Institute of Oceanology, Chinese Academy of Sciences
Article Information
- PENG Saijun, LIU Qingqing, WANG Lei, SUN Tingting, SHIGANOVA Tamara, DONG Zhijun
- Molecular identification and population differentiation of Aurelia spp. ephyrae in sea cucumber aquaculture ponds of northern China
- Journal of Oceanology and Limnology, 39(3): 989-996
- http://dx.doi.org/10.1007/s00343-020-0022-9
Article History
- Received Jan. 13, 2020
- accepted in principle Apr. 23, 2020
- accepted for publication Jun. 10, 2020
2 University of Chinese Academy of Sciences, Beijing 100049, China;
3 Center for Ocean Mega-Science, Chinese Academy of Sciences, Qingdao 266071, China;
4 Shirshov Institute of Oceanology Russian Academy of Sciences, Moscow 117997 Nakhimovskyi pr., 36, Russia
Jellyfish blooms have become frequent in the last few decades as a result of global climate change and human activities that have led to local ecological shifts and had harmful effects on biodiversity and commercial stocks (Arai, 2001; Mills, 2001; Purcell, 2005; Uye, 2008; Richardson et al., 2009; Falkenhaug, 2014). Representatives of genus Aurelia are among the most common species that form jellyfish blooms. There have been many reports of Aurelia species blooms across the world, including in China, Japan, Korea, Spain, and Tunisia (Toyokawa et al., 2000; Dong et al., 2010; Baxter et al., 2011; Uye, 2011; Wang et al., 2012; Purcell et al., 2013; Bosch-Belmar, et al., 2016). The genus Aurelia belongs to Cnidaria, Scyphozoa, Semaeostomeae, Ulmaridae and is considered the most common genus of scyphozoan jellyfish worldwide (Lucas, 2001; Chinese Zoology Editorial Committee, Chinese Academy of Sciences, 2002; Schroth et al., 2005). Aurelia spp. have a metagenetic life cycle with an asexual benthic generation and sexual planktonic medusae (meroplanktonic species) (Lucas, 2001).
Many researchers believed in the past that genus Aurelia consisted of 3 valid species (Aurelia aurita, Aurelia limbata, and Aurelia labiata) and 10 cryptic species (Aurelia sp.1–Aurelia sp.10) globally (Dawson and Jacobs, 2001; Schroth et al., 2002; Dawson, 2003, 2005; Ki et al., 2008). However, Scorrano et al. (2017) showed that Aurelia sp.1 (Aurelia coerulea) and Aurelia sp.8 (Aurelia solida) can be considered valid species, while Aurelia sp.5 (Aurelia relicta sp. nov.) must be described as a new species. There are geographical differences in distribution between species. For example, A. coerulea (Aurelia sp.1) occurs mainly in warmtemperate regions, including China, Australia, California, France, Japan, and Korea, while Aurelia sp.2 occurs mainly in Brazil; Aurelia sp.3 and sp.6 in Palau; Aurelia sp.4 in Indonesia, Palau, and Hawaii; A. relicta sp. nov. (Aurelia sp.5) in the Mljet Lake of Croatia; Aurelia sp.7 in New Zealand and Tasmania; A. solida (Aurelia sp.8) in the Northern Adriatic Sea and the Gulf of Lyon; Aurelia sp.9 in the Gulf of Mexico; and Aurelia sp.10 in Alaska and European seas (Dawson and Jacobs, 2001; Schroth et al., 2002; Dawson, 2003, 2005; Ki et al., 2008; Ramšak et al., 2012; Dong et al., 2015; Scorrano et al., 2017). From 2003 to 2014, Aurelia spp. medusae and ephyrae blooms frequently occurred in the Yellow Sea and Bohai Sea in China, including in aquaculture ponds, water power plants, docks, and so on. In coastal sea cucumber culture ponds, the typical blooms of Aurelia spp. ephyrae have been observed. The local farmers call them "red jellyfish" because during blooms they cause the surface of the aquaculture ponds to appear red.
