Institute of Oceanology, Chinese Academy of Sciences
Article Information
- HAO Ya, GUAN Chen, ZHAO Xinyu, QU Tongfei, TANG Xuexi, WANG Ying
- Investigation of the decline of Ulva prolifera in the Subei Shoal and Qingdao based on physiological changes
- Journal of Oceanology and Limnology, 39(3): 918-927
- http://dx.doi.org/10.1007/s00343-020-0140-4
Article History
- Received Mar. 30, 2020
- accepted in principle Apr. 27, 2020
- accepted for publication May. 24, 2020
2 Laboratory for Marine Ecology and Environmental Science, Qingdao National Laboratory for Marine Science and Technology, Qingdao 266071, China
Since 2007, Yellow Sea green tide occurred in coastal China almost every year, and large-scale green tides caused considerable negative impact on offshore aquaculture, shipping, and tourism, resulting in many economic losses (Liu et al., 2009; Hu et al., 2010; Pang et al., 2010; Zhang et al., 2014, 2019). Ulva prolifera has been identified as the causative macroalgal species based on morphological taxonomy and molecular biology techniques (Shen et al., 2012; Han et al., 2013; Zhao et al., 2013). Combining the results of a number of previous studies and satellite remote-sensing data (Zhang et al., 2014; Bao et al., 2015), it can be found that the green tide of U. prolifera was originated from Pyropia aquaculture rafts in the Subei Shoal prior to the formation of large-scale green tides in the southern Yellow Sea (Liu et al., 2010, 2013; Fan et al., 2012; Huo et al., 2016).
In recent years, the Pyropia aquaculture rafts, which have continuously expanded to over 20 000 ha, were the adherent base for the growth of floating green macroalgae (Zhu and Chang, 2001; Song et al., 2015). Under the influence of natural and human factors, U. prolifera falling off the rafts has become an important source of floating green macroalgae. The green tide of U. prolifera usually initiated in the Subei Shoal about 2 weeks after the harvest of Pyropia, and a large amount of U. prolifera was gradually exported from the Subei Shoal to the southern Yellow Sea. The algal mass drifted northward under the driving forces of seasonal winds and winddriven surface currents (Keesing et al., 2011; Fu et al., 2019). The green tide of U. prolifera continued to expand in the course of drifting north, and eventually developed into a large-scale green tide ecological disaster (Cui et al., 2012; Liu and Zhou, 2018). Because of the particular geographic location of the Shandong Peninsula, massive amounts of U. prolifera were dominated the coast of the peninsula (Qiao et al., 2011). In the Subei Shoal, which is the area of origin of the green tide, decline of the U. prolifera population generally occurs in July, and the population of U. prolifera that migrated to the nearshore of Qingdao generally disappears in August (Fig. 1).

|
| Fig.1 The occurrence and decline of the Yellow Sea green tide |
A considerable amount of research has been carried out on the occurrence and development of the green tide, but relatively little attention has been paid to the decline of the green tide. In this study, we focused on the relationship between environmental factors and the decline of the U. prolifera population by comparing the growth rate, photosynthetic capacity, lipid membrane peroxidation, and tissue structure of U. prolifera samples in the outbreak and declinedecline stages. The environmental factors in each stage were measured, followed by correlation analysis between the environmental factors and the physiological indicators of the U. prolifera samples. The correlation analysis results were used to determine the key environment factors leading to the decline of the U. prolifera population. We aimed to explore the ecological reasons for the decline of the U. prolifera green tide and provide ecological data for the prevention of the green tide in the South Yellow Sea of China.
2 MATERIAL AND METHOD 2.1 Sample collectionFree-floating U. prolifera were collected in Xiaoyangkou, Rudong, Jiangsu Province on May 10 and July 10, and Taipingjiao, Qingdao, Shandong Province on August 10 (Fig. 2). Samples collected in July and August were used as the research object for the decline of the green tide, and the samples collected during the early stage of the green tide outbreak in May were used as the comparison. Complete algal U. prolifera was collected at the sampling site and rinsed with sterile seawater to remove surface impurities. Field experiments were performed at the sampling site, which included the measurement of relative growth rate (RGR) and photosynthetic fluorescence parameters, and the preliminary fixation of transmission electronic microscopy (TEM) tissue sections. The remaining portion of U. prolifera samples was stored in a -80-℃ refrigerator for subsequent chlorophyll and lipid membrane peroxidation analysis.

