2007. Differences in SEM-AVS and ERM-ERL predictions of sediment impacts from metals in two US Virgin Islands marinas. Marine Pollution Bulletin, 54(2): 180-185.
DOI:10.1016/j.marpolbul.2006.09.009 |
Holmer M, Storkholm P. 2001. Sulphate reduction and sulphur cycling in lake sediments: a review. Freshwater Biology, 46(4): 431-451.
DOI:10.1046/j.1365-2427.2001.00687.x |
Huerta-Diaz M A, Tessier A, Carignan R. 1998. Geochemistry of trace metals associated with reduced sulfur in freshwater sediments. Applied Geochemistry, 13(2): 213-233.
DOI:10.1016/S0883-2927(97)00060-7 |
Jain A, Loeppert R H. 2000. Effect of competing anions on the adsorption of arsenate and arsenite by ferrihydrite. Journal of Environmental Quality, 29(5): 1 422-1 430.
DOI:10.2134/jeq2000.00472425002900050008x |
Ji H B, Li H X, Zhang Y, Ding H J, Gao Y, Xing Y X. 2018. Distribution and risk assessment of heavy metals in overlying water, porewater, and sediments of Yongding River in a coal mine brownfield. Journal of Soils and Sediments, 18(2): 624-639.
DOI:10.1007/s11368-017-1833-y |
Jia Y L, Chen W, Zuo Y X, Lin L Z, Song L R. 2018. Heavy metal migration and risk transference associated with cyanobacterial blooms in eutrophic freshwater. Science of the Total Environment, 613-614: 1 324-1 330.
DOI:10.1016/j.scitotenv.2017.09.180 |
Kao S J, Lin F J, Liu K K. 2003. Organic carbon and nitrogen contents and their isotopic compositions in surficial sediments from the East China Sea shelf and the southern Okinawa Trough. Deep Sea Research Part Ⅱ: Topical Studies in Oceanography, 50(6-7): 1 203-1 217.
DOI:10.1016/S0967-0645(03)00018-3 |
Kaushik A, Kansal A, Santosh, Meena, Kumari S, Kaushik C P. 2009. Heavy metal contamination of river Yamuna, Haryana, India: assessment by metal enrichment factor of the sediments. Journal of Hazardous Materials, 164(1): 265-270.
DOI:10.1016/j.jhazmat.2008.08.031 |
Li H B, Cao M H, Wu D S, Yang L F, Fang D. 2017. Stabilization efficiency and mechanism of heavy metals in contaminated lake sediments by ferrous sulfide. Chinese Journal of Environmental Engineering, 11(5): 3 258-3 263.
(in Chinese with English abstract) |
Li X, Jing L. 2015. Speciation and degrees of contamination of metals in sediments from upstream and downstream reaches along the catchment of the southern Bohai Sea, China. International Journal of Environmental Research and Public Health, 12(7): 7 959-7 973.
DOI:10.3390/ijerph120707959 |
Liu J D, Liu J, Saito Y, Yang Z G, Yue B J, Wang H, Kong X H. 2014. Sedimentary evolution of the holocene subaqueous clinoform off the southern Shandong Peninsula in the Western South Yellow Sea. Journal of Ocean University of China, 13(5): 747-760.
DOI:10.1007/s11802-014-2227-z |
Liu J W, Liu X S, Wang M, Qiao Y L, Zheng Y F, Zhang X H. 2015. Bacterial and Archaeal communities in sediments of the north Chinese marginal seas. Microbial Ecology, 70(1): 105-117.
DOI:10.1007/s00248-014-0553-8 |
Machado W, Carvalho M F, Santelli R E, Maddock J E L. 2004. Reactive sulfides relationship with metals in sediments from an eutrophicated estuary in Southeast Brazil. Marine Pollution Bulletin, 49(1-2): 89-92.
DOI:10.1016/j.marpolbul.2004.01.012 |
Malkin S Y, Rao A M F, Seitaj D, Vasquez-Cardenas D, Zetsche E M, Hidalgo-Martinez S, Boschker H T S, Meysman F J R. 2014. Natural occurrence of microbial Sulphur oxidation by long-range electron transport in the seafloor. The ISME Journal, 8(9): 1 843-1 854.
DOI:10.1038/ismej.2014.41 |
Massas I, Ehaliotis C, Gerontidis S, Sarris E. 2009. Elevated heavy metal concentrations in top soils of an Aegean island town (Greece): total and available forms, origin and distribution. Environmental Monitoring and Assessment, 151(1-4): 105-116.