Mitochondrial DNA is widely used in the study of species population genetic analysis due to its simple structure, high base mutation rate, rapid evolution rate, high sensitivity, maternal inheritance, and general lack of recombination (Brown et al., 1979; Whitmore et al., 1994; Hallerman, 2003). Mitochondrial 16S ribosomal RNA (rRNA) is a very commonly used molecular marker. Dong et al. (2017) determined by phylogenetic analyses of 16S rRNA that the "red jellyfish" ephyrae, which appeared in the culture ponds of the Rongcheng Shidao area in China belonged to A. coerulea. The ephyrae in aquaculture ponds is hypothesized to enter the ponds via water flow from the Yellow Sea and Bohai Sea. However, genetic analysis of Aurelia spp. medusae and ephyrae has not yet been carried out in other culture ponds. This study analyzes the genetic structure and diversity of the Aurelia spp. population in sea cucumber aquaculture ponds along the coast of the Bohai and Yellow Seas based on the 16S rRNA gene. In total, 81 Aurelia spp. ephyrae were collected from multiple sea cucumber culture ponds in 4 regions and 85 Aurelia spp. medusae were collected from 4 coastal locations. The aim of our study was to identify species of Aurelia spp. ephyrae in sea cucumber aquaculture ponds in the Bohai and Yellow Seas. Finally, the genetic differentiation between ephyrae populations collected in the sea cucumber aquaculture ponds and A. coerulea medusae collected in coastal waters were also revealed using the 16S rRNA gene.
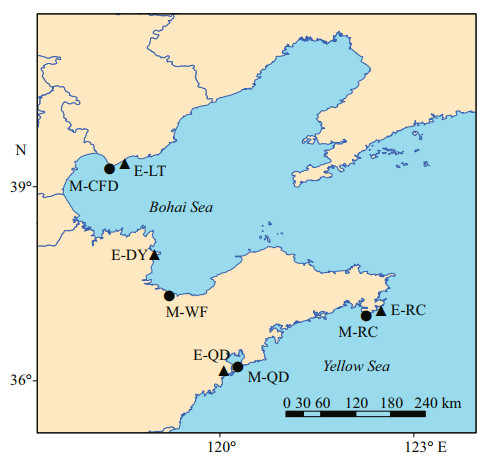
2 MATERIAL AND METHOD 2.1 Sample collectionFrom 2014 to 2016, a total of 166 Aurelia spp. individuals were collected in the Bohai Sea and Yellow Sea during the local jellyfish blooming periods (Supplementary Table S1; Fig. 1). Of these, 85 medusae were collected across four geographic locations: Caofeidian (M-CFD), Qingdao (M-QD), Rongcheng (M-RC), and Weifang (M-WF), while 81 ephyrae were obtained from Apostichopus japonicus aquaculture ponds in Laoting (E-LT), Qingdao (EQD), Rongcheng (E-RC), and Dongying (E-DY). The whole ephyrae and the medusae tissue extracted from the bell margin were preserved in 99% ethanol and then stored at -20 ℃ until DNA extraction.
|
| Fig.1 Sampling sites of Aurelia spp. in Chinese coastal waters ● represents A. coerulea medusae, ▲ represents A. coerulea ephyrae. |
The genomic DNA of medusae tissue was extracted using the TIANamp Marine Animals DNA Kit (TIANGEN, Beijing, China), while the genomic DNA of ephyrae was extracted using the CTAB (Cetyltrimethyl Ammonium Bromide) method. DNA from both life stages was stored at -20 ℃. A region of the 16S rRNA gene was amplified using the published primers (16S-H 5′-CAT AAT TCA ACA TCG AGG-3′ and 16S-L 5′-GAC TGT TTA CCA AAA ACA TA-3′) (Ender and Schierwater, 2003). Polymerase Chain Reactions (PCR) were performed in a total volume of 50 μL containing 50–100-ng genomic DNA, 1×PCR buffer, 2.5-U Taq DNA polymerase, 1.5-mmol/L MgCl2, 0.2-mmol/L dNTPs, and 0.25-mmol/L primers. The protocol for 16S rRNA amplification was as follows: 5 cycles of 4 ℃ for 1 min, 45 ℃ for 50 s and 72 ℃ for 1 min; 30 cycles of 94 ℃ for 50 s, 50 ℃ for 1 min and 72 ℃ for 1 min; and a final elongation at 72 ℃ for 5 min. The PCR products were analyzed using electrophoresis on a 1% agarose gel, stained with GenecolourTM (Biotium, USA).