|
| Fig.2 Sites of Ulva prolifera samples collection in the Subei Shoal off Rudong, Jiangsu, and the coast of Qingdao, Shandong a. the sampling site of U. prolifera floating to Qingdao in the green tide recession; b. the sampling site of early floating U. prolifera; c. the sampling site of U. prolifera that stayed in Rudong in the green tide recession. The area enclosed by a dotted line indicates the main rafting aquaculture area of Porphyra located in the Subei Shoal. |
Various environmental parameters were measured during the sample collection. Photosynthetically active radiation (PAR) was measured using a dual radiation meter (Zhuochuan, China). A standard thermometer (Fuyang Instrument Co., China) was used to measure temperature. Salinity was measured using a salinity meter (Lichen, China), and pH was measured using a portable pH meter (Mettler Toledo, Switzerland).
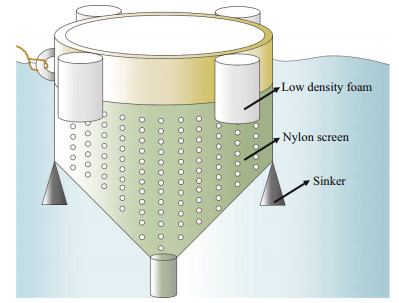
2.2 Growth rateRelative growth rates (RGRs) of U. prolifera samples were tested at the sampling site in a sevenday period. The U. prolifera samples with initial biomass of 20 g were cultured in mesocosm (Fig. 3). The culture conditions were consistent with the environmental factors at the corresponding times (Table 1). Algal material was reweighed after seven days, and relative growth rates were calculated as follows (Wu et al., 2018):
|
| Fig.3 Schematic diagram of the mesocosm experiment for measurement of relative growth rate |
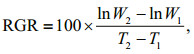 (1)
(1)where W1 is the initial fresh weight and W2 is the fresh weight after Δt (T2–T1) days.
2.3 Pigments extractionSamples were ground to powder in liquid nitrogen and placed in a centrifuge tube. A precooling extraction buffer of 90% (v/v) acetone buffer (5 mL) was used to extract the pigments at 4 ℃ in darkness. The mixture was then centrifuged at 10 000 r/min and 4 ℃, and the supernatant was filtered and analyzed spectrophotometrically (TU-1800, PERSEE, China). The chlorophyll a (Chl a), chlorophyll b (Chl b), and total chlorophyll (Chl) contents were calculated as follows (Arnon, 1949):
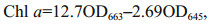 (2)
(2)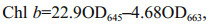 (3)
(3) (4)
(4)A Dual-PAM-100 fluorometer (Walz, Germany) was used to measure the photosynthetic performance of U. prolifera samples. Samples were acclimated in the dark for 20 min prior to measurements. The settings of the Dual-PAM-F fluorometer followed previous research in this laboratory (Zhao et al., 2016). The maximum quantum yield (Fv/Fm) of photosystem II (PSII) can be calculated by the equation:
 (5)
(5)where Fm is the maximum fluorescence for darkadapted tissue, F0 is the minimum fluorescence for dark adapted tissue, and Fv is the maximum fluorescence. YII is the effective quantum yield of PSII, and can be used to evaluate photosynthetic activity in the thalli.
The rapid light response curves (RLCs) were measured after the induction curve experiment. When the RLCs were measured, the samples were exposed to a light intensity gradient (PAR: 0, 25, 40, 56, 73, 129, 169, 276, 342, 534, 828, and 1 597 μmol photons/(m2·s)). The RLCs were fitted using Platt's empirical equation to obtain the following parameters: rETRmax (maximum relative electron transport rate), α (photosynthetic rate in the light-limited region of the RLCs), β (photoinhibition parameter), and Ek (minimum saturating irradiance) (Platt et al., 1980).