DOI:10.1007/s10661-008-0253-2 |
McKee B A, Aller R C, Allison M A, Bianchi T S, Kineke G C. 2004. Transport and transformation of dissolved and particulate materials on continental margins influenced by major rivers: benthic boundary layer and seabed processes. Continental Shelf Research, 24(7-8): 899-926.
DOI:10.1016/j.csr.2004.02.009 |
Morse J W, Luther Ⅲ G W I. 1999. Chemical influences on trace metal-sulfide interactions in anoxic sediments. Geochimica et Cosmochimica Acta, 63(19-20): 3 373-3 378.
DOI:10.1016/S0016-7037(99)00258-6 |
Mǜller G. 1969. Index of geoaccumulation in sediments of the Rhine River. GeoJournal, 2(3): 109-118.
|
Nagajyoti P C, Lee K D, Sreekanth T V M. 2010. Heavy metals, occurrence and toxicity for plants: a review. Environmental Chemistry Letters, 8(3): 199-216.
|
Nedwell D B, Abram J W. 1978. Bacterial sulphate reduction in relation to sulphur geochemistry in two contrasting areas of saltmarsh sediment. Estuarine and Coastal Marine Science, 6(4): 341-351.
DOI:10.1016/0302-3524(78)90126-3 |
Pester M, Knorr K H, Friedrich M W, Wagner M, Loy A. 2012. Sulfate-reducing microorganisms in wetlands—fameless actors in carbon cycling and climate change. Frontiers in Microbiology, 3: 72.
DOI:10.3389/fmicb.2012.00072 |
Rauret G, López-Sánchez J F, Sahuquillo A, Rubio R, Davidson C, Ure A, Quevauviller P. 1999. Improvement of the BCR three step sequential extraction procedure prior to the certification of new sediment and soil reference materials. Journal of Environmental Monitoring, 1(1): 57-61.
DOI:10.1039/A807854H |
Sakan S M, Đorđević D S, Manojlović D D, Predrag P S. 2009. Assessment of heavy metal pollutants accumulation in the Tisza River sediments. Journal of Environmental Management, 90(11): 3 382-3 390.
DOI:10.1016/j.jenvman.2009.05.013 |
Sampei Y, Matsumoto E. 2001. C/N ratios in a sediment core from Nakaumi Lagoon, Southwest Japan—usefulness as an organic source indicator. Geochemical Journal, 35(3): 189-205.
DOI:10.2343/geochemj.35.189 |
Shyleshchandran M N, Mohan M, Ramasamy E V. 2018. Risk assessment of heavy metals in Vembanad Lake sediments (south-west coast of India), based on acid-volatile sulfide (AVS)-simultaneously extracted metal (SEM) approach. Environmental Science and Pollution Research, 25(8): 7 333-7 345.
DOI:10.1007/s11356-017-0997-8 |
Simeonov V, Massart D L, Andreev G, Tsakovski S. 2000. Assessment of metal pollution based on multivariate statistical modeling of 'hot spot' sediments from the Black Sea. Chemosphere, 41(9): 1 411-1 417.
DOI:10.1016/S0045-6535(99)00540-8 |
Song H Y, Liu J Q, Yin P, Zhang Y. 2017. Distribution, enrichment and source of heavy metals in Rizhao offshore area, southeast Shandong province. Marine Pollution Bulletin, 119(2): 175-180.
DOI:10.1016/j.marpolbul.2017.04.017 |
State Environmental Protection Administration of China (SEPA). 2004. Marine Sediment Quality (GB 18668-2002). Standards Press of China, Beijing.
(in Chinese)
|
Stockdale A, Davison W, Zhang H, Hamilton-Taylor J. 2010. The association of cobalt with iron and manganese (oxyhydr)oxides in marine sediment. Aquatic Geochemistry, 16(4): 575-585.
DOI:10.1007/s10498-010-9092-1 |
Taylor S R, McLennan S M. 1995. The geochemical evolution of the continental crust. Reviews of Geophysics, 33(2): 241-265.
DOI:10.1029/95RG00262 |
Tessier A P, Campbell P G C, Bisson M X. 1979. Sequential extraction procedure for the speciation of particulate trace metals. Analytical chemistry, 51(7): 844-851.
DOI:10.1021/ac50043a017 |
Tomlinson D L, Wilson J G, Harris C R, Jeffrey D W. 1980. Problems in the assessment of heavy-metal levels in estuaries and the formation of a pollution index. Helgoländer Meeresuntersuchungen, 33(1): 566-575.
DOI:10.1007/BF02414780 |
Trivedi P, Axe L. 2000. Modeling Cd and Zn sorption to hydrous metal oxides. Environmental Science & Technology, 34(11): 2 215-2 223.