PCR products were purified and sequenced directly using ABI 3730 automated DNA sequencer at Shanghai Sangon Biological Engineering Technology & Service Co., Ltd., China. All PCR products were sequenced in both directions to obtain accurate sequences. 16S rRNA sequences were aligned using CLUSTALX 1.83, and were verified, edited and assembled with BioEdit 7.1. To ensure correct alignment, the sequences were conducted with MEGA 7.0.
2.3 Data analysesThe nucleotide composition and variation between sites were analyzed in MEGA 7.0 (Kumar et al., 2016). The haplotype diversity (Hd) and nucleotide diversity (π) were calculated using DnaSP 5.10 (Librado and Rozas, 2009). Levels of overall interpopulation differentiation as well as differentiation between different region populations and populationpairwise differentiation were estimated using Φ-statistics, which give an analogue of F-statistics calculated within the analysis of molecular variance (AMOVA) framework, calculated using the Arlequin 3.5 (Excoffier and Lischer, 2010). A median-joining network showing the intuitive and accurate relationships between the haplotypes was constructed using the Network 4.6 (Bandelt et al., 1999). The sequences of A. coerulea samples in this study can be downloaded from Genbank (MF981165–MF981181, Supplementary Table S2).
3 RESULT 3.1 Species identification and genetic variability16S rRNA sequences were obtained from 166 individuals. The aligned sequence length of the 16S rRNA sequences was 532 bp. BLASTn analysis indicated that both the ephyrae collected in the aquaculture ponds and medusae collected in coastal waters belong to A. coerulea.
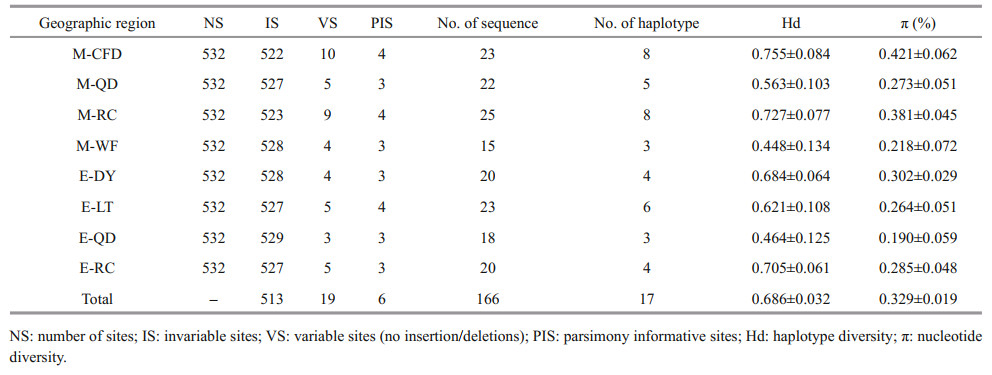
Nineteen polymorphic sites were detected in 532 sites, with a mutation rate of 3.57%, including 6 parsimony information sites, accounting for 1.13%, and 13 singlet nucleotide mutation sites, accounting for 2.44%. Seventeen haplotypes were defined in 19 polymorphic sites, defined as Hap 1–17. The genetic distance between haplotypes ranged from 0.2% to 1.5%, and the average genetic distance was 0.7%. The average contents of bases A, T, C, and G were 25.7%, 35.4%, 19.4%, and 19.5%, respectively. The A+T content (61.1%) was significantly greater than the C+G content (38.9%). The Hd and π of each geographic region are shown in Table 1. Across all samples, the haplotype diversity ranged from 0.448±0.134 to 0.755±0.084, while the nucleotide diversity ranged from 0.190%±0.059% to 0.421%±0.062%. Overall haplotype diversity in samples was 0.686±0.032, and the corresponding nucleotide diversity was 0.329%±0.019%, showing a higher haplotype diversity and lower nucleotide diversity. The highest haplotype diversity was found in the M-CFD population and the lowest in the M-WF population. The M-CFD population also had the highest nucleotide diversity and the E-QD population had the lowest. The haplotype diversity of ephyrae populations were generally lower than that of medusae populations with close sampling points. The Hd of E-LT was lower than M-CFD, the same results still existed in E-QD and M-QD, E-RC and M-RC.