2.5 Analysis of lipid peroxidationThe level of lipid peroxidation was studied as malondialdehyde (MDA) formation (Buege and Aust, 1978). Total levels of soluble protein of the crude extract for antioxidant enzyme activities ((g protein)/mL or (g pro)/mL) were determined using a commercial protein assay kit (Nanjing Jiancheng, China). The MDA content was determined using a commercial assay kit (Nanjing Jiancheng, China) and results are expressed as n·mol MDA per mg of total soluble protein (nmol MDA/(mg pro)).
2.6 Transmission electron microscope observationFor TEM analysis, samples of 1-mm length were fixed in 5-mL 2.5% glutaraldehyde and 2% paraformaldehyde in 0.01-mol/L phosphate buffer saline (PBS) (pH 7.2) for 24 h at 4 ℃ (Farias et al., 2017). After initial fixation, samples were sent to Servicebio technology company (Wuhan, China) for subsequent preparation, block cutting and transmission electron microscopy analysis. The specimens were observed with a transmission electronic microscopy (JEOL JEM-1011, Japan).
2.7 Data analysisAll data were obtained by analyzing three biological replicates and displayed as mean±standard deviation (SD). The environmental parameters and laboratory data were tested using one-way ANOVA and a Fisher LSD mean comparison. Bivariate Pearson's correlation analysis was performed to assess the relationships of the physiological indices of U. prolifera and environmental factors. The data were analyzed using IBM SPSS Statistics 22 (SPSS Inc., USA), and P-values of less than 0.05 were considered statistically significant.
3 RESULT 3.1 Environmental factorThe environmental parameters of the sampling locations are shown in Table 1. The survey data indicate that no significant difference was detected in the parameters of salinity or pH between the three stages (one-way ANOVA; P < 0.05). The temperature of Rudong in July and that of Qingdao in August were significantly higher than that of Rudong in May (oneway ANOVA; P < 0.05). The PAR value of Qingdao in August was significantly higher than those of Rudong in May and July (one-way ANOVA; P < 0.05).
3.2 Growth rateThe RGR of U. prolifera in three stages are shown in Fig. 4. The culture conditions of the samples collected in each phase were consistent with the onsite environmental conditions of the corresponding month (Table 1). The growth rate of U. prolifera in May was the highest, reaching 17.13%±1.29%. The samples of U. prolifera in July and August showed negative growth, and the negative growth rate of U. prolifera in August reached -17.54%±6.2%, which was significantly higher than that in July (one-way ANOVA; P < 0.05).

|
| Fig.4 Relative growth rate (RGR) of U. prolifera samples collected in three stages Values are means±SD (n=3), and different letters indicate significantly difference (one-way ANOVA, P < 0.05). RD: Rudong; QD: Qingdao, the same below. |
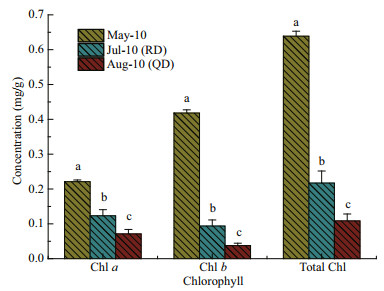
The results for pigment content of U. prolifera samples in three stages are shown in Fig. 5. The Chl-a, Chl-b, and total Chl concentrations of U. prolifera samples collected in Rudong in May were significantly higher than those of Rudong in July and Qingdao in August (one-way ANOVA; P < 0.05). The Chl-a, Chl-b, and total Chl concentrations of the U. prolifera samples from Qingdao in August were all clearly the lowest of the three stages (one-way ANOVA; P < 0.05).