DOI:10.1021/es991110c |
Turekian K K, Wedepoh K H. 1961. Distribution of the elements in some major units of the Earth's crust. GSA Bulletin, 72(2): 175-192.
DOI:10.1130/0016-7606(1961)72[175:DOTEIS]2.0.CO;2 |
van de Velde S J, Hylén A, Kononets M, Marzocchi U, Leermakers M, Choumiline K, Hall P O J, Meysman F J R. 2020. Elevated sedimentary removal of Fe, Mn, and trace elements following a transient oxygenation event in the Eastern Gotland Basin, central Baltic Sea. Geochimica et Cosmochimica Acta, 271: 16-32.
DOI:10.1016/j.gca.2019.11.034 |
van de Velde S, Callebaut I, Gao Y, Meysman F J R. 2017. Impact of electrogenic sulfur oxidation on trace metal cycling in a coastal sediment. Chemical Geology, 452: 9-23.
DOI:10.1016/j.chemgeo.2017.01.028 |
van de Velde S, Lesven L, Burdorf L D W, Hidalgo-Martinez S, Geelhoed J S, Van Rijswijk P, Gao Y, Meysman F J R. 2016. The impact of electrogenic sulfur oxidation on the biogeochemistry of coastal sediments: a field study. Geochimica et Cosmochimica Acta, 194: 211-232.
DOI:10.1016/j.gca.2016.08.038 |
Varol M, Şen B. 2012. Assessment of nutrient and heavy metal contamination in surface water and sediments of the upper Tigris River, Turkey. CATENA, 92: 1-10.
DOI:10.1016/j.catena.2011.11.011 |
Viers J, Dupré B, Gaillardet J. 2009. Chemical composition of suspended sediments in World Rivers: new insights from a new database. Science of the Total Environment, 407(2): 853-868.
DOI:10.1016/j.scitotenv.2008.09.053 |
Wang C L, Zou X Q, Feng Z Y, Hao Z, Gao J H. 2018. Distribution and transport of heavy metals in estuarine-inner shelf regions of the East China Sea. Science of the Total Environment, 644: 298-305.
DOI:10.1016/j.scitotenv.2018.06.383 |
Xing L, Tao S Q, Zhang H L, Liu Y G, Yu Z G, Zhao M X. 2011. Distributions and origins of lipid biomarkers in surface sediments from the southern Yellow Sea. Applied Geochemistry, 26(8): 1 584-1 593.
DOI:10.1016/j.apgeochem.2011.06.024 |
Xu X D, Cao Z M, Zhang Z X, Li R H, Hu B Q. 2016. Spatial distribution and pollution assessment of heavy metals in the surface sediments of the Bohai and Yellow Seas. Marine Pollution Bulletin, 110(1): 596-602.
DOI:10.1016/j.marpolbul.2016.05.079 |
Yang Z S, Liu J P. 2007. A unique Yellow River-derived distal subaqueous delta in the Yellow Sea. Marine Geology, 240(1-4): 169-176.
DOI:10.1016/j.margeo.2007.02.008 |
Yuan C G, Shi J B, He B, Liu J F, Liang L N, Jiang G B. 2004. Speciation of heavy metals in marine sediments from the East China Sea by ICP-MS with sequential extraction. Environment International, 30(6): 769-783.
DOI:10.1016/j.envint.2004.01.001 |
Yuan H M, Song J M, Li X G, Li N, Duan L Q. 2012. Distribution and contamination of heavy metals in surface sediments of the South Yellow Sea. Marine Pollution Bulletin, 64(10): 2 151-2 159.
DOI:10.1016/j.marpolbul.2012.07.040 |
Zhang H L, Walker T R, Davis E, Ma G F. 2019. Ecological risk assessment of metals in small craft harbour sediments in Nova Scotia, Canada. Marine Pollution Bulletin, 146: 466-475.
DOI:10.1016/j.marpolbul.2019.06.068 |
Zhao L, Liu J Q, Cai G Q, Huang L, Luo W D. 2020. Distribution, source, and pollution assessment of heavy metals in Sanya offshore area, south Hainan Island of China. Marine Pollution Bulletin, 160: 111561.
DOI:10.1016/j.marpolbul.2020.111561 |
Zhuang W, Gao X L. 2014. Assessment of heavy metal impact on sediment quality of the Xiaoqinghe estuary in the coastal Laizhou Bay, Bohai Sea: inconsistency between two commonly used criteria. Marine Pollution Bulletin, 83(1): 352-357.
DOI:10.1016/j.marpolbul.2014.03.039 |
 2021, Vol. 39
2021, Vol. 39