|
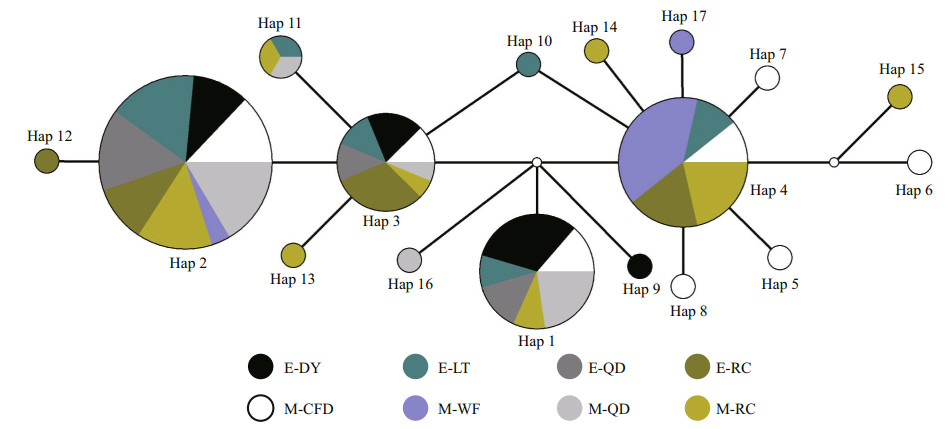
Based on the median-joining haplotype network method, a network relationship diagram of A. coerulea haplotypes was constructed (Fig. 2). There were 5 haplotypes (Hap 1, Hap 2, Hap 3, Hap 4, and Hap 11) that were found in individuals from more than one geographic region. Hap 2 was the most frequent haplotype, occurring across all eight geographic regions. Hap 1 was found in the M-CFD, M-QD, E-QD, E-DY, M-RC, and E-LT populations. Hap 3 occurred in all populations except M-WF. Hap 4 was found in the M-CFD, M-RC, M-WF, E-LT, and E-RC populations and Hap 11 occurred in the M-QD, M-RC, and E-LT populations. With the exception of E-QD, other populations had their own specific haplotypes. There was no significant correlation between the haplotypes and the geographic distribution of populations.
|
| Fig.2 Median-joining networks for all A. coerulea 16S rRNA haplotypes The color of the circle indicates the geographic region, and the size of the circle indicates the haplotype frequency. Each branch between any two shapes represents a single nucleotide substitution. |
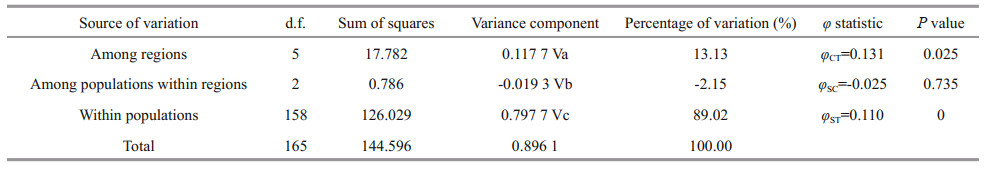
The analysis of molecular variance (AMOVA) revealed that 89.02% of the genetic variation occurred within populations (P < 0.01), whereas 13.13% occurred among regions (P < 0.05). Thus, the AMOVA revealed significant genetic differentiation between the six regions (CFD, QD, RC, WF, DY, LT) (φCT= 0.131, P=0.025), and extremely significant genetic differentiation within populations in the total samples (φST= 0.110, P=0) (Table 2). However, there was no significant genetic differentiation among populations within regions (P=0.735).