|
| Fig.5 Photosynthetic pigment content of U. prolifera samples in three stages Data are mg/g fresh weight for each pigment. Values are means±SD (n=3), and different letters indicate significantly difference (oneway ANOVA, P < 0.05). |
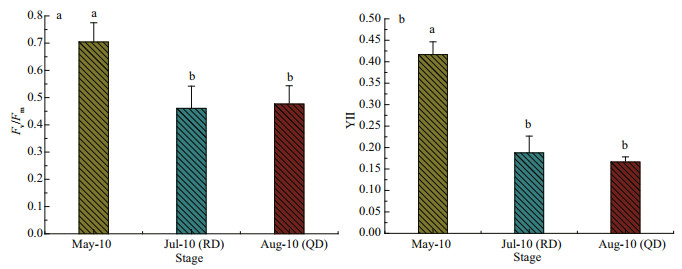
Figure 6a shows the difference of Fv/Fm in the thalli of U. prolifera in the three phases. The Fv/Fm value of U. prolifera samples from Rudong in May was significantly higher than those from Rudong in July and Qingdao in August (Fig. 6a). Similar results can be seen in the effective quantum yields of PSII (YII) (Fig. 6b), and the YII value for U. prolifera samples from Rudong in May was noticeably higher than those from Rudong in July and Qingdao in August.
|
| Fig.6 Results of induction curve parameters measurement a. the value of Fv/Fm; b. the effective quantum yield of PSII (YII). Values are means±SD (n=3), and different letters indicate significantly difference (one-way, ANOVA, P < 0.05). |
The results of the RLCs are shown in Fig. 7. The RLCs of the samples from Rudong in May show a linear rise at the beginning followed by stabilization as the intensity of the light increases. The convexity of the curves was noticeably lower for both samples from Rudong in July and from Qingdao in August compared with that from Rudong in May (one-way ANOVA; P < 0.05).

|
| Fig.7 Mean relative electron transport rate (rETR) of rapid light response curves (RLCs) of Ulva prolifera in three stages Values are means±SD (n=3). |
A further conclusion can be drawn based on the results of the RLCs. Table 2 shows that the values of the parameters rETRmax and Ek of U. prolifera samples collected in Rudong in May were significantly higher than those of Rudong in July and Qingdao in August (one-way ANOVA; P < 0.05). The β values of samples collected in Rudong in May were noticeably lower than those from both Rudong in July and Qingdao in August (one-way ANOVA; P < 0.05). We observed that the value of α from samples collected in Rudong in July was significantly lower than the other two stages (one-way ANOVA; P < 0.05).
Figure 8 shows the contents of MDA. The MDA contents of the samples were noticeably higher for samples both from Rudong in July and from Qingdao in August compared with those from Rudong in May, and the MDA contents of the samples from Qingdao in August were significantly higher than those from Rudong in July (one-way ANOVA; P < 0.05).

|
| Fig.8 The content of MDA from Ulva prolifera samples collected in three stages Values are means±SD (n=3), and different superscripted letters indicate significantly difference (one-way ANOVA, P < 0.05). |
The transmission electronic microscopy results are shown in Fig. 9. Transmission electronic micrographs of U. prolifera samples from the outbreak of the green tide in May showed that the cell structure was intact, and several protein cores and starch grains were surrounded by chloroplasts inside and the inner edge of the cell wall was smooth (Fig. 9a). Compared with the U. prolifera samples from the green tide outbreak phase, transmission electronic micrographs of U. prolifera samples from the decline phase of the green tide, which included both the samples from Rudong in July and those from Qingdao in August, showed that the number of chloroplasts in the cells was significantly reduced; the nucleus, protein nucleus, and starch granules disappeared; the inner edge of the cell wall was ruptured and deformed; and lipid droplets increased (Fig. 9b & c). Overall, there was significant cellular structure damage in the U. prolifera samples of the decline period of the green tide.