|
Population-pairwise Fst values ranged from -0.033 5 (M-RC/M-CFD) to 0.542 3 (E-QD/M-WF). These results indicated that the E-DY, E-LT, E-RC, M-RC, E-QD, M-QD, and M-CFD populations were significantly differentiated from the M-WF population (Fst range: 0.176 2 to 0.542 3, P < 0.05). The genetic differentiation between A. coerulea medusae collected in coastal waters and ephyrae populations collected in the sea cucumber aquaculture ponds was not significant. Moderate genetic differentiation existed between the M-WF population and the M-RC and M-CFD populations (0.15 < Fst < 0.25). Between the M-WF population and the E-DY, E-LT, E-RC, E-QD, M-QD populations, the Fst values were all greater than 0.25, and the corresponding Nm was between 0 and 1, indicating a large genetic differentiation between populations (Table 3). Nm>4 indicates more frequent gene exchange between groups.

|
Most researchers believed that the genus Aurelia, which includes 5 valid species, consists of at least 13 species and 16 genetic branches (Dawson and Jacobs, 2001; Dawson, 2003; Ki et al., 2008; Scorrano et al., 2017). A. aurita has the widest distribution, except the polar regions; therefore, unidentified Aurelia spp. have been named A. aurita in most previous reports. Previous research based on 16S rDNA and COI identified Aurelia spp. in Chinese seas as A. coerulea, but there is no definitive research which determines to which species or genetic branch the Aurelia spp. ephyrae found in sea cucumber culture ponds belong. BLASTn analysis indicated that the sequences identified in this study were highly similar (99%) to the sequences of A. coerulea reported by He et al. (2015) (Genbank accession number: KF962395) and Wang et al. (2013) (Genbank accession number: JX845344), indicating that the Aurelia spp. in this study were A. coerulea.
The genetic differentiation between A. coerulea ephyrae populations collected in the sea cucumber aquaculture ponds and medusae collected in coastal waters was not significant. Furthermore, among the ephyrae populations in sea cucumber aquaculture ponds and the medusae populations in the coastal waters with close sampling points, the haplotype diversity of ephyrae populations were generally lower than that of medusae populations. The Hd of E-LT was lower than M-CFD, the same results still existed in E-QD and M-QD, E-RC and M-RC. These results suggested that the ephyrae populations in the sea cucumber culture ponds were part of the same genetic group as the medusae populations in the coastal waters. It is likely that A. coerulea planulae of some haplotypes flow in the sea cucumber aquaculture ponds through tides or pumps and settle in the artificial structures because adult medusae of Aurelia frequently occur near the A. japonicus culture ponds during the summer. The water inlets and outlets in the sea cucumber aquaculture ponds are covered with nylon fishing nets to prevent the entrance of potential predators and the escape of farmed sea cucumbers; however, they cannot prevent the exchange of A. coerulea planulae between the A. japonicus culture ponds and coastal waters.
Phylogeographic analysis based on a network relationship diagram suggested that there was no significant correlation between the haplotypes and the distribution of geographic populations. However, pairwise fixation index values showed significant genetic differentiation between A. coerulea medusa population of WF and other populations, which means that there was a certain degree of gene flow among the 8 populations, but the dispersal of A. coerulea among WF and other locations was relatively limited. We speculate that this is related to biological characteristics of A. coerulea and marine transportation. A. coerulea, as a typical species of zooplankton, its ability to move long distances is relatively weak, and has the characteristics of alternated phenology and weak diffusion ability of polyps in its life cycle. The weak diffusion ability of benthic stages of seasonal meroplanktonic species, such as Rhizostoma octopus and A. coerulea, means they more likely to have distinct genetic population structures than holoplanktonic species, such as Pelagia noctiluca (Stopar et al., 2010; Ramšak et al., 2012; Lee et al., 2013). In addition, polyps, the key stage of the expansion of the A. coerulea populations, are often attached to artificial structures such as coastal docks and ports. Thus, the areas where A. coerulea medusae are gathered in large numbers are usually coastal waters and shoals areas, which are rare in deep-sea areas; this further limits the diffusion range of A. coerulea populations. Similarly, Li et al. (2016) found that there was significant genetic differentiation between Rhopilema esculentum sampled in the Yellow Sea and Bohai Sea. The Caofeidian population did not show significant genetic differentiation from the Yellow Sea populations, which may be due to the developed coastal transportation industry. Bolton and Graham (2006) proposed ballast water and hull carrying in marine transportation play an important role in importing new haplotypes or new species. Compared with Qingdao, Rongcheng and Caofeidian, the scale of coastal transportation in Weifang is still relatively insufficient, which allows Weifang population to have a certain degree of gene exchange with other groups, but also limits the frequency and scope of gene flow.
5 CONCLUSIONBased on mitochondrial 16S rRNA region, the ephyrae collected in the aquaculture ponds in the Bohai and Yellow Seas were identified as A. coerulea. The haplotype diversity and nucleotide diversity of the total population of A. coerulea were showing a higher haplotype diversity and lower nucleotide diversity. The haplotype diversity of ephyrae populations were generally lower than that of medusae populations with close sampling points. The genetic differentiation between ephyrae collected in the sea cucumber aquaculture ponds and A. coerulea medusae populations collected in coastal waters was not significant. Thus, the ephyrae populations in the sea cucumber culture ponds were part of the same genetic group as the medusae populations in the coastal waters. Phylogeographic analysis of the 16S rRNA region revealed that there was no significant correlation between the haplotypes and the geographic distribution of populations. Pairwise fixation index values showed significant genetic differentiation and limited gene flow between A. coerulea medusae population of Weifang and other populations.
6 DATA AVAILABILITY STATEMENTAll data generated and/or analyzed during this study are available from the corresponding author upon request.
Arai M N. 2001. Pelagic coelenterates and eutrophication: a review. Hydrobiologia, 451(1-3): 69-87.
DOI:10.1007/978-94-010-0722-1_7 |
Bandelt H J, Forster P, Röhl A. 1999. Median-joining networks for inferring intraspecific phylogenies. Molecular Biology and Evolution, 16(1): 37-48.
DOI:10.1093/oxfordjournals.molbev.a026036 |
Baxter E J, Rodger H D, McAllen R, Doyle T K. 2011. Gill disorders in marine-farmed salmon: investigating the role of hydrozoan jellyfish. Aquaculture Environment Interactions, 1(3): 245-257.
DOI:10.3354/aei00024 |
Bolton T F, Graham W M. 2006. Jellyfish on the rocks: Bioinvasion threat of the international trade in aquarium live rock. Biological Invasions, 8(4): 651-653.
DOI:10.1007/s10530-005-2017-z |
Bosch-Belmar M, Rabet C M, Dhaouadi R, Chalghaf R, Yahia M N D, Fuentes V, Piraino S, Yahia O K D. 2016. Jellyfish stings trigger gill disorders and increased mortality in farmed Sparus aurata (Linnaeus, 1758) in the Mediterranean sea. PLoS One, 11(4): e0154239.
DOI:10.1371/journal.pone.0154239 |
Brown W M, George M Jr, Wilson A C. 1979. Rapid evolution of animal mitochondrial DNA. Proceedings of the National Academy of Sciences of the United States of America, 76(4): 1967-1971.
DOI:10.1073/pnas.76.4.1967 |
Chinese Zoology Editorial Committee, Chinese Academy of Sciences. 2002. Fauna Sinica (Volume 27): Scyphozoa, Hydrozoa. Science Press, Beijing. p. 30-34. (in Chinese)
|
Dawson M N, Jacobs D K. 2001. Molecular evidence for cryptic species of Aurelia aurita (Cnidaria, Scyphozoa). The Biological Bulletin, 200(1): 92-96.
DOI:10.2307/1543089 |
Dawson M N. 2003. Macro-morphological variation among cryptic species of the moon jellyfish, Aurelia (Cnidaria: Scyphozoa). Marine Biology, 143(2): 369-379.