|
| Fig.9 Ultrastructure of Ulva prolifera samples in the three stages revealed in transmission electronic microscopy a. samples of May; b. samples of July; c. samples of August. C: chloroplast; CW: cell wall; L: lipids; N: nucleus; n: nucleolus; P: pyrenoid; rER: rough endoplasmic reticulum; S: starch granules; V: vacuole (black hollow arrows indicate ruptured and deformed cell walls). |
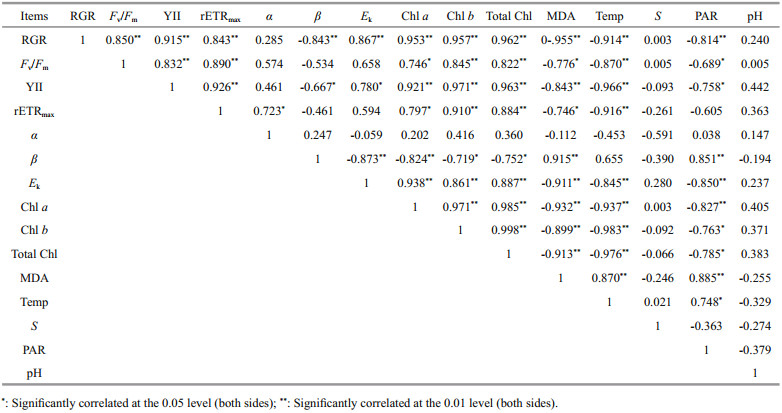
Table 3 shows that the RGR of U. prolifera was significantly positively correlated with the values of Fv/Fm, YII, rETRmax, Ek, and chlorophyll content, and significantly negatively correlated with the photoinhibition parameter β, MDA content, ambient temperature, and PAR intensity (one-way ANOVA; P < 0.05). MDA content was significantly positively correlated with ambient temperature and PAR intensity (one-way ANOVA; P < 0.05). The photoinhibition parameter β and PAR intensity showed a significant positive correlation (one-way ANOVA; P < 0.05). The correlation between salinity and each physiological index of U. prolifera was not significant (one-way ANOVA; P>0.05). In addition, there was no significant correlation between the pH of seawater in the survey area and each physiological index of U. prolifera (one-way ANOVA; P>0.05).
|
We collected samples of U. prolifera from the outbreak of the green tide in the Subei Shoal in May and samples of U. prolifera from the decline phase of the green tide in the Subei Shoal in July, as well as from Qingdao in August. The growth rate, photosynthetic capacity, lipid membrane peroxidation, and tissue ultrastructure of U. prolifera samples in the decline phase were studied.
Through experiments, we could conclude that the photosynthetic ability and chlorophyll content of U. prolifera in the decline period were significantly lower than those in the outbreak phase. These physiological changes adversely affected the material synthesis process of U. prolifera, and the growth of the U. prolifera population slowed compared with the rapid population growth in the outbreak stage. In the decline phase, the MDA content of U. prolifera was significantly higher than that in the outbreak phase, and the lipid membrane peroxidation was severely aggravated, which led to the loss of structural integrity of the tissue cells. Therefore, the intensification of lipid membrane peroxidation and further cell necrosis were the main internal causes of the decline of the U. prolifera population.
4.2 External drivers of U. prolifera population declinePrevious research confirmed that algal growth rates are regulated by environmental factors such as temperature, irradiance, and nutrient level (Taylor et al., 2001; Kim et al., 2016; Wu et al., 2018). High temperatures would inhibit the growth of U. prolifera (Wu et al., 2018). Through the correlation analysis between the physiological indicators of U. prolifera samples and environmental factors of the three phases, we could conclude that high temperature was the most important external environmental factor leading to inefficient photosynthesis, increased lipid peroxidation, and structural damage of U. prolifera. Therefore, we conclude that the long-term and sustained high temperature conditions were the main cause of the decline of the U. prolifera population. The PAR intensity in Qingdao in August was higher compared with that in Rudong in July, which further deepened the lipid peroxidation level of U. prolifera and led to higher photoinhibition (Table 2), and eventually presented as a higher negative growth rate than that in Rudong in July.
Given the relatively stable salinity and pH conditions in the space-time span of the Yellow Sea, their correlation with the changes of physiological process parameters of U. prolifera was not significant (Table 3). The spatio-temporal variation of nutrient content, seawater transparency and other marine physicochemical indicators in the survey area and their effects on the structure of the U. prolifera population need further field investigation and statistical verification.
4.3 Analysis of U. prolifera population decline in the Subei Shoal and offshore of QingdaoAs the origin area of the South Yellow Sea green tide, there was a large amount of biomass of the primordial population of U. prolifera in the Subei Shoal from April to June. A large amount of U. prolifera was exported from the Subei Shoal to the South Yellow Sea under the dual effects of seasonal winds and wind-driven surface currents (Keesing et al., 2011; Liu and Zhou, 2018; Fu et al., 2019). The temperature of seawater increased in mid-July (Fan et al., 2015), and the environmental stress on the U. prolifera population caused by persistent high temperature increased. The chlorophyll content and photosynthetic efficiency decreased significantly (Figs. 5–7), and the tissue cells ruptured (Fig. 9), which ultimately led to the decline of the U. prolifera population.