DOI:10.1007/s00227-003-1070-3 |
Dawson M N. 2005. Cyanea capillata is not a cosmopolitan jellyfish: morphological and molecular evidence for C. annaskala and C. rosea (Scyphozoa: Semaeostomeae: Cyaneidae) in south-eastern Australia. Invertebrate Systematics, 19(4): 361-370.
DOI:10.1071/IS03035 |
Dong Z J, Liu D Y, Keesing J K. 2010. Jellyfish blooms in China: dominant species, causes and consequences. Marine Pollution Bulletin, 60(7): 954-963.
DOI:10.1016/j.marpolbul.2010.04.022 |
Dong Z J, Liu Z Y, Liu D Y. 2015. Genetic characterization of the scyphozoan jellyfish Aurelia spp. in Chinese coastal waters using mitochondrial markers. Biochemical Systematics and Ecology, 60: 15-23.
DOI:10.1016/j.bse.2015.02.018 |
Dong Z J, Sun T T, Liu Q Q, Sun F F. 2017. High density aggregations of the Aurelia sp. 1 ephyrae in a Chinese coastal aquaculture pond. Aquatic Ecosystem Health & Management, 20(4): 465-471.
|
Ender A, Schierwater B. 2003. Placozoa are not derived cnidarians: evidence from molecular morphology. Molecular Biology and Evolution, 20(1): 130-134.
DOI:10.1093/molbev/msg018 |
Excoffier L, Lischer H E L. 2010. Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Molecular Ecology Resources, 10(3): 564-567.
DOI:10.1111/j.1755-0998.2010.02847.x |
Falkenhaug T. 2014. Review of jellyfish blooms in the Mediterranean and black Sea. Marine Biology Research, 10(10): 1038-1039.
DOI:10.1080/17451000.2014.880790 |
Hallerman E M. 2003. Population Genetics: Principles and Applications for Fisheries Scientists. American Fisheries Society, Bethesda. p. 261-290.
|
He J, Zheng L M, Zhang W J, Lin Y S. 2015. Life cycle reversal in Aurelia sp. 1 (Cnidaria, Scyphozoa). PLoS One, 10(12): e0145314.
DOI:10.1371/journal.pone.0145314 |
Ki J S, Hwang D S, Shin K, Yoon W D, Lim D, Kang Y S, Lee Y, Lee J S. 2008. Recent moon jelly (Aurelia sp. 1) blooms in Korean coastal waters suggest global expansion: examples inferred from mitochondrial COI and nuclear ITS-5.8S rDNA sequences. ICES Journal of Marine Science, 65(3): 443-452.
DOI:10.1093/icesjms/fsn018 |
Kumar S, Stecher G, Tamura K. 2016. MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Molecular Biology and Evolution, 33(7): 1870-1874.
DOI:10.1093/molbev/msw054 |
Lee P L M, Dawson M N, Neill S P, Robins P E, Houghton J D R, Doyle T K, Hays G C. 2013. Identification of genetically and oceanographically distinct blooms of jellyfish. Journal of the Royal Society Interface, 10(80): 20120920.
DOI:10.1098/rsif.2012.0920 |
Li Y L, Wang B, Wang W B, Dong J. 2016. Genetic diversity analysis of jellyfish Rhopilema esculentum in Liaodong bay based on the COI sequence. Fisheries Science, 35(4): 404-409.
(in Chinese with English abstract) DOI:10.16378/j.cnki.1003-1111.2016.04.016 |
Librado P, Rozas J. 2009. DnaSP V5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics, 25(11): 1451-1452.
DOI:10.1093/bioinformatics/btp187 |
Lucas C H. 2001. Reproduction and life history strategies of the common jellyfish, Aurelia aurita, in relation to its ambient environment. Hydrobiologia, 451(1-3): 229-246.
DOI:10.1023/A:1011836326717 |
Mills C E. 2001. Jellyfish blooms: are populations increasing globally in response to changing ocean conditions?. Hydrobiologia, 451(1-3): 55-68.
DOI:10.1023/A:1011888006302 |
Purcell J E. 2005. Climate effects on formation of jellyfish and ctenophore blooms: a review. Journal of the Marine Biological Association of the United Kingdom, 85(3): 461-476.