Because of the higher latitude, the ambient temperature rising in Qingdao offshore occurred later than that in the Subei Shoal, and the U. prolifera population that constituted the green tide drifted over a long distance before reaching offshore of Qingdao. The harsh environment had a negative impact on the physiological activities and organizational structure of U. prolifera during the drift course (Mou et al., 2013; Zhao et al., 2016). The ambient temperature and the light intensity offshore of Qingdao increased in August, and as a result, the external environmental stress experienced by the U. prolifera population blocked on the coast of Qingdao was intensified.
The U. prolifera samples of this phase showed low photosynthetic efficiency and photoinhibition, and exacerbated lipid membrane peroxidation, cell necrosis. The seawater temperature in the Subei Shoal increased earlier than that offshore of the Shandong Peninsula because of the difference in latitude. Therefore, the decline of the U. prolifera population in the Subei Shoal was earlier than that off Qingdao. Macroscopically, the U. prolifera population that constitutes the green tide of the South Yellow Sea had lost the supplementary source of biomass after the decline of the U. prolifera population remaining in the Subei Shoal in mid-July. Affected by topography, the U. prolifera population was stranded on the coast of the Shandong Peninsula during northward drifting and eventually died out because of the high temperature and high intensity radiation.
5 CONCLUSIONThrough determination of the physiological parameters of U. prolifera samples in the outbreak phase and the decline phase, and combined multiparameter correlation analysis of environmental factors, we conclude that the most important environmental factor leading to the decline of the U. prolifera population was temperature. High ambient temperature could lead to intensification of the lipid peroxidation of U. prolifera and reduce photosynthesis efficiency and chlorophyll content, which ultimately led to negative growth and decline of the U. prolifera population. In addition, the PAR intensity was significantly positively correlated with the photoinhibition parameter. Excessive PAR intensity could cause photoinhibition during the photosynthetic process, which was also an important environmental factor leading to the decline of the U. prolifera population. The salinity and pH of the seawater were not inducing factors for the decline of the U. prolifera population. In conclusion, it can be inferred from this experiment that the decline of the U. prolifera population remaining in the Subei Shoal was mainly attributed to the increase of temperature, and that this local decline in the Subei Shoal was earlier than that of the U. prolifera population blocked offshore of Qingdao because of the difference in latitude between the two places. In addition, the impact of this longdistance drift process on the physiological activity and population dynamics of U. prolifera requires further research and demonstration.
6 DATA AVAILABILITY STATEMENTThe datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Arnon D I. 1949. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta Vulgaris. Plant Physiology, 24(1): 1-15.
DOI:10.1104/pp.24.1.1 |
Bao M, Guan W B, Yang Y, Cao Z Y, Chen Q. 2015. Drifting trajectories of green algae in the western Yellow Sea during the spring and summer of 2012. Estuarine, Coastal and Shelf Science, 163: 9-16.
DOI:10.1016/j.ecss.2015.02.009 |
Buege J A, Aust S D. 1978. Microsomal lipid peroxidation. Methods in Enzymology, 52: 302-310.
DOI:10.1016/S0076-6879(78)52032-6 |
Cui T W, Zhang J, Sun L E, Jia Y J, Zhao W J, Wang Z L, Meng J M. 2012. Satellite monitoring of massive green macroalgae bloom (GMB): Imaging ability comparison of multi-source data and drifting velocity estimation. International Journal of Remote Sensing, 33(17): 5 513-5 527.
DOI:10.1080/01431161.2012.663112 |
Fan S L, Fu M Z, Li Y, Wang Z L, Fang S, Jiang M J, Wang H P, Sun P, Qu P. 2012. Origin and development of Huanghai (Yellow) Sea green-tides in 2009 and 2010. Acta Oceanologica Sinica, 34(6): 187-194.
(in Chinese) |
Fan S L, Fu M Z, Wang Z L, Zhang X L, Song W, Li Y, Liu G X, Shi X Y, Wang X N, Zhu M Y. 2015. Temporal variation of green macroalgal assemblage on Porphyra aquaculture rafts in the Subei Shoal, China. Estuarine, Coastal and Shelf Science, 163: 23-28.
DOI:10.1016/j.ecss.2015.03.016 |
Farias D R, Simioni C, Poltronieri E, Bouzon Z L, Macleod C K. 2017. Fine-tuning transmission electron microscopy methods to evaluate the cellular architecture of Ulvacean seaweeds (Chlorophyta). Micron, 96: 48-56.
DOI:10.1016/j.micron.2017.02.003 |
Fu M Z, Fan S L, Wang Z L, Song W, Sun K M, Han H B, Xiao J, Shen S D. 2019. Buoyancy potential of dominant green macroalgal species in the Yellow Sea's green tides, China. Marine Pollution Bulletin, 140: 301-307.
DOI:10.1016/j.marpolbul.2019.01.056 |
Han W, Chen L P, Zhang J H, Tian X L, Hua L, He Q, Huo Y Z, Yu K F, Shi D J, Ma J H, He P M. 2013. Seasonal variation of dominant free-floating and attached Ulva species in Rudong coastal area, China. Harmful Algae, 28: 46-54.
DOI:10.1016/j.hal.2013.05.018 |
Hu C M, Li D Q, Chen C S, Ge J Z, Muller-Karger F E, Liu J P, Yu F, He M X. 2010. On the recurrent Ulva prolifera blooms in the Yellow Sea and East China Sea. Journal of Geophysical Research, 115(C5): C05017.
DOI:10.1029/2009JC005561 |
Huo Y Z, Han H B, Hua L, Wei Z L, Yu K F, Shi H H, Kim J K, Yarish C, He P M. 2016. Tracing the origin of green macroalgal blooms based on the large scale spatiotemporal distribution of Ulva microscopic propagules and settled mature Ulva vegetative thalli in coastal regions of the Yellow Sea, China. Harmful Algae, 59: 91-99.
DOI:10.1016/j.hal.2016.09.005 |
Keesing J K, Liu D Y, Fearns P, Garcia R. 2011. Inter- and intra-annual patterns of Ulva prolifera green tides in the Yellow Sea during 2007-2009, their origin and relationship to the expansion of coastal seaweed aquaculture in China. Marine Pollution Bulletin, 62(6): 1 169-1 182.
DOI:10.1016/j.marpolbul.2011.03.040 |
Kim J K, Yarish C, Pereira R. 2016. Tolerances to hypo-osmotic and temperature stresses in native and invasive species of Gracilaria (Rhodophyta). Phycologia, 55(3): 257-264.
DOI:10.2216/15-90.1 |
Liu D Y, Keesing J K, Dong Z J, Zhen Y, Di B P, Shi Y J, Fearns P, Shi P. 2010. Recurrence of the world's largest green-tide in 2009 in Yellow Sea, China: Porphyra yezoensis aquaculture rafts confirmed as nursery for macroalgal blooms. Marine Pollution Bulletin, 60(9): 1 423-1 432.
DOI:10.1016/j.marpolbul.2010.05.015 |
Liu D Y, Keesing J K, He P M, Wang Z L, Shi Y J, Wang Y J. 2013. The world's largest macroalgal bloom in the Yellow Sea, China: formation and implications. Estuarine, Coastal and Shelf Science, 129: 2-10.
DOI:10.1016/j.ecss.2013.05.021 |
Liu D Y, Keesing J K, Xing Q G, Shi P. 2009. World's largest macroalgal bloom caused by expansion of seaweed aquaculture in China. Marine Pollution Bulletin, 58(6): 888-895.
DOI:10.1016/j.marpolbul.2009.01.013 |
Liu D Y, Zhou M J. 2018. Green tides of the Yellow Sea: Massive free-floating blooms of Ulva prolifera. In: Glibert P M, Berdalet E, Burford M A, Pitcher G C, Zhou M L eds. Global Ecology and Oceanography of Harmful Algal Blooms. Springer, Cham, Germany. p. 317–326, https: //doi.org/10.1007/978-3-319-70069-4_16
|
Mou S, Zhang X, Dong M, Fan X, Xu J, Cao S, Xu D, Wang W, Ye N. 2013. Photoprotection in the green tidal alga Ulva prolifera: Role of LHCSR and PsbS proteins in response to high light stress. Plant Biology, 15(6): 1 033-1 039.