DOI:10.1017/S0025315405011409 |
Purcell J E, Baxter E J, Fuentes V L. 2013. Jellyfish as products and problems of aquaculture. In: Allan G, Burnell G eds. Advances in Aquaculture Hatchery Technology. Cambridge: Woodhead Publishing: 404-430, https://doi.org/10.1533/9780857097460.2.404.
|
Ramšak A, Stopar K, Malej A. 2012. Comparative phylogeography of meroplanktonic species, Aurelia spp. and Rhizostomapulmo (Cnidaria: Scyphozoa) in European seas. Hydrobiologia, 690(1): 69-80.
DOI:10.1007/s10750-012-1053-9 |
Richardson A J, Bakun A, Hays G C, Gibbons M J. 2009. The jellyfish joyride: causes, consequences and management responses to a more gelatinous future. Trends in Ecology & Evolution, 24(6): 312-322.
DOI:10.1016/j.tree.2009.01.010 |
Schroth W, Jarms G, Streit B, Schierwater B. 2002. Speciation and phylogeography in the cosmopolitan marine moon jelly, Aurelia sp. BMC Evolutionary Biology, 2: 1.
DOI:10.1186/1471-2148-2-1 |
Schroth W, Ender A, Schierwater B. 2005. Molecular biomarkers and adaptation to environmental stress in moon jelly (Aurelia spp. ). Marine Biotechnology, 7(5): 449-461.
DOI:10.1007/s10126-004-4095-9 |
Scorrano S, Aglieri G, Boero F, Dawson M N, Piraino S. 2017. Unmasking Aurelia species in the Mediterranean Sea: an integrative morphometric and molecular approach. Zoological Journal of the Linnean Society, 180(2): 243-267.
DOI:10.1111/zoj.12494 |
Stopar K, Ramšak A, Trontelj P, Malej A. 2010. Lack of genetic structure in the jellyfish Pelagia noctiluca (Cnidaria: Scyphozoa: Semaeostomeae) across European seas. Molecular Phylogenetics and Evolution, 57(1): 417-428.
DOI:10.1016/j.ympev.2010.07.004 |
Tajima F, Nei M. 1984. Estimation of evolutionary distance between nucleotide sequences. Molecular Biology and Evolution, 1(3): 269-285.
|
Toyokawa M, Furota T, Terazaki M. 2000. Life history and seasonal abundance of Aurelia aurita medusae in Tokyo Bay, Japan. Plankton Biology and Ecology, 47(1): 48-58.
|
Uye S I. 2008. Blooms of the giant jellyfish Nemopilema nomurai: a threat to the fisheries sustainability of the East Asian Marginal Seas. Plankton Benthos Res., 3(S1): 125-131.
DOI:10.3800/pbr.3.125 |
Uye S I. 2011. Human forcing of the copepod-fish-jellyfish triangular trophic relationship. Hydrobiologia, 666(1): 71-83.
DOI:10.1007/s10750-010-0208-9 |
Wang J Y, Yu Z G, Zhen Y, Mi T Z, Yao Q Z, Wang G S. 2012. Research advances in the effects of environmental factors on the growth and development of Aurelia spp. Chinese Journal of Applied Ecology, 23(11): 3207-3217.
(in Chinese with English abstract) DOI:10.13287/j.1001-9332.2012.0483 |
Wang J Y, Zhen Y, Wang G S, Mi T Z, Yu Z G. 2013. Molecular identification and detection of moon jellyfish (Aurelia sp.) based on partial sequencing of mitochondrial 16S rDNA and COI. Chinese Journal of Applied Ecology, 24(3): 847-852.
(in Chinese with English abstract) DOI:10.13287/j.1001-9332.2013.0239 |
Whitmore D H, Thai T H, Craft C M. 1994. The largemouth bass cytochrome b gene. J. Fish Biol., 44(4): 637-645.
DOI:10.1111/j.1095-8649.1994.tb01240.x |
 2021, Vol. 39
2021, Vol. 39