DOI:10.1111/j.1438-8677.2012.00712.x |
Pang S J, Liu F, Shan T F, Shan T F, Xu N, Zhang Z H, Gao S Q, Chopin T, Sun S. 2010. Tracking the algal origin of the Ulva bloom in the Yellow Sea by a combination of molecular, morphological and physiological analyses. Marine Environmental Research, 69(4): 207-215.
DOI:10.1016/j.marenvres.2009.10.007 |
Platt T, Gallegos C, Harrison W G. 1980. Photoinhibition of photosynthesis in natural assemblages of marine phytoplankton. Journal of Marine Research, 38(4): 687-701.
|
Qiao F L, Wang G S, Lü X G, Dai D J. 2011. Drift characteristics of green macroalgae in the Yellow Sea in 2008 and 2010. Chinese Science Bulletin, 56(21): 2 236-2 242.
DOI:10.1007/s11434-011-4551-7 |
Shen Q, Li H Y, Li Y, Wang Z L, Liu J S, Yang W D. 2012. Molecular identification of green algae from the rafts based infrastructure of Porphyra yezoensis. Marine Pollution Bulletin, 64(10): 2 077-2 082.
DOI:10.1016/j.marpolbul.2012.07.021 |
Song W, Li Y, Fang S, Wang Z L, Xiao J, Li R X, Fu M Z, Zhu M Y, Zhang X L. 2015. Temporal and spatial distributions of green algae micro-propagules in the coastal waters of the Subei Shoal, China. Estuarine, Coastal and Shelf Science, 163: 29-35.
DOI:10.1016/j.ecss.2014.08.006 |
Taylor R, Fletcher R L, Raven J A. 2001. Preliminary studies on the growth of selected 'green tide' algae in laboratory culture: effects of irradiance, temperature, salinity and nutrients on growth rate. Botanica Marina, 44(4): 327-336.
DOI:10.1515/BOT.2001.042 |
Wu H L, Gao G, Zhong Z H, Li X S, Xu J T. 2018. Physiological acclimation of the green tidal alga Ulva prolifera to a fast-changing environment. Marine Environmental Research, 137: 1-7.
DOI:10.1016/j.marenvres.2018.02.018 |
Zhang J H, Huo Y Z, Wu H L, Yu K F, Kim J K, Yarish C, Qin Y T, Liu C C, Xu R, He P M. 2014. The origin of the Ulva macroalgal blooms in the Yellow Sea in 2013. Marine Pollution Bulletin, 89(1-2): 276-283.
DOI:10.1016/j.marpolbul.2014.09.049 |
Zhang J H, Shi J T, Gao S, Huo Y Z, Cui J J, Shen H, Liu G Y, He P M. 2019. Annual patterns of macroalgal blooms in the Yellow Sea during 2007-2017. PLoS One, 14(1): e0210460.
DOI:10.1371/journal.pone.0210460 |
Zhao J, Jiang P, Liu Z Y, Wei W, Lin H Z, Li F C, Wang J F, Qin S. 2013. The yellow sea green tides were dominated by one species, Ulva (Enteromorpha) prolifera, from 2007 to 2011. Chinese Science Bulletin, 58(19): 2 298-2 302.
DOI:10.1007/s11434-012-5441-3 |
Zhao X Y, Tang X X, Zhang H X, Qu T F, Wang Y. 2016. Photosynthetic adaptation strategy of Ulva prolifera floating on the sea surface to environmental changes. Plant Physiology and Biochemistry, 107: 116-125.
DOI:10.1016/j.plaphy.2016.05.036 |
Zhu Y R, Chang R F. 2001. Sediment dynamics study on the origin of the radial sand ridges in the southern Yellow Sea. Studia Marina Sinica, 43(1): 38-50.
(in Chinese) |
 2021, Vol. 39
2021, Vol. 39




